Volume 32, Number 2—February 2026
Research
Leptotrombidium imphalum Chiggers as Vector for Scrub Typhus in Human Settlements, India, 2022–2023
Cite This Article
Citation for Media
Abstract
Scrub typhus is a common bacterial infection in many parts of Asia. The causative agent, Orientia tsutsugamushi, is transmitted by trombiculid mite (chigger) larvae that require small mammals as maintaining hosts. We studied the prevalence of O. tsutsugamushi infection in mites and small mammals in villages and land surrounding them in South India to determine high-risk settings. We identified 12,431 mite larvae on 883 small mammals, 32% of which were bandicoot rats, 31% black rats, and 31% Asian house shrews. Leptotrombidium imphalum was the most common mite species and the only species associated with O. tsutsugamushi infection (prevalence 3.6%). Orientia infection increased with mite population size on a host. Host numbers, the L. imphalum index, and the prevalence of Orientia infection in chiggers were considerably higher within human settlements than in surrounding fields, suggesting that most human scrub typhus infection occurs inside villages rather than during agricultural work.
Scrub typhus is caused by intracellular bacteria of the genus Orientia (family Rickettsiaceae, order Rickettsiales) (1). Scrub typhus, caused by O. tsutsugamushi, occurs predominantly in South Asia, East Asia, and Southeast Asia (2). Severe infection is characterized by acute respiratory distress syndrome, shock, renal failure, and meningoencephalitis (3). Orientia bacteria are transmitted by the bite of trombiculid mite larvae (chiggers) (4) that use small mammals such as rodents and shrews as their primary maintaining hosts (4). Humans are regarded as accidental hosts. The India subcontinent is a region marked by a high occurrence of scrub typhus, accounting for up to 35% of hospital admissions for fever (3,5–9).
Groups regarded at risk for scrub typhus, including farmers and military personnel, are thought to acquire infected chiggers in agricultural fields and within disturbed ecosystems such as forest edges (4). However, agricultural and other outdoor activities were only weakly associated with scrub typhus in rural settings in South India (10,11). That finding was confirmed in a cohort study on risk factors for scrub typhus in the state of Tamil Nadu, in which agricultural activities, taking animals for grazing, firewood collection, and open defecation did not increase the risk for scrub typhus (12). Furthermore, persons residing at the edge of a village were not at a higher risk than those in the village center. To better understand scrub typhus transmission in South India, we trapped small mammals in highly endemic villages to estimate parameters of scrub typhus ecology.
We conducted our study in the context of a human cohort study on scrub typhus in 32,279 persons of all ages living in 37 scrub typhus–endemic villages in Vellore and Ranipet, 2 districts of Tamil Nadu (13). Study villages had an average size of ≈225 households; mean household size was 4.0. Approximately 55% of the population practiced part-time or full-time agriculture. The Institutional Animal Ethics Committee of the Christian Medical College Vellore (Ref 09/2019) and the Animal Welfare and Ethical Review Board of the London School of Hygiene and Tropical Medicine (Ref 2019-10) approved the study. We followed institutional and national (India and United Kingdom) standards of animal care and use.
Trapping of Small Mammals
For logistical reasons, we included in this study 25 of the 37 villages in the human cohort study that were within 45 minutes driving distance from the study center. We conducted trapping continuously during August 2022–September 2023, visiting each of the 25 villages twice. We aimed to visit each village once in the rainy season (approximately June–December) and once in the dry season (January–May). At each village visit, we set traps for 4 consecutive nights, Mondays through Thursdays, choosing different trap locations at each of the 2 visits. We used locally available single-capture cage traps, 27 cm × 15 cm × 12.5 cm, that contained a bowl of water and coconut and peanut butter as bait. We set traps in 3 different habitats: village center, village edge, and fields surrounding the village (Appendix Figure 1). We used Google Maps (https://www.google.com/maps) to visualize geographic village centers with households closest to the center chosen for trapping within the household compound but outside the house. We chose the 2 edges of the village nearest the village center and selected households located at the edge for trapping within the compound. We allowed for some flexibility in selecting the village edge to account for unevenly shaped village borders and access to surrounding fields.
We set traps in the field at >50 m from the village edge, measured from the houses on the village edge selected for trapping; we determined locations of the traps by the presence of trees to which traps were chained to reduce loss. Because success was highest in the center and lowest in the field, we set 4 traps in the center, 6 traps at the edge, and 10 traps in the fields to achieve similar catches in all 3 habitats. Traps were open during the night and closed during the day to reduce catching nontarget animals. Each morning, we inspected traps for catches and brought captured target animals (small mammals) to the study center inside the cage in an air-conditioned vehicle. At the study center, we euthanized the animals by carbon dioxide inhalation, then measured body, tail, hindfoot, and ear length for species identification, using published guidance (14). We inspected animals for any visible ectoparasites. We clipped their ears at the base and stored them in absolute ethanol at −70°C. For animals on which we found mite colonies on their hindlegs, we clipped and stored the hindlegs. We dissected the animals and removed spleen tissue for immediate storage at −70°C without ethanol (15).
