Volume 28, Number 11—November 2022
Research
Effects of the COVID-19 Pandemic on Incidence and Epidemiology of Catheter-Related Bacteremia, Spain
Cite This Article
Citation for Media
Abstract
We compared hospital-acquired catheter-related bacteremia (CRB) episodes diagnosed at acute care hospitals in Catalonia, Spain, during the COVID-19 pandemic in 2020 with those detected during 2007–2019. We compared the annual observed and predicted CRB rates by using the negative binomial regression model and calculated stratified annual root mean squared errors. A total of 10,030 episodes were diagnosed during 2007–2020. During 2020, the observed CRB incidence rate was 0.29/103 patient-days, whereas the predicted CRB rate was 0.14/103 patient-days. The root mean squared error was 0.153. Thus, a substantial increase in hospital-acquired CRB cases was observed during the COVID-19 pandemic in 2020 compared with the rate predicted from 2007–2019. The incidence rate was expected to increase by 1.07 (95% CI 1–1.15) for every 1,000 COVID-19–related hospital admissions. We recommend maintaining all CRB prevention efforts regardless of the coexistence of other challenges, such as the COVID-19 pandemic.
In December 2019, the first cases of COVID-19 were reported in Wuhan, China (1). On March 11, 2020, the World Health Organization declared COVID-19 a global pandemic because of the spread of SARS-CoV-2 infections worldwide (2). Subsequent waves related to the spread of different SARS-CoV-2 serotypes forced healthcare systems and, specifically, acute care hospitals to modify their structural and human resource organization (3); scheduled elective surgeries were cancelled, and healthcare workers had to change their specific clinical roles to address the abrupt increase in admissions of SARS-CoV-2–infected patients. To reduce SARS-CoV-2 nosocomial transmission, airborne and contact precaution measures were reinforced, personal protective equipment was worn by healthcare providers, and strict hand hygiene measures were observed at most centers (4).
Hand hygiene is a cornerstone of healthcare-associated infection (HAI) prevention, and reductions in Clostridioides difficile colitis incidence (5,6) and surgical-site infections (7,8) have been observed in different settings during the COVID-19 pandemic. However, reductions in other HAIs, such as catheter-associated urinary tract infections, ventilator-associated pneumonia, or catheter-related bacteremia (CRB) (9,10), were not observed. In addition, multidrug-resistant microorganisms were increasingly involved in these other HAIs (10–12).
CRB is one of the most frequent HAIs (13,14) and represents a major health challenge because of its high association with illness and death (15,16). CRB is currently considered a leading safety concern in healthcare settings and is a clinical practice quality indicator (17). For these reasons, CRB surveillance is mandatory in most countries (18–20).
In Catalonia, Spain, CRB surveillance is guided by the VINCat program of the Catalan Health Service (21), which provides a surveillance system for healthcare-associated nosocomial infections. The VINCat program was launched in 2006; the main objective of this program is to reduce the incidence of HAIs through continuous active monitoring and implementation of preventive programs (21). During recent decades, the incidence of healthcare-acquired CRB has decreased in most hospitals, especially in intensive care units (ICUs), because of the application of preventive measures (22,23). Some of the most critical evidence-based preventive interventions have been using appropriate barrier precautions and hand hygiene before handling catheters, disinfecting skin with chlorhexidine solutions, using appropriate catheter materials, carefully selecting insertion sites that avoid the femoral site, and withdrawing catheters whenever possible (24). During the COVID-19 pandemic, adherence to some of these preventive measures has notably affected HAI incidence rates (11); however, the effect of COVID-19 on CRB incidence is not definitively known. The aim of this study was to assess the effects of the COVID-19 pandemic on the incidence of hospital-acquired CRB.
Clinical Setting
Bacteremia associated with the use of venous catheters was continuously monitored under the VINCat program. All nosocomial episodes of CRB diagnosed in adult patients at each participating hospital were prospectively followed and reported to the VINCat program by infection control teams. CRB cases were identified by daily evaluation of all patients with bacteria-positive blood cultures.
Hospitals participating in the VINCat program are classified into 3 categories according to the number of beds available for hospitalization: >500 beds (group I), 200–499 beds (group II), and <200 beds (group III). Data from each hospital are continuously monitored and presented in general clinical sessions. A public annual report is published on the VINCat website (21).
