Volume 31, Number 6—June 2025
Research
Prospective Multicenter Surveillance of Non–H. pylori Helicobacter Infections during Medical Checkups, Japan
Cite This Article
Citation for Media
Abstract
To evaluate non–H. pylori Helicobacter (NHPH) infections in Japan, we enrolled 673 consecutive patients who underwent gastric endoscopy during annual medical checkups at 4 hospitals during April 2022–February 2023. We collected intragastric fluid and serum samples to detect NHPH infection by PCR and serologic tests. The prevalence of NHPH was 3% (20/673); 70% (14/20) of patients were infected with H. suis and 30% (6/20) with non–H. suis NHPH species. All 14 H. suis–infected patients were men and had a history of pork offal ingestion. Among non–H. suis NHPH-infected patients, 50% (3/6) owned pet cats, whereas only 22% (145/667) of other patients owned cats. Endoscopic evaluation revealed marbled crack-like gastritis was present in 93% (13/14) of H. suis–infected patients, a significantly higher prevalence than for H. pylori–infected (28.6%) and H. pylori eradication therapy (27.6%) groups. Pork offal ingestion and having pet cats increase risk for Helicobacter spp. infections.
Non–H. pylori Helicobacter (NHPH) species are helical corkscrew-like bacteria; Helicobacter spp. have been reported since the 1980s to infect the human stomach (1). In humans, NHPH infection causes gastric diseases, such as peptic ulcers, chronic gastritis, gastric cancer, and gastric mucosa-associated lymphoid tissue (MALT) lymphoma (2–5). Although H. suis is the most common NHPH species that infects the human stomach (4,6,7), its pathogenicity remains unclear because isolation has not previously been feasible. The recent successful isolation of H. suis from patients with gastric disease demonstrated H. suis pathogenicity in the human stomach (8), ushering in a new stage of NHPH research.
NHPH infection rates have mostly been analyzed in patients with specific gastric-related diseases (2–5,9–12). Most studies have used gastric biopsy to detect NHPH infection, making it difficult from an ethical standpoint to investigate NHPH infection in asymptomatic persons. Recent studies have shown the usefulness of gastric juice samples for H. pylori diagnoses (13,14), and we recently developed a method to detect NHPH infections by using gastric juice samples (15,16) without performing a biopsy. In addition, we developed a highly sensitive serologic diagnostic method for H. suis infection in humans (15,16). We conducted a multicenter survey of NHPH infection among patients who underwent gastric endoscopy during medical checkups in Japan by using those methods. We evaluated NHPH infection rates and regional differences, identified bacterial species, and determined associations between NHPH stomach infections, demographics, and endoscopic findings.
Ethics
We explained the study to all participants and obtained written informed consent from each. The study was approved by the Ethics Committees of Kyorin University Hospital, Junpukai Health Maintenance Center (Kurashiki, Japan), Kenwakai Hospital, and Hokkaido Cancer Society (approval no. 810, project no. R03-052-52) and registered with the University Hospital Medical Information Network, Clinical Trials Registry (UMIN registration no. UMIN000054538).
