Volume 25, Number 2—February 2019
Research
Echinococcus multilocularis Infection, Southern Ontario, Canada
Cite This Article
Citation for Media
Abstract
Alveolar echinococcosis, the disease caused by infection with the intermediate stage of the Echinococcus multilocularis tapeworm, is typically fatal in humans and dogs when left untreated. Since 2012, alveolar echinococcosis has been diagnosed in 5 dogs, 3 lemurs, and 1 chipmunk in southern Ontario, Canada, a region previously considered free of these tapeworms. Because of human and animal health concerns, we estimated prevalence of infection in wild canids across southern Ontario. During 2015–2017, we collected fecal samples from 460 wild canids (416 coyotes, 44 foxes) during postmortem examination and analyzed them by using a semiautomated magnetic capture probe DNA extraction and real-time PCR method for E. multilocularis DNA. Surprisingly, 23% (95% CI 20%–27%) of samples tested positive. By using a spatial scan test, we identified an infection cluster (relative risk 2.26; p = 0.002) in the western-central region of the province. The cluster encompasses areas of dense human population, suggesting zoonotic transmission.
Alveolar echinococcosis (AE) is a chronic infection caused by the larval stage of the Echinococcus multilocularis tapeworm and commonly manifests within the liver. In humans and dogs, AE is typically fatal when left untreated. The E. multilocularis tapeworm has a wide distribution in the Northern Hemisphere, including extensive endemic regions in North America, Europe, and Asia (1), and is usually maintained in a life cycle that involves 2 mammalian hosts. Wild canids (e.g., foxes and coyotes), dogs, and (less commonly) cats act as definitive hosts, which harbor adult parasites in the small intestine without apparent clinical disease. Once mature, adult parasites release eggs, which are shed in the definitive host’s feces. Intermediate hosts (e.g., small rodents) acquire the larval stage by ingestion of infective eggs in the environment. The life cycle is completed when a definitive host consumes an intermediate host containing the larval stage. Humans and dogs can experience AE when eggs of the parasite are consumed.
In humans, AE is characterized by a lengthy clinical incubation period of 5–15 years, during which the larval stage typically proliferates within the liver, behaving similarly to infiltrative hepatic neoplasia (2). Humans with clinical AE cases typically experience cholestatic jaundice, abdominal pain, fatigue, and weight loss (3). The preferred treatment is complete excision of parasitic tissue and radical resection of host tissue, depending on the site and size of the lesion, presence of metastases, and patient comorbidities (4). Benzimidazole chemotherapy is initiated at the time of diagnosis (5). In cases of total surgical resection, treatment is continued for a minimum of 2 years to reduce the likelihood of relapse (5). In case-patients who are not surgical candidates, chemotherapy treatment might be prescribed indefinitely to slow the progression of disease (6). Historically, in patients from Alaska, France, and Germany, the average survival rate 10 years after diagnosis was 29% when left untreated (7). The advent of benzimidazole chemotherapy has increased the 10-year survival rate to ≈80% (8).
Red foxes (Vulpes vulpes) are commonly the primary definitive host for E. multilocularis tapeworms in Europe and North America (1). More recently, studies have shown that coyotes (Canis latrans) also maintain the parasite in North America (9,10). This development is important because coyotes can expedite the spread of E. multilocularis tapeworms because they have larger home ranges compared with red foxes (11).
The area of endemicity of the E. multilocularis tapeworm in North America was thought to include 2 distinct regions: the north tundra zone and the north central region. The north tundra zone begins on the west coast of Alaska and extends north and east to occupy most of the Canadian Arctic; the distribution is consistent with that of the Arctic fox (10,12). The north central region includes the southern portions of the Canada provinces of Alberta, Saskatchewan, and Manitoba, along with 13 neighboring US states (North Dakota, South Dakota, Iowa, Minnesota, Montana, Wyoming, Nebraska, Illinois, Wisconsin, Indiana, Ohio, Missouri, and Michigan) (9,10,13). Recent reports suggest that the distribution is expanding or perhaps is wider than previously thought; for example, in 2009, a dog from the Quesnel region in British Columbia, with no travel history outside of that province, was diagnosed with AE (14). A subsequent study determined that ≈33% of wild canids in that region were infected with E. multilocularis tapeworms, suggesting a new endemic area (15).
