Volume 26, Number 7—July 2020
Dispatch
Inactivation of Severe Acute Respiratory Syndrome Coronavirus 2 by WHO-Recommended Hand Rub Formulations and Alcohols
Cite This Article
Citation for Media
Abstract
Infection control instructions call for use of alcohol-based hand rub solutions to inactivate severe acute respiratory syndrome coronavirus 2. We determined the virucidal activity of World Health Organization–recommended hand rub formulations, at full strength and multiple dilutions, and of the active ingredients. All disinfectants demonstrated efficient virus inactivation.
Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) is the third highly pathogenic human coronavirus to cross the species barrier into the human population during the past 20 years (1–3). SARS-CoV-2 infection is associated with coronavirus disease (COVID-19), which is characterized by severe respiratory distress, fever, and cough and high rates of mortality, especially in older persons and those with underlying health conditions (3). The World Health Organization (WHO) declared SARS-CoV-2 a pandemic on March 11, 2020 (4), and by April 8, a total of 1,447,466 confirmed cases and 83,471 deaths from SARS-CoV-2 had been reported worldwide (5).
Human-to-human transmission of SARS-CoV-2 is efficient, and infected persons can transmit the virus even when they have no, or only mild, symptoms (3). Because no antiviral drugs or vaccines are available, virus containment and prevention of infection are the current highest priorities. To limit virus spread, effective hand hygiene is crucial. Therefore, easily available but efficient disinfectants are needed. WHO’s guidelines for hand hygiene in healthcare suggest 2 alcohol-based formulations for hand sanitization to reduce the infectivity and spread of pathogens (6). WHO’s recommendations are based on fast-acting, broad-spectrum microbicidal activity, along with accessibility and safety. The original WHO formulations failed to meet the efficacy requirements of European Norm 1500 in previous tests (7). However, Suchomel et al. (8) suggested modified versions with increased concentrations of ethanol: 80% (wt/wt) (85.5% [vol/vol]; formulation I), or isopropanol, 75% (wt/wt) (81.3% [vol/vol]; formulations II). Later, they complemented these by reducing the glycerol concentrations (9).
We previously showed that these modified WHO formulations were able to inactivate severe acute respiratory syndrome coronavirus (SARS-CoV) and Middle East respiratory syndrome coronavirus (MERS-CoV; 10), which are related to SARS-CoV-2. Current recommendations to inactivate SARS-CoV-2 were translated from findings of other coronaviruses (11). To evaluate whether these alcohol-based disinfectants also effectively inactivate SARS-CoV-2, we tested different concentrations of the original and modified WHO formulations I and II (6,9), ethanol, and 2-propanol for virucidal activity.
We propagated SARS-CoV-2 (SARS-CoV-2/München-1.1/2020/929) on VeroE6 cells (kindly provided by M. Müller and C. Drosten; Charité, Berlin, Germany). We cultured VeroE6 cells in Dulbecco’s modified minimal essential medium supplemented with 10% heat inactivated fetal bovine serum, 1% nonessential amino acids, 100 µg/mL of streptomycin and 100 IU/mL of penicillin, and 15 mMol of HEPES (Gibco; ThermoFisher, https://www.thermofisher.com).
Original WHO formulation I consists of 80% (vol/vol) ethanol, 1.45% (vol/vol) glycerol, and 0.125% (vol/vol) hydrogen peroxide. Original WHO formulation II consists of 75% (vol/vol) 2-propanol, 1.45% (vol/vol) glycerol, and 0.125% (vol/vol) hydrogen peroxide. The modified WHO formulation I used in our study consists of 80% (wt/wt) ethanol, 0.725% (vol/vol) glycerol, and 0.125% (vol/vol) hydrogen peroxide. The modified isopropyl-based WHO formulation II contains 75% (wt/wt) 2-propanol, 0.725% (vol/vol) glycerol, and 0.125% (vol/vol) hydrogen peroxide (9). We also prepared ethanol (CAS 64–17–5) and 2-propanol (CAS 67–63–0) in vol/vol dilutions for investigation.