Study Design, Inclusion Criteria, and Sample Collection
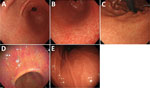
We enrolled consecutive patients who underwent an upper endoscopy during annual medical checkups at 4 hospitals in Japan during April 2022–February 2023. Hospital A is in Hokkaido, hospital B is in Tokyo, hospital C is in Nagano, and hospital D is in Okayama. We excluded patients who underwent gastrectomy. We collected gastric washes (intragastric fluid) and serum samples from all patients to test for NHPH infection by PCR (fluids) and for H. suis by ELISA (serum samples). We collected the following clinical information: age; sex; endoscopic findings; and histories of H. pylori infection, H. pylori eradication therapy, pork offal ingestion, and pet ownership. We assessed the endoscopic grade of gastric mucosal atrophy by using the Kimura and Takemoto classification (17). To verify the endoscopic characteristics of NHPH infection, we confirmed the presence or absence of white-marbled appearance (18) and crack-like mucosa (11). We defined white-marbled appearance as a mottled mucosa with a white mesh pattern from the gastric antrum to the angulus (Figure 1, panel A; Appendix Figure 1) and crack-like mucosa as a mesh pattern composed of faded, depressed, and varying width lines on a coarse surface from the gastric antrum to angulus (Figure 1, panel B; Appendix Figure 1). Although both appearances are similar to atrophy, they have a white mesh; however, the crack-like mucosa appearance also has depressions in the white mesh. We defined a white-marbled with crack-like mucosa appearance as marbled crack-like (MARC) gastritis. In NHPH-infected patients, we also examined the presence and location of nodular gastritis from the antrum to angulus (Figure 1, panel C), spotty redness from the antrum to the body (Figure 1, panel D), and the regular arrangement of collecting venules (RAC; Figure 1, panel E) (19). We defined endoscopic findings as normal if no atrophic gastritis, peptic ulcers, gastric cancer, nodular gastritis, white-marble appearance, or crack-like mucosal gastritis were present.
We divided H. pylori infections into the following 3 groups: infected with H. pylori (H. pylori infection group), history of H. pylori infection treated with eradication therapy (posteradication group), and no history of H. pylori infection (no H. pylori infection group). Diagnosis of H. pylori infection was confirmed by endoscopy physicians (K.T., K.M., T.K., and M.K.) with >10 years of experience in diagnosing H. pylori infection, according to results of a urea breath test, serum H. pylori antibody test, fecal antigen test, rapid urease test, histologic studies, and endoscopic observation, according to the Kyoto classification of gastritis (20). The posteradication group also included patients with no history of H. pylori eradication but with an endoscopically suspected history of H. pylori infection, according to the Kyoto classification of gastritis.
NHPH Detection by PCR and ELISA
We detected NHPH infection by PCR of DNA extracted from intragastric fluid collected during endoscopic examination. We centrifuged 1 mL of intragastric fluid and collected the precipitates for DNA extraction by using the DNeasy Blood and Tissue Kit (QIAGEN, https://www.qiagen.com). We performed PCR by using NHPH species–specific primers targeting 16S rRNA and H. suis–specific primers targeting the hsvA gene, as previously described (8). We determined the samples to be H. suis positive by using both PCR primer sets. However, if PCR targeting 16S rRNA for NHPH species was positive and PCR targeting hsvA was negative, we identified those samples as non–H. suis NHPH positive.
We measured serum H. suis antibody titers as previously described (15,16). In brief, we determined H. suis antibody titers by using ELISA and calculated the cutoff index (COI) value as the ratio of the absorbance of the sample to the absorbance of the positive control, both at 450 nm. We deemed the samples as positive when COI values were >1.
Statistics
We examined associations between clinical characteristics of patients and the specific status of Helicobacter infection (i.e., infection by H. suis, non–H. suis NHPH, or H. pylori, as well as after H. pylori eradication and absence of Helicobacter infection without eradication therapy) by using the Fisher exact test for discrete characteristics and Wilcoxon rank-sum test for continuous characteristics. We also examined the association between specific hospitals and Helicobacter infection status by using the Fisher exact test. We evaluated differences in age and H. suis ELISA titers between groups by using the Kruskal-Wallis test. We used SPSS Statistics 29 (IBM, https://www.ibm.com) and R version 4.3.2 (The R Project for Statistical Computing, https://www.r-project.org) for statistical analyses and set statistical significance at p<0.05.
Prevalence of NHPH Infection
Five patients were excluded from the analysis; 2 at hospital B and 1 at hospital D underwent remnant gastrectomy, and intragastric fluid or serum could not be obtained from 2 other patients at hospital B. We included 673 participants in the study: 100 at hospital A, 198 at hospital B, 199 at hospital C, and 179 at hospital D. The median patient age was 55 (range 29–91) years; 57.6% were male and 42.4% female (Table 1).