Definitions
We defined catheter-related bacteremia as the detection of bacterial growth in patient blood using a venous catheter; >1 set of blood cultures were obtained from a peripheral vein and 2 sets were obtained to identify habitual skin-colonizing microorganisms, such as coagulase-negative staphylococci, Micrococcus spp., Propionibacterium acnes, Bacillus spp., and Corynebacterium spp. Positive bacterial cultures had to be associated with clinical manifestations of infection, such as fever, chills, or hypotension, and absence of any apparent alternative source of bloodstream infection (BSI). The conditions had to be accompanied by >1 of the following criteria: >15 CFU per catheter segment in semiquantitative cultures or >103 CFU per catheter segment in quantitative cultures that detected the same microorganism found in peripheral blood cultures; quantitative blood cultures that detected the same microorganism and showed a difference of >5:1 between the blood obtained from the lumen of a venous catheter and that obtained from a peripheral vein by puncture; difference of >2 hours between positive bacterial cultures obtained from a peripheral vein and the lumen of a venous catheter; presence of inflammatory signs or purulent secretions in the insertion point or the subcutaneous tunnel of a venous catheter (a culture of the secretion showing growth of the same microorganism detected in the blood cultures was also useful); and resolution of clinical signs and symptoms after catheter withdrawal with or without appropriate antibiotic treatment. For the clinical diagnosis of peripheral venous CRB, we required signs of phlebitis (induration, pain, or signs of inflammation at the insertion point or the catheter route).
Exclusion Criteria
We excluded patients if they were under 18 years of age, were outpatients, and had a hospital stay <48 hours at the time of BSI detection. We also excluded those who had CRB detected at an outpatient service or had CRB associated with arterial catheters.
Microbiology
Two sets of 2 blood samples from a peripheral vein were obtained from all patients with a suspected BSI. An additional blood sample was also obtained through the catheter. When possible, the catheter tip was cultured after removal. Blood samples were processed at the microbiology laboratories of each center in accordance with standard operating procedures. All microorganisms were identified by using standard microbiological techniques at each center.
Statistical Analysis
We reported categorical variables as the number of cases and percentages and continuous variables as means +SD or medians with interquartile ranges, depending on whether the distribution was normal or nonnormal. We assessed normality of variables graphically by using quantile-quantile and density plots. We calculated the CRB incidence rate by dividing the total number of episodes of CRB by the total number of hospital stays (patient-days) in 1 year.
We used a negative binomial regression model to assess the rate trend of CRBs diagnosed at VINCat hospitals each year during 2007–2019. We used the number of admissions per year as the offset variable, number of events as the dependent variable, and year as the main independent variable. We performed stratified analyses according to hospital ward, catheter type, catheter insertion site, catheter use, and type of identified microorganism. We reported the annual rate of CRBs diagnosed per 1,000 patient-days and the incidence rate ratio (IRR) and 95% CI for each model. We focused the interpretation of the IRR on the annual rate of increase or decrease.
We plotted and compared the annual CRB rates observed during 2007–2020 and annual CRB rates predicted by our model. We calculated the average root mean squared error (RMSE) of the model predictions for CRB rates during 2007–2019 and compared the RMSEs between the expected rate according to the model and observed rate in 2020. We replicated these analyses after stratifying by hospital ward, catheter type, catheter insertion site, catheter use, and type of microorganism.
We evaluated the conditions of application in all models and calculated the 95% CI for each estimator. We arbitrarily set the level of statistical significance at 5%. We performed the analyses using the statistical package R version 4.0.3 (The R Project for Statistical Computing, https://www.r-project.org) for Windows.
Ethical Considerations
Participation in the VINCat program was voluntary, and data confidentiality was guaranteed. This study was evaluated and approved by the Parc Taulí Hospital Research Ethics Committee, Sabadell, Spain.