We performed virucidal activity studies by using a quantitative suspension test with 30 s exposure time (6). In brief, we mixed 1 part virus suspension with 1 part organic load (0.3% bovine serum albumin as an interfering substance) and 8 parts disinfectant solution of different concentrations. After a 30 s exposure, we serially diluted samples and determined the 50% tissue culture infectious dose (TCID50) per milliliter by using crystal violet staining and subsequently scoring the number of wells displaying cytopathic effects. We calculated TCID50 by the Spearman-Kärber algorithm, as described (12). We monitored the cytotoxic effects of disinfectants by using crystal violet staining and optical analysis for altered density and morphology of the cellular monolayer in the absence of virus. We quantified cytotoxic effects analogous to the TCID50/mL of the virus infectivity.
We determined dose-response curves as percent normalized virus inactivation versus percent log disinfectant concentration by nonlinear regression using the robust fitting method on the normalized TCID50 data implemented in Prism version 8.0.3 (GraphPad, https://www.graphpad.com). We plotted reference curves for SARS-CoV, MERS-CoV, and bovine CoV (BCoV) by using previously published data (9). BCoV is often used as surrogate for highly pathogenic human CoVs. We assessed the mean TCID50 and standard deviations of means from 3 individual experiments. We identified outliers by using Grubb’s test in Prism. We calculated reduction factors (RFs) for each treatment condition as follows:
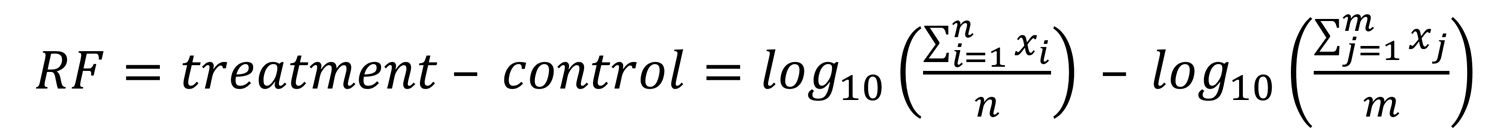
Our results showed that SARS-CoV-2 was highly susceptible to the original and the modified WHO formulations (Figure 1). The original and modified versions of formulation I efficiently inactivated the virus. The original formulation I of 80% (vol/vol) ethanol had an RF of >3.8 (Figure 1, panel A) and the modified formulation I of 80% (wt/wt) ethanol had an RF of >5.9 (Figure 1, panel C). Dilutions >40% were still effective (Figure 1, panels A and C). Subsequent regression analysis of modified formulation I revealed similar inactivation profiles compared with SARS-CoV, MERS-CoV, and BCoV. (Figure 1, panel C). The original and modified versions of formulation II also were effective. The original formulation II of 75% (vol/vol) 2-propanol had a log10-reduction of >3.8 (Figure 1, panel B) and the modified formulation II of 75% (wt/wt) 2-propanol had a log10-reduction of >5.9. Dilution >30% (vol/vol) also resulted in complete viral inactivation (Figure 1, panel D). Regression analysis of modified WHO formulation II showed the inactivation profile of SARS-CoV-2 was comparable to those of SARS-CoV, BCoV, and MERS-CoV (Figure 1, panel D).
We also investigated the susceptibility of SARS-CoV-2 against the active components of the WHO-recommended formulations, which are also the active ingredients of commercially available hand disinfectants. Ethanol (Figure 2, panel A) and 2-propanol (Figure 2, panel B) were able to reduce viral titers to background levels in 30 s with RFs of between 4.8 and ≥5.9. Furthermore, we noted that a concentration of >30% (vol/vol) ethanol or 2-propanol is sufficient for complete viral inactivation (Figure 2).
We found that SARS-CoV-2 was efficiently inactivated by WHO-recommended formulations, supporting their use in healthcare systems and viral outbreaks. Of note, both the original and modified formulations were able to reduce viral titers to background level within 30 s. In addition, ethanol and 2-propanol were efficient in inactivating the virus in 30 s at a concentration of >30% (vol/vol). Alcohol constitutes the basis for many hand rubs routinely used in healthcare settings. One caveat of this study is the defined inactivation time of exactly 30 s, which is the time recommended but not routinely performed in practice. Our findings are crucial to minimize viral transmission and maximize virus inactivation in the current SARS-CoV-2 outbreak.