Intragastric fluid from all patients was tested for NHPH infection. NHPH infections were found in 3% (20/673) of patients; H. suis infection was found in 70% (14/20) and non–H. suis NHPH infections in 30% (6/20) of those patients. Among the 20 patients, 15 were in the no H. pylori infection group, 3 in the H. pylori infection group, and 2 in the posteradication group. Diagnoses of the 3 NHPH-positive patients in the H. pylori infection group were made by using either the urea breath test, serum H. pylori antibody test, or endoscopic findings. However, we found those patients to be H. pylori negative by PCR targeting the H. pylori–specific region of the 16S rRNA gene (21). Therefore, those 3 patients were considered to be false positives for H. pylori because of their NHPH infection. The 2 NHPH-positive patients in the posteradication group had no history of H. pylori eradication; 1 of those patients was H. pylori negative by serologic and histologic analyses, and both showed H. pylori–negative status by PCR. Therefore, both patients were placed in the posteradication group according to endoscopic observations of NHPH infection and Kyoto classification of gastritis. After detecting NHPH species in intragastric fluid by PCR, patients were recategorized as follows: no Helicobacter infection, n = 378; posteradication, n = 254; H. pylori infection, n = 21; H. suis infection, n = 14; and non–H. suis NHPH infection, n = 6. The proportion of patients with H. suis infections was significantly higher in hospital C than in the other hospitals (p<10−5) (Table 2).
Comparison of Clinical Characteristics among Groups
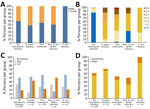
We compared clinical characteristics of patients in each of the recategorized groups (Table 3). The H. suis infection group consisted entirely of men (100%), which differed from the combined 4 groups not infected with H. suis (57% men; p = 0.0005) (Figure 2, panel A). When compared with the non–H. suis NHPH infection group (66.7%), the percentage of atrophic gastritis was 100% in the H. suis (p = 0.04) and H. pylori (p = 0.04) infection groups and 97.6% in the posteradication group (p = 0.01) (Table 3). The percentage of nodular gastritis was 42.9% for H. suis, 50.0% for non–H. suis NHPH, and 19.0% for H. pylori infection groups (p = 0.2 overall). The overall percentage of nodular gastritis among those 3 groups was 32%, which was significantly higher than that in the no Helicobacter infection (0%; p<0.0001) and posteradication (2.8%; p<0.0001) groups. In addition, a white marbled appearance (18) was observed in 78.6% and crack-like mucosa (11) in 85.7% of the H. suis infection group, indicating endoscopic characteristics of H. suis infection. The percentage of patients with at least 1 of the 2 endoscopic characteristics of NHPH infection was 92.9% (13/14) among the H. suis–infected patients, which was significantly higher than that among H. pylori–infected (28.6% [6/21]; p = 0.0003) and posteradication (27.6% [70/254]; p<0.0001) groups.
The atrophic grade of patients with H. suis infection was mostly C-2, and no patient had an atrophic grade greater than C-2. Among H. pylori–infected patients, most had atrophic grades of C-2; however, some had O-1 (4.8%) and O-2 (9.5%) grades (Table 3; Figure 2, panel B).
Among the non–H. suis NHPH-infected patients, 50% (3/6) had a history of owning cats, which was higher than the 22% (145/667) observed among the other patients in the study. However, the difference was not statistically significant (p = 0.12) because of the small sample size of non–H. suis NHPH-infected patients (Figure 2, panel C). All (14/14) H. suis–infected patients had a pork offal ingestion history, a proportion that was significantly higher than that in the combined 4 groups not infected with H. suis (384/659; p = 0.0006) (Figure 2, panel D).
Endoscopic Characteristics of Patients with NHPH Infection
We determined the type and site of gastritis and the presence or absence of RAC in the 20 NHPH-infected patients (Appendix Table). In each patient, gastritis was observed from the glandular border around the angulus to the pyloric antrum (Figure 1; Appendix Figure 1). Endoscopic gastritis results differed by NHPH species. The percentages of MARC gastritis were significantly different between the H. suis–infected (92.9% [13/14]) and non–H. suis NHPH–infected (33.3% [2/6]) groups (p = 0.014), whereas spotty redness was observed at similar rates in the H. suis–infected (35.7% [5/14]) and non–H. suis NHPH–infected (50.0% [3/6]) groups (p = 0.6). Gastritis of the pyloric canal was found in 50.0% (7/14) of the H. suis infection group, whereas it was absent in 83.3% (5/6) of the non–H. suis NHPH infection group, although the difference was not statistically significant (p = 0.3). RAC was absent in 1 patient who had petechial erythema spread over the entire gastric body.