Study Periods
During 2007–2020, a total of 10,030 nosocomial episodes of CRB were diagnosed. Data from the 2007–2019 period have been analyzed and described previously (25). In summary, during 2007–2019, a total of 9,290 episodes of CRB were diagnosed. The mean annual incidence was 0.2 episodes/103 patient-days, 73.7% of episodes occurred in non-ICU wards, 62.7% of episodes were related to central vascular catheters, 24.1% of episodes were related to peripheral venous catheters, and 13.3% of episodes were related to peripherally inserted central venous catheters (25). The incidence rate of CRB decreased substantially over the 2007–2019 study period (IRR 0.94, 95% CI 0.93–0.96), especially in ICU wards. CRB episodes caused by central vascular catheters fell markedly (IRR 0.90, 95% CI 0.89–0.92), whereas those associated with peripherally inserted catheters increased.
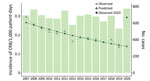
In 2020, a total of 774 CRB episodes were diagnosed at the participating hospitals. We determined that the incidence rate was 0.29 episodes/103 patient-days (Figure 1). Of 774 episodes, 297 (40.1%) were acquired in conventional medical wards, 127 (17.2%) in surgical wards, and 316 (42.7%) in ICUs. We found that the catheters most frequently implicated in CRB were central venous catheters (412 cases, 55.7%), peripheral catheters (169 cases, 22.8%), and peripherally inserted central venous catheters (159 cases, 21.5%). Catheters causing CRB were located in the arm/forearm (323 cases, 43.6%), jugular (237 cases, 32.0%), subclavian (116 cases, 15.7%), or femoral (52 cases, 7.03%) sites. The catheters were used for medication and serum infusion (583 cases, 78.8%), parenteral nutrition (146 cases, 19.7%), or hemodialysis (11 cases, 1.5%). The most frequent causes of CRB were coagulase-negative staphylococci (299 cases, 41.3%), Staphylococcus aureus (155 cases, 21.4%), gram-negative enterobacteria (112 cases, 15.5%), enterococci (72 cases, 9.9%), Candida sp. (45 cases, 6.2%), and Pseudomonas aeruginosa (34 cases, 4.7%).
Comparison of Observed and Expected Incidence Rates
According to the case mix index observed during 2007–2019, we predicted that the incidence rate for CRB in 2020 was 0.14 episodes/103 patient-days. However, we observed 0.29 episodes/103 patient-days (observed/predicted [O/P] ratio 2.10, 95% CI 1.95–2.25) in 2020. The RMSE was 0.015 during 2007–2019 and 0.153 in 2020 (Figure 1). Disparities between predicted and observed rates were consistent among the different participating hospitals (Appendix Figure 1).
In conventional surgical and medical wards, we found that the predicted incidence rate for CRB was 0.12 episodes/103 patient-days, and the observed rate was 0.19/103 patient-days in 2020 (O/P 1.51, 95% CI 1.37–1.65). However, in ICUs, we predicted the incidence rate was 0.48 episodes/103 patient-days, but the observed rate was 1.62/103 patient-days in 2020 (O/P 3.42, 95% CI 3.04–3.79). The average RMSE was 0.013 for conventional wards and 0.069 for ICUs during 2007–2019, whereas, in 2020, the RMSE was 0.062 for conventional wards and 1.147 for ICUs (Table 1; Figure 2).
We observed an incidence rate of 0.064 for CRB caused by peripheral catheters in 2020; the predicted rate according to the negative binomial regression model was 0.05 (O/P 1.24, 95% CI 1.06–1.43). When central catheters were used, the observed rate for CRB was 0.16, and the predicted rate was 0.06 (O/P 2.54, 95% CI 2.29–2.78). When peripherally inserted central catheters were used, the observed rate for CRB was 0.06, and the predicted rate was 0.04 (O/P 1.73, 95% CI 1.46–2.00). We observed increases in RMSEs in 2020 compared with the 2007–2019 period for peripheral catheters (0.012 vs. 0.007), central catheters (0.094 vs. 0.008), and peripherally inserted central catheters (0.025 vs. 0.004) (Table 1; Figure 2). In addition, we determined that the number of observed CRB episodes in 2020 were higher than predicted episodes depending on the location of the catheter; increased incidence was more pronounced in catheters located in femoral (O/P 3.11, 95% CI 2.27–3.96), jugular (O/P 2.64, 95% CI 2.30–2.97), and subclavian (O/P 1.88, 95% CI 1.53–2.22) sites (Table 1; Appendix Figure 2).