Before 2012, Ontario was considered free of E. multilocularis tapeworms. Since then, AE has been diagnosed in 5 dogs, 3 privately owned lemurs (Lemur catta), and a wild-caught eastern chipmunk (Tamias striatus) in the region surrounding the western shores of Lake Ontario in southern Ontario (16–21; A.S. Peregrine, unpub. data). The primary organ of involvement was the liver in all except 1 case, which involved only a subcutaneous lesion. To the authors’ knowledge, only 1 of the aforementioned dogs had traveled outside this region; the other animals must have acquired the infection locally, probably as a result of ingestion of canid feces containing E. multilocularis eggs. Canine AE is a rare disease that most likely occurs when dogs ingest a substantial number of eggs (22). Collectively, these cases suggest that parts of southern Ontario have substantial levels of infection among wild canids.
Although southern Ontario encompasses an extensive geographic area (136,907 km2), it is the most densely populated region of the province, with ≈12 million residents (23). At the time of the aforementioned cases of AE in animals, human AE was not a disease of public health importance (i.e., it was not reportable) in Ontario; therefore, whether autochthonous human cases were occurring in the province was unknown. Nevertheless, the presence of E. multilocularis tapeworms represented a potentially serious threat to human and animal health.
In light of these developments, we sensed an urgent need to accurately define areas in southern Ontario where the E. multilocularis tapeworm occurs and to identify the areas of highest risk within this region. We therefore conducted a study to estimate the prevalence and geographic distribution of E. multilocularis infection among foxes and coyotes across southern Ontario.
Carcass Collection and Necropsy
We obtained wild canid carcasses through collaboration with licensed hunters and trappers and the Ontario Ministry of Natural Resources and Forestry. We disseminated information about the project to hunter and trapper groups in southern Ontario that ordinarily harvest coyotes and foxes for their pelts. Submission of a carcass was contingent on provision of the geographic location of origin of the harvested carcass. We obtained carcasses over 2 collection periods: November 1, 2015–August 10, 2016, and August 30, 2016–March 27, 2017. During each collection period, hunters and trappers were limited to 10 carcass submissions of each species. No animals were killed for the purpose of this study. We submitted frozen and fresh carcasses for a limited postmortem examination. We removed the large intestine from each carcass and stored it at −80°C for a minimum of 5 days to eliminate infectivity of the eggs (24), after which we collected 2 aliquots of rectal fecal material (3 g each) from each intestinal sample and stored them at −20°C before analysis.
Magnetic Capture Probe DNA Extraction and Real-Time PCR
At the end of each collection period, we sent fecal samples to the Section for Microbiology at the National Veterinary Institute, Uppsala, Sweden, for analysis using a semiautomated magnetic capture probe DNA extraction and real-time hydrolysis PCR (MC-PCR) method for the presence of E. multilocularis DNA (25). Compared with the sedimentation and counting technique, which is typically considered the reference standard for the diagnosis of infection in wild canids (26), the MC-PCR method is less labor intensive and is well suited for processing large numbers of samples (25). When applied to foxes, MC-PCR has an overall sensitivity of 88% (81% with <100 parasites and 96% sensitivity with >100 parasites) and a minimum specificity of 99.9% (25,27). For each batch of extracted samples that was examined, we used 1 positive and 2 negative controls to validate the extraction process and real-time PCR; the positive control was a known positive fox fecal homogenate. We analyzed all samples in duplicate and considered a sample positive if >1 of the duplicates tested positive.
Statistical and Spatial Analyses
Human exposure or case follow-up of AE falls within the legislative mandate for public health in Ontario on the basis of the geographic boundaries of each public health unit (PHU). Therefore, we visualized the prevalence of infection in wild canids across southern Ontario by using choropleth maps organized by the administrative boundaries of the 29 southern Ontario PHUs (Figure 1). To account for potentially unreliable prevalence estimates in certain PHUs resulting from small sample sizes, we used a Bayesian estimation method with local priors to smooth prevalence estimates (28,29). We performed Bayesian smoothing by using R 3.4.2 with R packages maptools 0.8–39 and spdep 0.6–13 (R Foundation for Statistical Computing, http://cran.r-project.org). Graphic displays were produced by using QGIS 2.14.3 (http://www.qgis.org).