H. suis ELISA
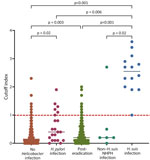
The COI for the H. suis ELISA was significantly higher for the H. suis infection group than for the other groups (Figure 3). All patients in the H. suis infection group were considered H. suis positive according to H. suis ELISA criteria (16). In the non–H. suis NHPH infection group, 1 (16.7%) patient was H. suis positive and 5 (83.3%) were H. suis negative, according to ELISA. We determined that 28.6% (6/21) of patients in the H. pylori infection, 7.1% (18/254) in the posteradication, and 3.4% (13/378) in the no Helicobacter infection groups were H. suis positive, according to H. suis ELISA. Among the 13 H. suis–positive patients in the no Helicobacter infection group, atrophic gastritis was found in 3 patients, suggesting the possibility of previous H. suis infection or false-negative results by PCR of intragastric fluid.
In this study of consecutive patients who underwent gastric endoscopy during medical checkups in Japan, the NHPH infection rate was 3%. NHPH infection rates of 0.25%–6.1% have been reported previously (2–5,9–11), indicating that rates among healthy persons are not much lower than among patients with gastric diseases. A 10-year study on NHPH infection rates in a single institution in Japan showed the infection rate increased from 1.3% to 3.35% (30 of 896 patients) in the last 2 years and 4 months of the study period because of increased recognition of characteristic endoscopic findings (11). Therefore, NHPH infections might be missed in many patients with gastric diseases. In our survey, the H. pylori infection rate was 3.6%, and 38% of patients had a history of H. pylori infection. The estimated H. pylori infection rate in persons born in Japan in 1968 was reported to be 37.5% (22). Considering the median age (55 years) of the patients in our study, our sample represents a major section of the population in Japan.
Gastric NHPH species have been found in patients with gastric diseases by using culture tests, including H. suis (8), which also infects pigs and monkeys, and H. ailrurogastricus (23), H. felis (24), and H. bizzozeronii (25,26), which infect cats and dogs. Other species that infect dogs and cats, such as H. heilmannii and H. salomonis, have also been detected by PCR in patients with gastric diseases (2–5,9–12). H. suis is the most prevalent NHPH species infecting humans in Japan and other countries, whereas other species infecting dogs and cats have also been detected in most studies (2–5,9–12). In this study, H. suis was the most prevalent NHPH species (14/20), consistent with previous reports in Japan (4,11).
All patients infected with H. suis in this study were men. The percentage of men in the group infected with non–H. suis NHPH species was 50%, similar (57.8%) to that in the entire tested study population. Other studies have also demonstrated a high prevalence of H. suis infections among men (11). Future studies should investigate the difference in prevalence of H. suis infection between men and women by focusing on aspects such as the transmission route of infection. The natural hosts of H. suis are pigs and monkeys, and pork meat is suspected to be a carrier of H. suis infection to humans. In this study, a relationship between the history of pork offal ingestion and H. suis infection was strongly suggested. Because a history of pork offal ingestion was found in only 40% of the no Helicobacter infection group, it was not always a risk factor for H. suis infection. However, hospital C, which had the highest percentage of H. suis infections, is located in an area where pork and pork offal are commonly eaten. A survey held in Japan in 2008 showed by using PCR that 74% of slaughtered sows were infected with Helicobacter spp. (27); however, updated surveys are needed to identify the current prevalence of H. suis infection in pigs.