In 2020, we found increases in observed CRB incidence rates compared with rates predicted by the binomial regression model according to catheter use and causative microorganisms. For hemodialysis, the observed CRB rate was 0.004, and the predicted rate was 0.003 (O/P 1.25, 95% CI 0.51–1.99). For parenteral nutrition, the observed CRB rate was 0.06, and the predicted rate was 0.03 (O/P 1.62, 95% CI 1.36–1.89). For other uses, the observed CRB rate was 0.22, and the predicted rate was 0.10 (O/P 2.14, 95% CI 1.97–2.31); the last category increased most notably (Table 1; Figure 2). Observed CRB rates were increased compared with predicted rates for all causative microorganisms, especially enterococci (O/P 5.41, 95% CI 4.16–6.65).
Relationship between Monthly CRB Incidence Rates and SARS-CoV-2 Admissions
The total number of hospital admissions and the proportion of patients affected by COVID-19 changed substantially during 2020 (Figure 3). We recorded more COVID-19–related admissions during February–June in both conventional wards and ICUs (Table 2; Figure 3). The peak rate of COVID-19 hospital admissions was 54.87 in March, and the lowest rate was 14.15 in January.
Concomitantly, CRB incidence rates also varied during 2020, reaching a peak in April (0.57 episodes of CRB/103 admissions), followed by August and December (0.44 episodes of CRB/103 admissions for each month) (Table 2). We observed the lowest CRB rate at the beginning of the year (0.13 episodes of CRB/103 admissions).
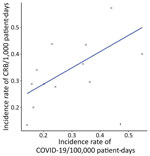
We observed an association between CRB and COVID-19 incidence rate. The CRB incidence rate was expected to increase by 1.07 (IRR 1.07, 95% CI 1–1.15) for every 1,000 COVID-19 admissions if all factors remained constant (Figure 4).
We demonstrated that the COVID-19 pandemic increased CRB incidence in 2020 in our hospitals in Catalonia, Spain. We found that months with the highest proportion of COVID-19 admissions were strongly associated with increased CRB incidence. We also described the most critical CRB characteristics that changed during the pandemic in 2020. Compared with previous years, we observed increased CRB incidence in both ICUs and conventional wards in 2020.
Other studies conducted around the same time observed increased HAI incidence rates during 2020, especially in ICUs. Catheter-associated urinary tract infections, ventilator-associated pneumonia, and CRB were the HAIs with the greatest increases (9–11). In contrast, other HAIs, such as nosocomial-acquired C. difficile colitis (5,6) or surgical-site infections (7,8, decreased during the same period. Of note, HAIs may be more frequently associated with patients receiving steroids or tocilizumab (26), although a specific association with BSI was not observed (27).
In most cases, the increased rates of CRB were likely associated with a lower adherence to specific preventive measures during the months when the pandemic caused the most hospital admissions, despite the generalized reinforcement of contact precautions and hand hygiene to reduce SARS-CoV-2 nosocomial transmission. Of note, in our hospital settings, alcohol-based product consumption for hand hygiene during 2020 increased 2.4-fold overall and 1.9-fold in ICUs compared with the previous year, and a similar trend was observed in a hospital in Taiwan (28). Therefore, although proper hand hygiene is necessary to prevent CRB and other HAIs, it is not sufficient to avoid HAIs if other measures are not performed during the insertion and care of vascular catheters. Specifically, since 2006, various evidence-based bundles for CRB interventions have been shown to reduce CRB, especially in the ICU setting. These bundles include handwashing, using full-barrier precautions, cleaning the skin with chlorhexidine, avoiding the femoral site if possible, and removing unnecessary catheters (22,23). Among the different preventive measures, both hand hygiene and catheter insertion measures were associated with reduced incidence of CRB, and they were most effective when both measures were applied simultaneously (24).