An underlying assumption for constructing CIs is independence of observations. Our data fail to meet this assumption because we cannot assume that the infection status of a wild canid is independent of others in the population. Thus, the dependence of the observations must be considered. Therefore, we constructed Agresti-Coull CIs for prevalence estimates by using Stata/SE 15.1 (StataCorp, http://www.stata.com) (30); this method has been recommended for data that violate the assumption of independence (31).
To assess for areas of high risk for infection (hotspots), we performed a 1-tailed spatial scan statistic by using a Bernoulli probability model with SaTScan 9.4.4 (https://www.satscan.org). We set the maximum size of the circular scanning window size to 50% of the total population. We estimated the statistical significance of the spatial clusters by using Monte Carlo hypothesis testing based on 999 iterations. We reported statistically significant primary and secondary nonoverlapping spatial clusters. We set the significance level for all analyses at 5% (α = 0.05). We performed the spatial scan test with the observations georeferenced to the centroids of the PHUs and then according to the latitude and longitude of PCR-positive and -negative tested wild canids to assess the consistency of results using different levels of spatial resolution.
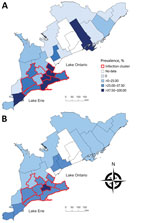
During November 2015–March 2017, we collected 460 wild canids (416 coyotes and 44 foxes) from 25 of the 29 southern Ontario PHUs and tested them for the presence of E. multilocularis DNA by using MC-PCR. We collected 205 wild canids (183 coyotes and 22 foxes) in the first collection period and 255 wild canids (233 coyotes and 22 foxes) in the second. During both collection periods, we collected >80% of the wild canids during the months of January, February, and March. Hunters and trappers consistently reported low fox population numbers throughout the duration of the project, resulting in a low number of sampled foxes. No canids were collected from PHUs 3535, 3553, 3555, and 3595 (Figure 1). Overall, 23% (95% CI 20%–27%) of wild canids, from 18 PHUs, tested positive for E. multilocularis (Table). Among coyotes, 24% (95% CI 20%–28%) tested positive; 21% (95% CI 11%–35%) of foxes tested positive. Raw prevalence ranged from 0% to 100% among PHUs (Figure 2). Smoothed prevalence by PHU estimates ranged from 4% to 46% (Table) and varied on a gradient of higher to lower prevalence from the southwestern to northeastern regions of southern Ontario (Figure 2).
The spatial scan test, georeferenced by PHU, detected a significant spatial cluster of high prevalence of infection (relative risk 2.26; p = 0.002) centered in PHU 3534, consisting of 10 contiguous PHUs (3527, 3531, 3534, 3536, 3537, 3544, 3546, 3552, 3554, and 3565) (Figure 2). The prevalence of infection among the 205 wild canids included in the cluster was 34% (95% CI 28%–40%). A second spatial scan test, using data georeferenced to each wild canid’s location of origin, detected a significant spatial cluster of high prevalence of infection (relative risk 2.53; p = 0.001), with a radius of 120 km also centered in PHU 3534. The prevalence of infection among the 216 wild canids included in the cluster was 34% (95% CI 28%–41%). No statistically significant nonoverlapping secondary high-risk spatial clusters were identified at either level of spatial resolution.
This report describes the prevalence of E. multilocularis infection in wild canids in Ontario. Because Ontario was previously considered free of the E. multilocularis tapeworm, we were surprised that 23% of the wild canids (107/460) tested positive for the parasite in our study. This finding is comparable to recent wild canid prevalence data from Edmonton, Alberta, Canada (9), where the E. multilocularis tapeworm has been recognized for decades (32). We anticipated that infection among wild canids would be confined to the region surrounding the western shores of Lake Ontario in southern Ontario, where the aforementioned cases of AE were observed. However, our findings indicate that E. multilocularis infection in wild canids is widely distributed across the western, central, and eastern regions of southern Ontario, with a high prevalence hotspot consisting of 10 PHUs in the western-central region (Figure 2). The combination of the high prevalence and wide geographic distribution of infection suggests that the E. multilocularis tapeworm was not a recent introduction into Ontario. In addition, to the authors’ knowledge, only 1 other study has investigated E. multilocularis tapeworms in the province; a survey of 302 red foxes from southern Ontario during 1979–1980 did not detect evidence of the parasite (33). Therefore, E. multilocularis tapeworms probably were introduced sometime after 1980.