Chigger Enumeration and Identification
We examined ears and legs of each animal under a stereomicroscope (StereoBlue EVO; Euromex, https://www.euromex.com). We enumerated chiggers on ears and legs separately; because the overall number found was large, we used samples of chiggers for species identification and molecular analysis. We selected up to 10 chiggers from each ear or leg for mounting on microscopy slides, using Hoyer medium for morphologic identification, and placed up to 20 in an Eppendorf tube in absolute ethanol for molecular analysis. To ensure that chigger species on slides and in Eppendorf tubes were approximately comparable, we alternately chose chiggers for slide mounting or molecular analysis in groups of up to 5. In shrews, we found more chiggers on hindlegs than ears. In those cases, we used chiggers from legs for mounting and for PCR; we counted chiggers found on ears but did not further process them.
We identified chiggers under a light microscope (iScope; Euromex) using a key for chigger mites in India (16). We confirmed identification of species from the genus Leptotrombidium and related genera using published keys (17,18).
DNA Extraction and Molecular Analysis
We extracted DNA from chiggers and spleen tissue using a DNeasy Blood & Tissue Kit (QIAGEN, https://www.qiagen.com). We used quantitative PCR to detect O. tsutsugamushi targeting the 47-kDa protein gene as previously described (19). For each morphologically identified rodent species, we selected 2 representative specimens for molecular barcoding (Appendix).
Statistical Analysis
We calculated the proportion of small mammals infested with chiggers by mammal and chigger species, as well as the chigger index (i.e., the mean number of chiggers per host) (4). To account for the sampling process of selecting a subset of chiggers for identification, we calculated the chigger index for different mammal and chigger species by extrapolation from the species composition observed for different locations on a host (each of 2 ears and hindlegs). We averaged the proportions of each chigger species from different locations on the same host and weighted the averages by the total number of chiggers on that location (mounted or not). We then applied the weighted average proportions to the total number of chiggers found on that host to arrive at the species-specific chigger index.
Similarly, we estimated the number of chiggers of each species in a pool undergoing PCR from the proportion of microscopically identified chiggers of that species at the same location from which chiggers for the PCR pool were selected. We calculated the association between the estimated number of chiggers of 1 species in the pool and O. tsutsugamushi PCR positivity of the same pool using generalized linear models (binomial family, identity link). We explored effect modification by habitat as a 3-category variable (village center, edge, field) using likelihood ratio tests. We estimated the prevalence of O. tsutsugamushi in individual chiggers, p, on the basis of pool size and pool positivity using complementary log-log models based on the formula (20)
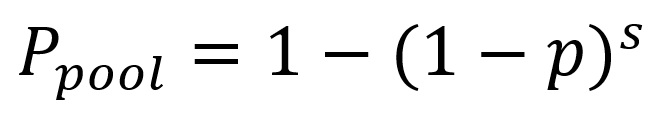
where Ppool is the proportion of pools that are positive for O. tsutsugamushi and s is the pool size (Appendix).
We estimated the association between the total number of Leptotrombidium imphalum chiggers on a host and the probability of O. tsutsugamushi PCR positivity of the chigger pool from the same host using logistic regression with PCR positivity as outcome and the estimated number of chiggers (chigger index) as explanatory variable, modeled as cubic splines with knots at 0, 100, and 200 chiggers. We resolved a pronounced right skew in the chigger index by applying the 4th root, and we adjusted for pool size because small mammals with a high chigger index tended to have larger pool sizes. We explored the association between chigger index and the probability of the spleen sample being PCR positive for O. tsutsugamushi. We expressed clustering of trap success and Orientia infection prevalence within villages as the intraclass correlation coefficient (ICC).
Small Mammal Trapping by Habitat and Season
We trapped 883 small mammals during 9,496 trap nights, an overall trap success of 9.3% (Table 1). Trap success was 20.3% in the village center (314 during 1,545 trap nights), 16.6% on the village edge (297/1,794) and 4.4% in the fields (272/6,152). There was some degree of clustering of the overall trap success within villages (ICC = 0.011; p = 0.001).