The first limitation of our study is that heterogeneity of COVID-19 pandemic responses existed between hospitals, resulting in lack of data on adherence to CRB preventive measures at each center. Second, there was a lack of clinical information regarding the presence of chronic diseases or clinical conditions that might influence CRB incidence rates. However, the availability of a large number of CRB episodes diagnosed by standardized definitions is a strength that enables generalization of our observations. In addition, CRB incidence rates were adjusted by patient-days rather than catheter-days, which enabled surveillance of all types of catheters inserted in all hospital wards.
In 2020, substantial resources were allocated for infection prevention to manage the SARS-CoV-2 outbreak, which also affected HAI prevention programs. Because CRB is a key healthcare quality indicator (29), our observations stress the importance of maintaining all prevention efforts, regardless of the coexistence of other challenges, such as the worldwide COVID-19 pandemic.
Dr. Gasch is a clinician, associate of the Infectious Diseases Service at the Corporació Sanitària Parc Taulí (Sabadell), and a member of the infection control team of the institution. He is an associate professor in the Faculty of Medicine at Autonomous University of Barcelona, Spain, and his research interests are focused on the study of bloodstream infections and infective endocarditis.
Acknowledgments
We thank Johanna-Milena Caro and Garazi Carrillo-Aguirre for their help obtaining data for this project.
The study is part of the doctoral thesis of L.B.C. in the medicine department at Autonomous University of Barcelona. O.G. received a personal research grant from Pla estratègic de recerca i innovació en salut (PERIS) 2019–2021 (Departament de Salut, Generalitat de Catalunya). This work was supported by the Ministry of Economy and Competitiveness, Spain, Carlos II Institute Expedient: PI20/01563, Red Española de Investigación en Patología Infecciosa (REIPI), Centro de Investigación Biomédica en Red (CIBER) de Enfermedades Respiratorias (CIBERES CB06/06/0037), and CIBER of Infectious Diseases (INFEC, CB21/13/00009, IDIBELL), an initiative of the Instituto de Salud Carlos III, Madrid, Spain.
Additional members of the Catalan Infection Control and Antimicrobial Stewardship Program: Alfredo Jover, Dolors Castellana (Hospital Universitari Arnau de Vilanova, Lleida, Spain); Montserrat Olona, Antonia García Pino (Hospital Universitari Joan XXIII, Tarragona, Spain); Josep Rebull Fatsini, M France Domènech (Hospital Verge de la Cinta, Tortosa, Spain); Miquel Pujol, Ana Hornero (Hospital Universitari de Bellvitge, L’Hospitalet de Llobregat, Spain); Joaquín López-Contreras, Engracia Fernández (Hospital de la Santa Creu i Sant Pau. Barcelona, Spain); Xavier Salgado, Dolors Domènech (Hospital Universitari Doctor Josep Trueta. Girona. Spain); Ana Lérida, Lydia Martin (Hospital deViladecans, Viladecans, Spain); Nieves Sopena, Irma Casas Garcia (Hospital Universitari Germans Tries i Pujol. Badalona. Spain); Rafel Pérez, Encarna Maraver (Althaia Xarxa Asistencia, Manresa, Spain); Eva Palau, Pepi Serrats (Clinica de Girona, Girona, Spain); José Antonio Martínez, Gemina Santana (Hospital Clínic i Provincial, Barcelona, Spain); Ana Martínez, Anna Martínez (Hospital de Campdevànol, Campdevànol, Spain); Lourdes Ferrer, M. José Moreno (Hospital Dos de Maig, Barcelona, Spain); Esther Calbo, Carolina Porta (Hospital Mútua de Terrassa, Terrassa, Spain); Alex Smithson (Hospital de l’Esperit Sant, Barcelona, Spain); Maria de la Roca Toda, Teresa Aliu (Hospital de Palamos, Palamós, Spain); Susanna Camps, Montserrat Ortega (Corporació Sanitària Parc Taulí, Sabadell, Spain); Vicens Diaz-Brito, Encarna Moreno (Hospital Sant Joan de Déu, Sant Boi, Spain); Carme Agustí, Miquel Perea Garcia (Hospital de Sant Celoni, Sant Celoni, Spain); Marta Andrés, Laura Grau Palafox (Hospital de Terrassa, Terrassa, Spain); Raquel Carrera, Anna Besolí (Consorci Hospitalari, Vic, Spain); Juan Pablo Horcajada, Cristina Gonzalez (Hospital del Mar/Esperança, Barcelona, Spain); Jordi Cuquet, Demelsa Maria Maldonado López (Hospital General, Granollers, Spain); Rosa Benítez, Mirella Duch (Serveis Assistencials, Badalona, Spain); David Blancas, Esther Moreno (Consorci Sanitari del Garraf, Vilanova, Spain); Naiara Villalba, Sara Martínez (Hospital Sant Bernabé, Berga, Spain); Àngels García Flores, Roser Ferrer (Hospital Sant Jaume, Calella, Spain); Josep Bisbe, Montse Blascó (Hospital Sant Jaume, Olot, Spain); Antoni Castro Salom, Ana Felisa López (Hospital Universitari Sant Joan, Reus, Spain); Joan Espinach Alvarós, Àngels Perez (Hospital Sant Joan de Deu, Martorell, Spain); David Castander, Elisabet Calaf (Hospital Sant Pau i Santa Tecla, Tarragona, Spain); Mercè Clarós (Hospital Sant Rafael, Barcelona, Spain); Núria Bosch Ros (Hospital Sta. Caterina, Girona, Spain); Irene Montardit (Hospital Sta. Maria, Lleida, Spain); Roser Porta, Pilar De la Cruz Sol (Hospital Universitari Quirón Dexeus, Barcelona, Spain); Mª Rosa Coll Colell, Rosa García Penche Sanches (Hospital Universitari Sagrat Cor, Barcelona, Spain); Josep Maria Tricas, Eva Redon (Fundació Privada Hopstital de Mollet, Mollet, Spain); Montse Brugués (Consorci Sanitari de l’Anoia, Igualada, Spain); Laura Linares, Maria Cusco (Hospital Comarcal de l’Alt Penedés, Vilafranca, Spain); Mª Pilar Barrufet‚ Elena Vidal (Consorci Hospitalari del Maresme, Mataró, Spain); Sandra Barbadillo, Mariló Marimón (Hospital Universitari General de Catalunya, Sant Cugat, Spain); Yolanda Meije, Montserrat Vaqué (Hospital de Barcelona, Barcelona, Spain); M. Rosa Laplace Enguinados, Blanca Vila (Hospital del Vendrell, El Vendrell, Spain); Ana Guadalupe Coloma, Lucrecia López (Hospital Moisés Broggi, Sant Joan Despí, Spain); Magda Campins, Carme Ferrer, Benito Almirante (Hospital Universitari. Vall d’Hebrón, Barcelona, Spain); Natalia Juan Serra, Josep Farguell Carrera (Hospital Quiron Salud Barcelona, Spain); Àngels Garcia Flores, Roser Ferrer (Hospital Comarcal de Blanes, Blanes, Spain); Marta Milian Sanz, Alexandra Moise (Pius Hospital de Valls, Valls, Spain); Ana Mª Jiménez Zarate, M. Carmen Eito Navasal (Institut Català d’Oncologia, L’Hospitalet, Spain); Maria Gracia Garcia Ramirez (Centre mèdicoquirúrgic, Reus, Spain); Ana Mª Jiménez Zarate (Institut Català d’Oncologia, Girona, Spain); Ana Mª Jiménez Zarate, Mar Armario Fernández (Institut Català d’Oncologia, Badalona, Spain).