How E. multilocularis tapeworms were introduced into Ontario is unclear. However, the spatial pattern of high infection prevalence among wild canids in the southern PHUs that border the northern shores of Lake Erie (Figure 2) might indicate a natural northeastern expansion from Michigan, a known endemic area (33). Also, the importation of dogs from endemic areas in North America or Europe, without any requirement for cestocide treatment, might have contributed to the introduction of E. multilocularis tapeworms into the province. Notably, molecular characterization of the metacestode stage from 1 of the southern Ontario dogs diagnosed with AE without travel history was consistent with E. multilocularis tapeworms of possible European origin (1), whereas another appeared to be North American in origin (K. Gesy, pers. comm., 2017 Dec 14). These findings strengthen the possibility that an importation event occurred, perhaps in addition to a natural range expansion. However, the meaning of this information remains unclear because data concerning the epidemiologic importance of individual strain variants are limited (34,35).
We measured an infection prevalence of 34% (95% CI 28%–40%) among wild canids within the southern Ontario hotspot. Consequently, a question of public health importance is to what extent the human population in southern Ontario is at risk for human AE. Across the endemic countries in Europe, where the prevalence of E. multilocularis infection in wild canids ranges from <1% to >50% (1), human AE is rare; the overall average annual incidence in these countries ranges from 0.03 to 0.3 cases/100,000 residents (36). However, substantial variation in risk exists across regions. For example, in areas with consistently high prevalence in wild canids (i.e., 35%–65% prevalence), the annual incidence of human AE can be as high as 8.1 cases/100,000 residents (37,38), which is similar to the prevalence estimates among wild canids in the southern Ontario hotspot that we describe. Furthermore, the location of the infection cluster encompasses multiple urban areas with human population densities of up to 1,700 residents/km2 (23). Therefore, transmission of E. multilocularis tapeworms should be considered a public health risk.
In areas endemic for E. multilocularis tapeworms, dog ownership has been associated with increased risk for human AE (37,39–41). Dog ownership might entail various human and dog behaviors that might lead to an increased risk for human infection with E. multilocularis tapeworms. These behaviors include leaving dogs outside unattended, walking dogs without a leash, allowing dogs to consume rodents, and inconsistent deworming of dogs (40). As such, monthly treatment with praziquantel is recommended for dogs that consume rodents in AE-endemic areas to prevent patent intestinal infections and therefore mitigate the risk for transmission to humans (36). The same is also recommended for dogs with hepatic AE because such dogs might also have concurrent intestinal infections (42). Thus, even in instances of canine hepatic AE, a follow-up investigation of possible exposure to E. multilocularis tapeworms for in-contact humans is warranted (43).
As of January 1, 2018, E. multilocularis infection was designated a reportable disease in animals in Ontario (44). Veterinarians and diagnostic laboratories are required to report animal cases directly to their local PHUs to minimize potential risks to human and public health. Furthermore, as of May 1, 2018, E. multilocularis infection in humans was designated a disease of public health importance (i.e., a disease that must be reported) in Ontario (45). Although human AE was not reportable before 2018, data from the Canadian Institute for Health Information indicate that >3 cases of human AE have been diagnosed in Ontario since 2014 (46); however, these data do not include information regarding patient travel or exposure histories. Therefore, whether these cases were locally acquired is unknown. Designating E. multilocularis infection as reportable in humans and animals is potentially important because, in AE-endemic areas (i.e., Europe), a large proportion of the economic burden associated with human AE is attributable to patients typically being diagnosed in the late stages of the disease, requiring lifelong chemotherapy and occasionally interventional procedures (e.g., percutaneous biliary and centroparasitic abscess drainage) (5,36). Therefore, the ability to anticipate E. multilocularis exposure and to diagnose early-stage human AE is essential to reduce the need for long-term treatment, thereby minimizing the economic burden associated with the disease. A limitation of having the infection reportable only in humans is that, given the long clinical incubation period of AE in humans, other persons potentially at risk would likely have been infected years earlier. Thus, in areas where E. multilocularis infection is endemic, a One Health surveillance approach that also requires mandatory reporting of E. multilocularis infection in animals to public health authorities could improve rates of prompt investigation of suspected exposure in persons and lead to earlier diagnosis.