Among NHPH-infected patients, 6 were infected with non–H. suis NHPH species. The non–H. suis NHPH species that infect dogs and cats are H. felis, H. bizzozeronii, H. heilmannii, H. ailirogastricus, H. cynogastricus, H. baculiformis, and H. salomonis. A survey of NHPH infections in dogs and cats in Japan indicated that 34.7% of dogs and 50% of cats were infected with NHPH species (28,29). Compared with other groups, 50% of patients infected with non–H. suis NHPH species had pet cats. Because not all patients had pets, pets might not be the only risk factor for non–H. suis NHPH infection. Multiple reports have suggested that non–H. suis NHPH species are transmitted from dogs and cats to humans (23–26), and studies demonstrating the genetic identity between strains from humans and pets will be needed to confirm that transmission. Species identification was not possible in this study because of the small number of bacterial cells in the samples. Further analysis using culture tests will be needed to identify non–H. suis NHPH species infecting humans.
No mixed infections of NHPH and H. pylori were observed; very low prevalences of mixed infections have been previously reported (7,30). Because NHPH infection was not observed in the posteradication group, H. pylori infection history might prevent NHPH infection, although further investigation is needed to clarify this possibility. Alternatively, NHPH might have also been eradicated by H. pylori eradication treatment. This study was conducted with patients who had undergone health checks in Japan; therefore, a substantial proportion of study participants had a history of H. pylori eradication therapy. Further analysis of a population with no history of H. pylori infection will be needed to clarify whether H. pylori infection prevents NHPH infection.
After we developed serologic tests for H. suis infection (16), we compared those results with PCR results. Serologic tests for H. suis infection revealed high detection sensitivity. Some patients in the no Helicobacter infection group tested positive in serologic tests for H. suis infection, and some of those patients had atrophic gastritis. Serologic tests will be useful to confirm PCR results and might help to detect overlooked H. suis infections. The serologic test for H. suis infection had low reactivity with the serum samples obtained from non–H. suis NHPH–infected patients. PCR remains the standard method for detecting NHPH infection, and a diagnostic method for non–H. suis NHPH infection is required. Some H. pylori–infected patients and patients with a history of H. pylori eradication showed positive serologic test results for H. suis infection, possibly because of cross-reactivity between H. pylori and NHPH species or NHPH infection might have been overlooked by PCR because of extremely low bacterial counts. Alternatively, the patients might have had a history of NHPH infection. Various possibilities exist; therefore, interviews regarding H. pylori eradication history are critical for accurately diagnosing NHPH infection by PCR and serologic tests. In addition to diagnostic testing, recognizing endoscopic characteristics of NHPH gastritis is essential. NHPH gastritis is antrum predominant and causes milder atrophy than H. pylori gastritis (2).
In this study, 94.1% (16/17) of patients with NHPH gastritis without H. pylori infection had RAC in the gastric corpus and had a Kimura-Takemoto classification of C-2 or lower, indicating that NHPH gastritis was localized within the angulus-antrum region and rarely extended to the gastric corpus (indicated by the presence of RAC). In addition, endoscopic gastritis of the pyloric canal was not observed in 60.0% of patients, suggesting that NHPH gastritis progresses from the angulus to the antrum, which differs from H. pylori gastritis, in which atrophy progresses from the antrum to the gastric body (Appendix Figure 2). In H. pylori gastritis, the incidence of gastric cancer increases as atrophy extends to the gastric body (31); however, in NHPH gastritis, atrophy does not extend to the gastric body, suggesting that gastric cancer might be less common. Because all asymptomatic NHPH-infected patients had MARC gastritis, nodular gastritis, or spotty redness similar to endoscopic gastritis, epidemiologic studies can determine whether NHPH gastritis is an etiopathogenic agent of gastric MALT lymphoma and peptic ulcers.
We found endoscopic characteristics of NHPH infection (i.e., MARC gastritis) in 93% of the H. suis–infected patients, which was more frequent than for H. pylori-infected patients (29%) and patients in the posteradication group (28%). Those findings were not observed in the non–H. suis NHPH group. Endoscopists should suspect NHPH infection, especially from H. suis, when NHPH-specific endoscopic characteristics are found in H. pylori–negative patients. Nodular gastritis was found in 42.9% (6/14) of the H. suis–infected group, similar to the rate in the non–H. suis NHPH infected group (50.0%), and was more frequent than in H. pylori–infected patients (19.0%), although the difference was not significant. Differences in the pathogenesis and endoscopic findings between H. suis and non–H. suis NHPH infections might become more apparent as the number of patients increases.