References
- Huang C, Wang Y, Li X, Ren L, Zhao J, Hu Y, et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet. 2020;395:497–506. DOIPubMedGoogle Scholar
- World Health Organization. Coronavirus disease 2019 (COVID-19) situation report—52 [cited 2022 Sep 21]. https://www.who.int/docs/default-source/coronaviruse/situation-reports/20200312-sitrep-52-covid-19.pdf
- Weiner-Lastinger LM, Dudeck MA, Allen-Bridson K, Dantes R, Gross C, Nkwata A, et al. Changes in the number of intensive care unit beds in US hospitals during the early months of the coronavirus disease 2019 (COVID-19) pandemic. Infect Control Hosp Epidemiol. 2021;•••:1–5. DOIPubMedGoogle Scholar
- Centers for Disease Control and Prevention. Infection control guidance for healthcare professionals about coronavirus (COVID-19) [cited 2022 Sep 21]. https://www.cdc.gov/coronavirus/2019-ncov/hcp/infection-control.html
- Bentivegna E, Alessio G, Spuntarelli V, Luciani M, Santino I, Simmaco M, et al. Impact of COVID-19 prevention measures on risk of health care-associated Clostridium difficile infection. Am J Infect Control. 2021;49:640–2. DOIPubMedGoogle Scholar
- Ponce-Alonso M, Sáez de la Fuente J, Rincón-Carlavilla A, Moreno-Nunez P, Martínez-García L, Escudero-Sánchez R, et al. Impact of the coronavirus disease 2019 (COVID-19) pandemic on nosocomial Clostridioides difficile infection. Infect Control Hosp Epidemiol. 2021;42:406–10. DOIPubMedGoogle Scholar
- Chacón-Quesada T, Rohde V, von der Brelie C. Less surgical site infections in neurosurgery during COVID-19 times-one potential benefit of the pandemic? Neurosurg Rev. 2021;44:3421–5. DOIPubMedGoogle Scholar
- Losurdo P, Paiano L, Samardzic N, Germani P, Bernardi L, Borelli M, et al. Impact of lockdown for SARS-CoV-2 (COVID-19) on surgical site infection rates: a monocentric observational cohort study. Updates Surg. 2020;72:1263–71. DOIPubMedGoogle Scholar
- Baccolini V, Migliara G, Isonne C, Dorelli B, Barone LC, Giannini D, et al. The impact of the COVID-19 pandemic on healthcare-associated infections in intensive care unit patients: a retrospective cohort study. Antimicrob Resist Infect Control. 2021;10:87. DOIPubMedGoogle Scholar
- Baker MA, Sands KE, Huang SS, Kleinman K, Septimus EJ, Varma N, et al.; CDC Prevention Epicenters Program. The impact of coronavirus disease 2019 (COVID-19) on healthcare-associated infections. Clin Infect Dis. 2022;74:1748–54. DOIPubMedGoogle Scholar
- Weiner-Lastinger LM, Pattabiraman V, Konnor RY, Patel PR, Wong E, Xu SY, et al. The impact of coronavirus disease 2019 (COVID-19) on healthcare-associated infections in 2020: A summary of data reported to the National Healthcare Safety Network. Infect Control Hosp Epidemiol. 2022;43:12–25. DOIPubMedGoogle Scholar
- Bhargava A, Riederer K, Sharma M, Fukushima EA, Johnson L, Saravolatz L. High rate of Multidrug-Resistant Organisms (MDROs) among COVID-19 patients presenting with bacteremia upon hospital admission. Am J Infect Control. 2021;49:1441–2. DOIPubMedGoogle Scholar
- Zarb P, Coignard B, Griskeviciene J, Muller A, Vankerckhoven V, Weist K, et al.; National Contact Points for the ECDC pilot point prevalence survey; Hospital Contact Points for the ECDC pilot point prevalence survey. The European Centre for Disease Prevention and Control (ECDC) pilot point prevalence survey of healthcare-associated infections and antimicrobial use. Euro Surveill. 2012;17:20316. DOIPubMedGoogle Scholar
- Zingg W, Metsini A, Balmelli C, Neofytos D, Behnke M, Gardiol C, et al. National point prevalence survey on healthcare-associated infections in acute care hospitals, Switzerland, 2017. Euro Surveill. 2019;24:
1800603 . - Zhong Y, Zhou L, Liu X, Deng L, Wu R, Xia Z, et al. Incidence, risk factors, and attributable mortality of catheter-related bloodstream infections in the intensive care unit after suspected catheters infection: a retrospective 10-year cohort study. Infect Dis Ther. 2021;10:985–99. DOIPubMedGoogle Scholar