Our study has several limitations. First, sample collection depended on carcass submission from hunters, trappers, and the Ontario Ministry of Natural Resources and Forestry. Although this convenience sampling allowed us to achieve a large sample size, it resulted in underrepresentation of parts of the study area. Because this approach resulted in PHUs with low sample sizes and thus potentially unreliable prevalence estimates, we used a Bayesian estimation method to smooth prevalence estimates. Second, the MC-PCR method used to detect E. multilocularis DNA is imperfect, having an overall sensitivity of 88% and specificity of 99% (25,27). However, we chose to employ this method because it is suitable for large-scale screening and its performance is comparable to what is considered the reference standard for diagnosis of infection in wild canids, the sedimentation and counting technique (26).
Our findings underscore the importance for continued surveillance among wild canids within and outside endemic areas in North America to monitor the spread of E. multilocularis tapeworms. In addition, an understanding of the prevalence of intestinal infections among dogs in AE-endemic areas would provide valuable information on potential exposure risk in human populations. Collectively, the data would guide public health and veterinary efforts in the development of targeted prevention strategies in this region.
Mr. Kotwa is a PhD candidate in the Department of Pathobiology at the University of Guelph. His research interests include interdisciplinary perspectives for evaluating emerging zoonotic diseases, with a specific concentration on investigating the role of wild canids as sentinel species for pathogens of public health and veterinary significance.
Acknowledgments
We thank the southern Ontario hunters and trappers, Aylmer District Stakeholders Committee, Nuisance Wildlife Control Inc., and the Ontario Ministry of Natural Resources and Forestry for coyote and fox submissions. The Canadian Wildlife Health Cooperative contributed invaluable resources and support for the processing of coyote and fox samples.
This study was supported by Bayer Animal Health, the National Center for Veterinary Parasitology, Natural Sciences and Engineering Research Council of Canada, Ontario Veterinary College, Ontario Animal Health Network, and Burroughs-Wellcome Fund.
References
- Deplazes P, Rinaldi L, Alvarez Rojas CA, Torgerson PR, Harandi MF, Romig T, et al. Global distribution of alveolar and cystic echinococcosis. Adv Parasitol. 2017;95:315–493.
- Kadry Z, Renner EC, Bachmann LM, Attigah N, Renner EL, Ammann RW, et al. Evaluation of treatment and long-term follow-up in patients with hepatic alveolar echinococcosis. Br J Surg. 2005;92:1110–6.
- Brunetti E, Kern P, Vuitton DA; Writing Panel for the WHO-IWGE. Expert consensus for the diagnosis and treatment of cystic and alveolar echinococcosis in humans. Acta Trop. 2010;114:1–16.
- Kern P. Clinical features and treatment of alveolar echinococcosis. Curr Opin Infect Dis. 2010;23:505–12.
- Brunetti E, White AC Jr. Cestode infestations: hydatid disease and cysticercosis. Infect Dis Clin North Am. 2012;26:421–35.
- Torgerson PR, Schweiger A, Deplazes P, Pohar M, Reichen J, Ammann RW, et al. Alveolar echinococcosis: from a deadly disease to a well-controlled infection. Relative survival and economic analysis in Switzerland over the last 35 years. J Hepatol. 2008;49:72–7.
- Ammann RW, Eckert J. Cestodes. Echinococcus. Gastroenterol Clin North Am. 1996;25:655–89.
- Pawlowski ZS, Kern EJ, Craig P, Dar KF, De Rosa F, Filice C, et al. Echinococcosis in humans: clinical aspects, diagnosis and treatment. In: Eckert J, Gemmel MA, Meslin F-X, Pawlowski ZS, editors. WHO/OIE manual on echinococcosis in humans and animals: a public health problem of global concern. Paris: World Health Organization/World Health Organization for Animal Health; 2001. p. 47–66.
- Catalano S, Lejeune M, Liccioli S, Verocai GG, Gesy KM, Jenkins EJ, et al. Echinococcus multilocularis in urban coyotes, Alberta, Canada. Emerg Infect Dis. 2012;18:1625–8.
- Massolo A, Liccioli S, Budke C, Klein C. Echinococcus multilocularis in North America: the great unknown. Parasite. 2014;21:73.
- Voigt DR, Berg WE. Coyote. In: Novak M, Baker JA, Obbard ME, Malloch B, editors. Wild furbearer management and conservation in North America. Toronto (Canada): Ontario Ministry of Natural Resources; 1987. p. 344–57.
- Eckert J, Conraths FJ, Tackmann K. Echinococcosis: an emerging or re-emerging zoonosis? Int J Parasitol. 2000;30:1283–94.