The greater bandicoot rat (Bandicota indica), the black rat (Rattus rattus), and the Asian house shrew (Suncus murinus) accounted for most of the trapped mammals, followed by mouse species (Mus spp.), three-striped Indian palm squirrel (Funambulus palmarum), Indian gerbil (Tatera indica), and Asian gray mongoose (Urva edwardsii). The bandicoot rats (GenBank accession no. PV918832) predominantly displayed an unusual tail to head/body ratio >1 (Appendix Figure 3). One mouse specimen (GenBank accession no. PV915568) showed 99.7% sequence identity with M. saxicola (GenBank accession no. MN964116). A further Mus specimen (accession no. PV915577) showed 98.3% identity with M. terricolor (accession no. KY018920). Greater bandicoot rats predominated in human habitats, whereas black rats, Indian gerbils, mice, and squirrels were more commonly trapped in the fields (Table 1). Asian house shrews were common in all 3 habitats.
In the fields, small mammal trapping showed pronounced seasonal variation with a low in the dry season and a high in October and November (Figure 1); we observed strong correlation between trappings and monthly human scrub typhus cases (r = 0.80). Trappings in village edge (r = −0.48) and center (r = −0.37) tended to be inversely correlated with scrub typhus cases.
Chigger Species by Host, Habitat, and Season
We collected 90,377 chiggers; of those, we identified 12,431 morphologically under the microscope and placed 10,740 in 759 pools for PCR analysis. L. imphalum was the most common chigger species, followed by Ericotrombidium bhattipadense and Schoengastiella ligula (Table 2). The L. imphalum indices were highest on Indian gerbils and house shrews. Otherwise, L. imphalum chiggers did not show a strong host preference, whereas E. bhattipadense and S. ligula chiggers had a strong host preference for B. indica rats. Trombicula hypodermata chiggers were found mainly on shrews, whereas Microtrombicula kajutekrii was mostly on black rats and Schoengastia tuberculata on bandicoot rats. One species of Hypotrombidium chiggers and 1 species of Walchia chiggers could not be identified to species level (Appendix Figures 4–7).
The total chigger index, as well as the indices for L. imphalum, E. bhattipadense, and S. ligula chiggers, were far higher in the village center and edge than in the fields (Figure 2). The L. imphalum index correlated moderately with monthly scrub typhus cases (r = 0.51) (Figure 3), whereas the E. bhattipadense index showed a strong negative correlation with scrub typhus, peaking in the hot months of April–July (r = −0.78). The S. ligula index showed no association (r = 0.08).
O. tsutsugamushi Infection by Chigger and Host Species
By real-time PCR, 122/759 pools (16.1%) tested positive for O. tsutsugamushi (Table 3). The coefficients indicate the change in the prevalence of positivity by PCR as a function of the expected number of chiggers of each species per pool, the latter extrapolated from the proportion of each chigger species among specimens examined by microscopy from the same ear or leg. Each additional L. imphalum chigger expected to be in a pool was associated with a 2.2% percentage point increase in the PCR pool positivity. By contrast, an increase in the number of most other chigger species was associated with a decreased probability of PCR positivity. Schoengastia tuberculata was associated with an increased PCR positivity, but with a wide confidence interval. L. imphalum chiggers were found concurrent with S. tuberculata in 9/13 locations where S. tuberculata was found on slides.
The percentage point increase in PCR positivity with each additional expected L. imphalum chigger was higher in the village center (+2.8%, 95% CI 2.0%–3.5%) and edge (+2.4, 95% CI 1.6%–3.1%) than in the field (+1.2%, 95% CI 0.5%–2.0%), with evidence for effect modification (p = 0.018). The overall prevalence of infection with O. tsutsugamushi among individual L. imphalum chiggers was 3.6%. The prevalence was 2.3 times higher in the village center (4.5%; prevalence ratio 2.3, 95% CI 1.3–4.1) and 2.2 times higher in the village edge (4.2%; prevalence ratio 2.2, 95% CI 1.2–3.8) than in the fields (1.9%). The prevalence of infection in chigger pools was highest in those from house shrews (29%), followed by bandicoot rats (14.3%) and black rats (8.5%) (Table 4).
We observed considerable clustering of chigger pool positivity within villages (ICC = 0.09; p<0.001), but no evidence of correlation between trap success and pool positivity by habitat (r = −0.09 for village center, r = −0.08 for edge, and r = −0.07 for fields).