Ms. Kratzel is a PhD candidate affiliated with the Institute of Virology and Immunology, Bern and Mittelhäusern, Switzerland, the Department of Infectious Diseases and Pathobiology, Vetsuisse Faculty, University of Bern, Bern, Switzerland and the Graduate School for Cellular and Biomedical Sciences, University of Bern. Her research interests include coronaviruses.
Acknowledgment
This article was preprinted at https://www.biorxiv.org/content/10.1101/2020.03.10.986711v1.
References
- Drosten C, Günther S, Preiser W, van der Werf S, Brodt HR, Becker S, et al. Identification of a novel coronavirus in patients with severe acute respiratory syndrome. N Engl J Med. 2003;348:1967–76. DOIPubMedGoogle Scholar
- Zaki AM, van Boheemen S, Bestebroer TM, Osterhaus ADME, Fouchier RAM. Isolation of a novel coronavirus from a man with pneumonia in Saudi Arabia. N Engl J Med. 2012;367:1814–20. DOIPubMedGoogle Scholar
- Lai C-C, Shih T-P, Ko W-C, Tang H-J, Hsueh P-R. Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) and coronavirus disease-2019 (COVID-19): the epidemic and the challenges. Int J Antimicrob Agents. 2020;55:
105924 . DOIPubMedGoogle Scholar - World Health Organization. WHO Director-General’s opening remarks at the media briefing on COVID-19: 11 March 2020 [cited 2020 Apr 8] https://www.who.int/dg/speeches/detail/who-director-general-s-opening-remarks-at-the-media-briefing-on-covid-19---11-march-2020
- Gardner L. Update January 31: modeling the spreading risk of 2019-nCoV. Johns Hopkins University Center for Systems Science and Engineering; 2020 Jan 31 [cited 2020 Mar 31] https://systems.jhu.edu/research/public-health/ncov-model-2
- World Health Organization. WHO guidelines on hand hygiene in health care: first global patient safety challenge clean care is safer care. Geneva: the Organization; 2009 [cited 2020 Apr 08] https://apps.who.int/iris/bitstream/handle/10665/44102/9789241597906_eng.pdf
- European Committee for Standardization. Chemical disinfectants and antiseptics. Hygienic hand disinfection. Test method and requirement. Brussels: the Committee; 2013 [cited 2020 Apr 8] https://standards.globalspec.com/std/1597777/EN%201500
- Suchomel M, Kundi M, Allegranzi B, Pittet D, Rotter ML. Testing of the World Health Organization-recommended formulations for surgical hand preparation and proposals for increased efficacy. J Hosp Infect. 2011;79:115–8. DOIPubMedGoogle Scholar
- Suchomel M, Kundi M, Pittet D, Rotter ML. Modified World Health Organization hand rub formulations comply with European efficacy requirements for preoperative surgical hand preparations. Infect Control Hosp Epidemiol. 2013;34:245–50. DOIPubMedGoogle Scholar
- Siddharta A, Pfaender S, Vielle NJ, Dijkman R, Friesland M, Becker B, et al. Virucidal activity of World Health Organization-recommended formulations against enveloped viruses, including Zika, Ebola, and emerging coronaviruses. J Infect Dis. 2017;215:902–6. DOIPubMedGoogle Scholar
- Kampf G, Todt D, Pfaender S, Steinmann E. Persistence of coronaviruses on inanimate surfaces and their inactivation with biocidal agents. J Hosp Infect. 2020;104:246–51. DOIPubMedGoogle Scholar
- George VG, Hierholzer JC, Ades EW. Cell culture. In: Virology methods manual. Mahy BWJ, Kangro HO, editors. Academic Press: London; 1996. p. 3–24.
Figures
Cite This ArticleOriginal Publication Date: April 13, 2020
Table of Contents – Volume 26, Number 7—July 2020
| EID Search Options |
|---|
|
|
|
|
|
|

Please use the form below to submit correspondence to the authors or contact them at the following address:
Stephanie Pfaender, Department for Molecular and Medical Virology, Ruhr-Universität Bochum, Universitätsstrasse 150, 44801 Bochum, Germany
Top