- Storandt ST, Virchow DR, Dryden MW, Hygnstrom SE, Kazacos KR. Distribution and prevalence of Echinococcus multilocularis in wild predators in Nebraska, Kansas, and Wyoming. J Parasitol. 2002;88:420–2.
- Peregrine AS, Jenkins EJ, Barnes B, Johnson S, Polley L, Barker IK, et al. Alveolar hydatid disease (Echinococcus multilocularis) in the liver of a Canadian dog in British Columbia, a newly endemic region. Can Vet J. 2012;53:870–4.
- Gesy K, Hill JE, Schwantje H, Liccioli S, Jenkins EJ. Establishment of a European-type strain of Echinococcus multilocularis in Canadian wildlife. Parasitology. 2013;140:1133–7.
- Skelding A, Brooks A, Stalker M, Mercer N, de Villa E, Gottstein B, et al. Hepatic alveolar hydatid disease (Echinococcus multilocularis) in a boxer dog from southern Ontario. Can Vet J. 2014;55:551–3.
- Oscos-Snowball A, Tan E, Peregrine AS, Foster R, Bronsoiler J, Gottstein B, et al. What is your diagnosis? Fluid aspirated from an abdominal mass in a dog. Vet Clin Pathol. 2015;44:167–8.
- French SK, Jajou S, Campbell GD, Cai HY, Kotwa JD, Peregrine AS, et al. Echinococcus multilocularis in a wild free-living eastern chipmunk (Tamias striatus) in southern Ontario: a case report and subsequent field study of wild small mammals. Vet Parasitol Reg Stud Reports. 2018;13:234–7.
- Peregrine AS, Cuq B. Echinococcus multilocularis: an emerging zoonotic disease. In: Proceedings of the American College of Veterinary Internal Medicine Forum; 2016 June 9–11; Denver, Colorado, USA.
- Pinard CJ, Cuq B, Gibson TWG, Brisson B, Plattner BL, Bienzle D, et al. Intolerance of albendazole therapy and failure to control hepatic alveolar echinococcosis in a dog in southern Ontario. In: Proceedings of the 61st Annual Meeting of the American Association of Veterinary Parasitologists; 2016 August 6–9; San Antonio, Texas, USA.
- Turner PV, Compo NR, Davidson S, McDowell R, Cai H, Gottstein B, et al. Diagnoses of alveolar echinococcosis in lemurs at an exotic animal sanctuary: implications for public health. In: Proceedings of the 66th Annual Meeting of the James Steele Conference on Diseases in Nature Transmissible to Man (DIN); 2016 May 25–27; San Antonio, Texas, USA.
- Corsini M, Geissbühler U, Howard J, Gottstein B, Spreng D, Frey CF. Clinical presentation, diagnosis, therapy and outcome of alveolar echinococcosis in dogs. Vet Rec. 2015;177:569.
- Statistics Canada. Population and dwelling count highlight tables, 2016 census [cited 2017 Sep 12]. http://www12.statcan.gc.ca/census-recensement/2016/dp-pd/hlt-fst/pd-pl/Table.cfm?Lang=Eng&T=201&S=3&O=D.
- Eckert J, Deplazes P, Craig PS, Gemmell MA, Gottstein B, Heath D, et al. Echinococcosis in animals: clinical aspects, diagnosis and treatment. In: Eckert J, Gemmell MA, Meslin FX, Pawlowski ZS, editors. WHO/OIE manual on echinococcosis in humans and animals: a public health problem of global concern. Paris: World Health Organization/World Organization for Animal Health; 2001. p. 80.
- Isaksson M, Hagström Å, Armua-Fernandez MT, Wahlström H, Ågren EO, Miller A, et al. A semi-automated magnetic capture probe based DNA extraction and real-time PCR method applied in the Swedish surveillance of Echinococcus multilocularis in red fox (Vulpes vulpes) faecal samples. Parasit Vectors. 2014;7:583.
- Deplazes P, Eckert J. Veterinary aspects of alveolar echinococcosis—a zoonosis of public health significance. Vet Parasitol. 2001;98:65–87.
- Wahlström H, Comin A, Isaksson M, Deplazes P. Detection of Echinococcus multilocularis by MC-PCR: evaluation of diagnostic sensitivity and specificity without gold standard. Infect Ecol Epidemiol. 2016;6:30173.