- Chou EH, Mann S, Hsu TC, Hsu WT, Liu CCY, Bhakta T, et al. Incidence, trends, and outcomes of infection sites among hospitalizations of sepsis: A nationwide study. PLoS One. 2020;15:
e0227752 . DOIPubMedGoogle Scholar - O’Grady NP, Alexander M, Burns LA, Dellinger EP, Garland J, Heard SO, et al.; Healthcare Infection Control Practices Advisory Committee. Guidelines for the prevention of intravascular catheter-related infections. Am J Infect Control. 2011;39(Suppl 1):S1–34. DOIPubMedGoogle Scholar
- European Union. Council recommendation of 9 June 2009 on patient safety, including the prevention and control of healthcare associated infections [cited 2022 Sep 21]. https://op.europa.eu/en/publication-detail/-/publication/8ae80abf-31cd-4577-b0be-4f2fe108f6f9
- Centers for Disease Control and Prevention. National Healthcare Safety Network (NHSN) patient safety component manual. 2022 January [cited 2022 Sep 21]. https://www.cdc.gov/nhsn/pdfs/pscmanual/pcsmanual_current.pdf
- Centers for Disease Control and Prevention. Current HAI progress report. 2020 National and state healthcare-associated infections progress report [cited 2022 Sep 21]. https://www.cdc.gov/hai/data/portal/progress-report.html
- Catalan Health Service. VINCat program [cited 2022 Sep 21]. https://catsalut.gencat.cat/ca/proveidors-professionals/vincat
- Pronovost P, Needham D, Berenholtz S, Sinopoli D, Chu H, Cosgrove S, et al. An intervention to decrease catheter-related bloodstream infections in the ICU. N Engl J Med. 2006;355:2725–32. DOIPubMedGoogle Scholar
- Palomar M, Álvarez-Lerma F, Riera A, Díaz MT, Torres F, Agra Y, et al.; Bacteremia Zero Working Group. Impact of a national multimodal intervention to prevent catheter-related bloodstream infection in the ICU: the Spanish experience. Crit Care Med. 2013;41:2364–72. DOIPubMedGoogle Scholar
- van der Kooi T, Sax H, Pittet D, van Dissel J, van Benthem B, Walder B, et al.; PROHIBIT consortium. Prevention of hospital infections by intervention and training (PROHIBIT): results of a pan-European cluster-randomized multicentre study to reduce central venous catheter-related bloodstream infections. Intensive Care Med. 2018;44:48–60. DOIPubMedGoogle Scholar
- Badia-Cebada L, Peñafiel J, Saliba P, Andrés M, Càmara J, Domenech D, et al. VINCat programme (Infection Control Catalan Programme). Trends in the epidemiology of catheter-related bloodstream infections; towards a paradigm shift, Spain, 2007 to 2019. Euro Surveill. 2022;27:
2100610 . DOIGoogle Scholar - Kumar G, Adams A, Hererra M, Rojas ER, Singh V, Sakhuja A, et al. Predictors and outcomes of healthcare-associated infections in COVID-19 patients. Int J Infect Dis. 2021;104:287–92. DOIPubMedGoogle Scholar
- Abelenda-Alonso G, Rombauts A, Gudiol C, Oriol I, Simonetti A, Coloma A, et al. Immunomodulatory therapy, risk factors and outcomes of hospital-acquired bloodstream infection in patients with severe COVID-19 pneumonia: a Spanish case-control matched multicentre study (BACTCOVID). Clin Microbiol Infect. 2021;27:1685–92. DOIPubMedGoogle Scholar
- Lo SH, Lin CY, Hung CT, He JJ, Lu PL. The impact of universal face masking and enhanced hand hygiene for COVID-19 disease prevention on the incidence of hospital-acquired infections in a Taiwanese hospital. Int J Infect Dis. 2021;104:15–8. DOIPubMedGoogle Scholar
- World Health Organization. Global patient safety action plan 2021–2030 [cited 2022 Sep 21]. https://www.who.int/teams/integrated-health-services/patient-safety/policy/global-patient-safety-action-plan
Figures
Tables
Cite This ArticleOriginal Publication Date: October 03, 2022
Table of Contents – Volume 28, Number 11—November 2022
| EID Search Options |
|---|
|
|
|
|
|
|


Please use the form below to submit correspondence to the authors or contact them at the following address:
Oriol Gasch, Hospital Parc Taulí de Sabadell, Institut d’Investigació i Innovació Parc Taulí (IL3PT), Universitat Autònoma de Barcelona, 08208 Sabadell, Spain
Top