We performed real-time PCR on spleen tissue of 874 small mammals, of which 99 (11.3%, 95% CI 9.3%–13.6%) tested positive for O. tsutsugamushi (Table 5). The prevalence of infection in spleen tissue was higher in small mammals caught in the field (15.2%) than in those caught at the village edge (10.3%) and center (8.9%; p = 0.019 by score test for trend), in contrast to infection in chiggers by location. The high prevalence of Orientia infection in spleen tissue of shrews in the fields (31%) is a likely cause of the trend (Table 5). Conversely, despite high prevalence of infection in chigger pools from bandicoot rats, the prevalence of infection in spleen tissue was 2.5%.
Adjusted for pool size, the prevalence of Orientia infection in chigger PCR pools increased with the estimated L. imphalum index on small mammals, leveling off at an index of ≈200 (Figure 4, panel A). By contrast, the association between the estimated L. imphalum index and prevalence of spleen tissue infection sharply plateaued at an L. imphalum index of ≈60 (Figure 4, panel B).
In this setting in South India where scrub typhus is a major public health problem (13), chigger numbers found on small mammals and the prevalence of O. tsutsugamushi infection in chiggers were higher within human settlements than in the land surrounding them. Several findings point to the peridomestic environment as a high-risk setting for infestation with O. tsutsugamushi–infected chigger mites. Trap success of small mammals, a proxy for density, was 4–5 times higher in village centers and edges than in the fields. Furthermore, chigger indices were higher in those locations, presumably because mite larvae more frequently encounter small mammals as suitable hosts (21), which they require to develop into the nymph and adult stages. L. imphalum chiggers, which have been implicated as a vector for scrub typhus in other foci across Asia (22,23), might be the dominant and perhaps only vector for scrub typhus in our setting. Of note, the estimated prevalence of O. tsutsugamushi infection in L. imphalum chiggers was more than twice as high in village center and edge than in the fields. Even when adjusting for pool size, a higher L. imphalum index, a potential marker of trombiculid mite density in the environment, was associated with a higher prevalence of infection in chigger pools (Figure 4, panel A). Our data suggest that, through mechanisms such as acquisition of infection from the host (24), cofeeding of chiggers (25,26), or horizontal transmission during other life stages, a high population density of Leptotrombidium mites may promote Orientia infection in the vector. Conclusive evidence only exists for vertical transmission (24,27).
A study in Yunnan, China, revealed close association between human habitats and Leptotrombidium deliense chiggers (28); that study and others from Thailand (29,30) found a low chigger species diversity in human settlements compared with less disturbed ecosystems. The occurrence of 1 Leptotrombidium chigger species, L. imphalum, in our study stands in contrast to earlier work from South India, which suggested a high Leptotrombidium chigger species diversity and an absence of L. imphalum chiggers in comparable settings (31–33). Future studies could include molecular barcoding to distinguish between morphologically similar species (34). In general, vector studies involving small mammals trapped in the peridomestic environment in South India have demonstrated high prevalence of Leptotrombidium infestation (33,35,36). Our study confirms that chigger mites regularly infest highly synanthropic small mammals, thus increasing the likelihood of infestation of humans.
As observed in earlier studies from India (36,37), S. murinus shrews carry large numbers of Leptotrombidium spp. chiggers (Table 2) and could play an important role in maintaining scrub typhus transmission. Bandicoot rats had lower L. imphalum indices than did the shrews but were highly synanthropic (Table 1) and likely to substantially contribute to L. imphalum density in villages. By contrast, the prevalence of infection in host spleen tissue plateaued at relatively low L. imphalum indices (Figure 4, panel B), suggesting that high exposure to O. tsutsugamushi might strengthen the immune response to the pathogen in hosts and enable them to rapidly clear the infection. The higher prevalence of Orientia infection in spleen tissue from hosts trapped in the field, compared with those from center and edge locations (Table 5), was driven by the high infection prevalence in house shrews, which might be less able to clear the infection than bandicoot rats.
Previous work has discussed causes of the strong seasonality of human scrub typhus in most settings (4,38). We found a strong correlation between human scrub typhus and trap success in the fields but not in village edge and center (Figure 1). The L. imphalum index correlated moderately with human scrub typhus, as in a study from Puducherry in South India (37). Small mammal trappings within villages increased in the dry season, inversely to human scrub typhus, suggesting that hosts may prefer the village environment in the dry season. Similarly, infestation with E. bhattipadense, the dominant chigger in the village center, correlated inversely with human scrub typhus. Trombiculid mites are able to complete their life cycle within burrows (39), perhaps independently of seasonal changes in temperature and humidity. Therefore, chigger numbers on small mammals may not accurately reflect human exposure to chiggers. Further, small mammals, in particular Asian house shrews (40), can have large home ranges (4) and can move between village and field (i.e., the trap location may be far away from the place where chiggers are acquired). Collecting questing chiggers in the environment may provide further insights, for example, by using the black plating method (41) in village center, edge, and field to estimate the risk for human exposure in each habitat, ideally complemented by human behavior studies with possible use of tracking devices. Such studies might also investigate the marked clustering of Orientia infection in chiggers we observed in our study and the observed variation in human scrub typhus risk and associated risk factors among study villages (12).