- Mollié A. Bayesian mapping of disease. In: Gilks WR, Richardson S, Spiegelhalter DJ, editors. Markov chain Monte Carlo in practice. London: Chapman & Hall/CRC Press; 1996. p. 359.
- Berke O. Choropleth mapping of regional count data of Echinococcus multilocularis among red foxes in Lower Saxony, Germany. Prev Vet Med. 2001;52:119–31.
- Agresti A, Coul BA. Approximate is better than “exact” for interval estimation of binomial proportions. Am Stat. 1998;52:119–26.
- Miao W, Gastwirth JL. The effect of dependence on confidence intervals for a population proportion. Am Stat. 2004;58:124–30.
- Holmes JC, Mahrt JL, Samuel WM. The occurrence of Echinococcus multilocularis Leuckart, 1863 in Alberta. Can J Zool. 1971;49:575–6.
- Storandt ST, Kazacos KR. Echinococcus multilocularis identified in Michigan with additional records from Ohio. J Parasitol. 2012;98:891–3.
- Thompson RC. The taxonomy, phylogeny and transmission of Echinococcus. Exp Parasitol. 2008;119:439–46.
- Lymbery AJ. Phylogenetic pattern, evolutionary processes and species delimitation in the genus Echinococcus. Adv Parasitol. 2017;95:111–45.
- Gottstein B, Stojkovic M, Vuitton DA, Millon L, Marcinkute A, Deplazes P. Threat of alveolar echinococcosis to public health—a challenge for Europe. Trends Parasitol. 2015;31:407–12.
- Piarroux M, Piarroux R, Knapp J, Bardonnet K, Dumortier J, Watelet J, et al. FrancEchino Surveillance Network. Populations at risk for alveolar echinococcosis, France. Emerg Infect Dis. 2013;19:721–8.
- Said-Ali Z, Grenouillet F, Knapp J, Bresson-Hadni S, Vuitton DA, Raoul F, et al. FrancEchino Surveillance Network. Detecting nested clusters of human alveolar echinococcosis. Parasitology. 2013;140:1693–700.
- Kreidl P, Allerberger F, Judmaier G, Auer H, Aspöck H, Hall AJ. Domestic pets as risk factors for alveolar hydatid disease in Austria. Am J Epidemiol. 1998;147:978–81.
- Kern P, Bardonnet K, Renner E, Auer H, Pawlowski Z, Ammann RW, et al. European Echinococcosis Registry. European echinococcosis registry: human alveolar echinococcosis, Europe, 1982-2000. Emerg Infect Dis. 2003;9:343–9.
- Conraths FJ, Probst C, Possenti A, Boufana B, Saulle R, La Torre G, et al. Potential risk factors associated with human alveolar echinococcosis: Systematic review and meta-analysis. PLoS Negl Trop Dis. 2017;11:e0005801.
- Deplazes P, Arnold P, Kaser-Holtz B, Gardelle O, Guscetti F, Halle M, et al. Concurrent infections of the liver and intestine with Echinococcus multilocularis in dogs. Arch Int Hidatid. 1997;31:202–3.
- Trotz-Williams LA, Mercer NJ, Walters JM, Wallace D, Gottstein B, Osterman-Lind E, et al. Public health follow‐up of suspected exposure to Echinococcus multilocularis in southwestern Ontario. Zoonoses Public Health. 2017;64:460–7.
- Government of Ontario. R.R.O. 1990, Reg. 557: communicable diseases—general, s 6.1(3) [cited 2018 Jan 31]. http://www.e-laws.gov.on.ca/html/regs/english/elaws_regs_900557_e.htm
- Government of Ontario. O. Reg. 135/18: designation of diseases, s 4 [cited 2018 Oct 3]. https://www.ontario.ca/laws/regulation/180135
- Government of Ontario. Infectious diseases protocol appendix A: disease specific chapters. 2018 [cited 2018 Oct 3]. http://www.health.gov.on.ca/en/pro/programs/publichealth/oph_standards/docs/E_multilocularis_chapter.pdf.
Figures
Table
Cite This ArticleOriginal Publication Date: January 07, 2019
Table of Contents – Volume 25, Number 2—February 2019
| EID Search Options |
|---|
|
|
|
|
|
|
Please use the form below to submit correspondence to the authors or contact them at the following address:
Jonathon D. Kotwa, University of Guelph, Department of Pathobiology, 419 Gordon St, Guelph, ON N1G 2W1, Canada
Top