The first limitation of our study is that the study population was highly health conscious and had a large number of patients who had undergone H. pylori eradication therapy. Because a difference in the prevalence of H. suis infection was detected among the study areas, the population background must have had a substantial influence on the prevalence of NHPH species. Continued surveys targeting other areas in Japan and other countries can provide comprehensive information to elucidate the prevalence of NHPH infections. Second, because the participants underwent medical checkups, the age of the target group was relatively high; very few young persons were in the study population. Although endoscopy cannot be performed at all ages, a study using serologic testing for H. suis infection might be useful to determine H. suis infection status in young persons.
In conclusion, the prevalence of NHPH infection in patients who underwent gastric endoscopy during medical checkups in Japan was 3.0%, similar to that of H. pylori infection (3.1%). Whether all NHPH-infected patients need to receive an eradication regimen remains controversial, as does the contribution of NHPH infections to gastric MALT lymphoma and peptic ulcers. In regions with decreasing rates of H. pylori infections, it is crucial to differentiate NHPH infections. Our findings provide critical information on the prevalence and endoscopic characteristics of NHPH infections in Japan. NHPH infections are zoonotic. Our findings that pork offal ingestion is a risk factor for H. suis infections and having pet cats increases risk for non–H. suis NHPH infection can help prevent gastric Helicobacter diseases.
Dr. Tokunaga is a professor in public health at Kyorin University School of Medicine in Japan. His main interests focus on gastric diseases, gastric Helicobacter infections, and preventive medicine.
Acknowledgment
This work was supported by the Japanese Society for Helicobacter Research, Japan Agency for Medical Research and Development (AMED grant nos. 23fk0108678 and 24ym0126140 to E.R.), Health Labour Sciences Research Grant (grant no. 21HA1005 to K.S., no. 24HA1002 to E.R.), and Ministry of Education, Culture, Sports, Science and Technology/Japan Society for the Promotion of Science KAKENHI program (grant no. 20K08365 to K.T., no. 23H02636 to E.R., and no. 24K11176 to K.T.). The authors declare no conflict of interest.
References
- Dent JC, McNulty CA, Uff JC, Wilkinson SP, Gear MW. Spiral organisms in the gastric antrum. Lancet. 1987;2:96. DOIPubMedGoogle Scholar
- Stolte M, Kroher G, Meining A, Morgner A, Bayerdörffer E, Bethke B. A comparison of Helicobacter pylori and H. heilmannii gastritis. A matched control study involving 404 patients. Scand J Gastroenterol. 1997;32:28–33. DOIPubMedGoogle Scholar
- Morgner A, Lehn N, Andersen LP, Thiede C, Bennedsen M, Trebesius K, et al. Helicobacter heilmannii-associated primary gastric low-grade MALT lymphoma: complete remission after curing the infection. Gastroenterology. 2000;118:821–8. DOIPubMedGoogle Scholar
- Nakamura M, Øverby A, Michimae H, Matsui H, Takahashi S, Mabe K, et al. PCR analysis and specific immunohistochemistry revealing a high prevalence of non-Helicobacter pylori Helicobacters in Helicobacter pylori-negative gastric disease patients in Japan: High susceptibility to an Hp eradication regimen. Helicobacter. 2020;25:
e12700 . DOIPubMedGoogle Scholar - Kadota H, Yuge R, Miyamoto R, Otani R, Takigawa H, Hayashi R, et al. Investigation of endoscopic findings in nine cases of Helicobacter suis-infected gastritis complicated by gastric mucosa-associated lymphoid tissue lymphoma. Helicobacter. 2022;27:
e12887 . DOIPubMedGoogle Scholar - Haesebrouck F, Pasmans F, Flahou B, Chiers K, Baele M, Meyns T, et al. Gastric helicobacters in domestic animals and nonhuman primates and their significance for human health. Clin Microbiol Rev. 2009;22:202–23. DOIPubMedGoogle Scholar
- De Groote D, Van Doorn LJ, Van den Bulck K, Vandamme P, Vieth M, Stolte M, et al. Detection of non-pylori Helicobacter species in “Helicobacter heilmannii”-infected humans. Helicobacter. 2005;10:398–406. DOIPubMedGoogle Scholar
- Rimbara E, Suzuki M, Matsui H, Nakamura M, Morimoto M, Sasakawa C, et al. Isolation and characterization of Helicobacter suis from human stomach. Proc Natl Acad Sci U S A. 2021;118:
e2026337118 . DOIPubMedGoogle Scholar - Heilmann KL, Borchard F. Gastritis due to spiral shaped bacteria other than Helicobacter pylori: clinical, histological, and ultrastructural findings. Gut. 1991;32:137–40. DOIPubMedGoogle Scholar
- Okiyama Y, Matsuzawa K, Hidaka E, Sano K, Akamatsu T, Ota H. Helicobacter heilmannii infection: clinical, endoscopic and histopathological features in Japanese patients. Pathol Int. 2005;55:398–404. DOIPubMedGoogle Scholar
- Tsukadaira T, Hayashi S, Ota H, Kobayashi N, Sekiguchi Y, Kodaira H, et al. Prevalence, clinical features, and esophagogastroduodenoscopy (EGD) findings of non-Helicobacter pylori Helicobacter infection: A study of 50 cases at a single facility in Japan. Helicobacter. 2021;26:
e12811 . DOIPubMedGoogle Scholar - Taillieu E, De Witte C, De Schepper H, Van Moerkercke W, Rutten S, Michiels S, et al. Clinical significance and impact of gastric non-Helicobacter pylori Helicobacter species in gastric disease. Aliment Pharmacol Ther. 2023;57:1432–44. DOIPubMedGoogle Scholar
- Rimbara E, Tamura R, Tanuma M, Noguchi N, Kawai T, Sasatsu M. Evaluation of clarithromycin resistance in Helicobacter pylori obtained from culture isolates, gastric juice, and feces. Helicobacter. 2009;14:156–7. DOIPubMedGoogle Scholar
- Tsuda M, Watanabe Y, Oikawa R, Watanabe R, Higashino M, Kubo K, et al. Clinical evaluation of a novel molecular diagnosis kit for detecting Helicobacter pylori and clarithromycin-resistant using intragastric fluid. Helicobacter. 2022;27:
e12933 . DOIPubMedGoogle Scholar - Matsui H, Suzuki M, Aoki S, Shibayama K, Tokunaga K, Suzuki H, et al. Protocol for detecting Helicobacter suis infection in gastric biopsies and serum by PCR and ELISA. STAR Protoc. 2023;4:
102556 . DOIPubMedGoogle Scholar - Matsui H, Rimbara E, Suzuki M, Tokunaga K, Suzuki H, Sano M, et al. Development of serological assays to identify Helicobacter suis and H. pylori infections. iScience. 2023;26:
106522 . DOIPubMedGoogle Scholar - Kimura K, Takemoto T. An endoscopic recognition of the atrophic border and its significance in chronic gastritis. Endoscopy. 1969;1:87–97. DOIGoogle Scholar
- Shiratori S, Mabe K, Yoshii S, Takakuwa Y, Sato M, Nakamura M, et al. Two cases of chronic gastritis with non-Helicobacter pylori Helicobacter infection. Intern Med. 2016;55:1865–9. DOIPubMedGoogle Scholar
- Yagi K, Nakamura A, Sekine A. Characteristic endoscopic and magnified endoscopic findings in the normal stomach without Helicobacter pylori infection. J Gastroenterol Hepatol. 2002;17:39–45. DOIPubMedGoogle Scholar
- Haruma K, Kato M, Inoue K, Murakami K, Kamada T, editors. Kyoto classification of gastritis. Tokyo: Nihon Medical Center; 2017.
- Okuda M, Osaki T, Kikuchi S, Ueda J, Lin Y, Yonezawa H, et al. Evaluation of a stool antigen test using a mAb for native catalase for diagnosis of Helicobacter pylori infection in children and adults. J Med Microbiol. 2014;63:1621–5. DOIPubMedGoogle Scholar
- Wang C, Nishiyama T, Kikuchi S, Inoue M, Sawada N, Tsugane S, et al. Changing trends in the prevalence of H. pylori infection in Japan (1908-2003): a systematic review and meta-regression analysis of 170,752 individuals. Sci Rep. 2017;7:15491. DOIPubMedGoogle Scholar
- Sano M, Rimbara E, Suzuki M, Matsui H, Hirai M, Aoki S, et al. Helicobacter ailurogastricus in patient with multiple refractory gastric ulcers, Japan. Emerg Infect Dis. 2023;29:833–5. DOIPubMedGoogle Scholar
- Wüppenhorst N, von Loewenich F, Hobmaier B, Vetter-Knoll M, Mohadjer S, Kist M. Culture of a gastric non-Helicobacter pylori Helicobacter from the stomach of a 14-year-old girl. Helicobacter. 2013;18:1–5. DOIPubMedGoogle Scholar
- Jalava K, On SL, Harrington CS, Andersen LP, Hänninen ML, Vandamme P. A cultured strain of “Helicobacter heilmannii,” a human gastric pathogen, identified as H. bizzozeronii: evidence for zoonotic potential of Helicobacter. Emerg Infect Dis. 2001;7:1036–8. DOIPubMedGoogle Scholar
- Kivistö R, Linros J, Rossi M, Rautelin H, Hänninen ML. Characterization of multiple Helicobacter bizzozeronii isolates from a Finnish patient with severe dyspeptic symptoms and chronic active gastritis. Helicobacter. 2010;15:58–66. DOIPubMedGoogle Scholar
- Kikuchi M, Iwaya O, Endo H, Sigeta F, Une M, Osaka S, et al. The presence of Helicobacter infection in sows. Nippon Juishikai Zasshi. 2004;57:310–2. DOIGoogle Scholar
- Kubota-Aizawa S, Ohno K, Kanemoto H, Nakashima K, Fukushima K, Uchida K, et al. Epidemiological study on feline gastric Helicobacter spp. in Japan. J Vet Med Sci. 2017;79:876–80. DOIPubMedGoogle Scholar
- Kubota-Aizawa S, Ohno K, Fukushima K, Kanemoto H, Nakashima K, Uchida K, et al. Epidemiological study of gastric Helicobacter spp. in dogs with gastrointestinal disease in Japan and diversity of Helicobacter heilmannii sensu stricto. Vet J. 2017;225:56–62. DOIPubMedGoogle Scholar
- Liu J, He L, Haesebrouck F, Gong Y, Flahou B, Cao Q, et al. Prevalence of coinfection with gastric non-Helicobacter pylori Helicobacter (NHPH) species in Helicobacter pylori-infected patients suffering from gastric disease in Beijing, China. Helicobacter. 2015;20:284–90. DOIPubMedGoogle Scholar
- Uemura N, Okamoto S, Yamamoto S, Matsumura N, Yamaguchi S, Yamakido M, et al. Helicobacter pylori infection and the development of gastric cancer. N Engl J Med. 2001;345:784–9. DOIPubMedGoogle Scholar
Figures
Tables
Cite This ArticleOriginal Publication Date: May 13, 2025
1These authors contributed equally to this article.
Table of Contents – Volume 31, Number 6—June 2025
| EID Search Options |
|---|
|
|
|
|
|
|
Please use the form below to submit correspondence to the authors or contact them at the following address:
Addresses for correspondence: Emiko Rimbara, Department of Bacteriology II, National Institute of Infectious Diseases; 4-7-1 Gakuen, Musashimurayama-shi, Tokyo 208-0011, Japan
Top