The seasonality of small mammal trappings might also be influenced by changes in food availability, especially in the dry season, when the baited traps could be more attractive. The bait used in this study, coconut with peanut butter, might attract host species differently. For example, Indian gerbils, common rodents in the Indian subcontinent that cause extensive crop damage (42), were caught in small numbers in this study. Given that it was the host with the highest L. imphalum index (Table 2), our results might have underestimated its role as a maintaining host of L. imphalum chiggers. Our study confirmed the utility of genetic barcoding in identifying common host species presenting with unusual morphology (Appendix Figure 3).
We based the species-specific chigger index on a relatively small sample of <20 mites per host undergoing morphologic identification. Furthermore, we included different mites for morphologic identification and O. tsutsugamushi detection using PCR, then estimated infection prevalence using regression models. Our approach does not rule out Orientia infection of a mite species at a very low prevalence. Ideally, the same chigger morphologically identified under the microscope would have been used for PCR, potentially following methods described previously (43), but that was not feasible given the large number of chiggers processed.
In conclusion, this study corroborates studies in South India suggesting human settlements as the setting in which most infestation with chigger mites infected with O. tsutsugamushi occurs (10–12,35). Our findings suggest a causal chain involving large numbers of small mammals in human settlements, enabling high trombiculid mite densities (21) and large populations of chigger mites, which might encourage the spread of O. tsutsugamushi within the vector population (25,26) and cause a high risk for human scrub typhus infection.
Dr. Devamani works as a doctor of community medicine at the Christian Medical College of Vellore. Her research interests include the epidemiology of rickettsial infections and fever etiology in rural settings.
Acknowledgments
We thank the residents of the participating villages in Vellore and Ranipet districts of Tamil Nadu, India for their support.
The study was supported by the Medical Research Council, UK (grant reference MR/S023275/1). A.A.S. was supported by the Ministry of Science and Higher Education of the Russian Federation (cooperative agreement no. 125013001089-0).
References
- Tamura A, Ohashi N, Urakami H, Miyamura S. Classification of Rickettsia tsutsugamushi in a new genus, Orientia gen. nov., as Orientia tsutsugamushi comb. nov. Int J Syst Bacteriol. 1995;45:589–91. DOIPubMedGoogle Scholar
- Kelly DJ, Fuerst PA, Ching WM, Richards AL. Scrub typhus: the geographic distribution of phenotypic and genotypic variants of Orientia tsutsugamushi. Clin Infect Dis. 2009;48(Suppl 3):S203–30. DOIPubMedGoogle Scholar
- Abhilash KP, Jeevan JA, Mitra S, Paul N, Murugan TP, Rangaraj A, et al. Acute undifferentiated febrile illness in patients presenting to a tertiary care hospital in South India: clinical spectrum and outcome. J Glob Infect Dis. 2016;8:147–54. DOIGoogle Scholar
- Elliott I, Pearson I, Dahal P, Thomas NV, Roberts T, Newton PN. Scrub typhus ecology: a systematic review of Orientia in vectors and hosts. Parasit Vectors. 2019;12:513. DOIGoogle Scholar
- Behera B, Biswal M, Das RR, Dey A, Jena J, Dhal S, et al. Clinico-epidemiological analysis of scrub typhus in hospitalised patients presenting with acute undifferentiated febrile illness: a hospital-based study from eastern India. Indian J Med Microbiol. 2019;37:278–80. DOIGoogle Scholar
- Bonell A, Lubell Y, Newton PN, Crump JA, Paris DH. Estimating the burden of scrub typhus: a systematic review. PLoS Negl Trop Dis. 2017;11:
e0005838 . DOIGoogle Scholar - Devamani CS, Prakash JAJ, Alexander N, Suzuki M, Schmidt WP. Hospitalisations and outpatient visits for undifferentiated fever attributable to scrub typhus in rural South India: retrospective cohort and nested case-control study. PLoS Negl Trop Dis. 2019;13:
e0007160 . DOIGoogle Scholar - Roopa KS, Karthika K, Sugumar M, Bammigatti C, Shamanna SB, Harish BN. Serodiagnosis of scrub typhus at a tertiary care hospital from southern India. J Clin Diagn Res. 2015;9:DC05–07. DOIGoogle Scholar
- Devasagayam E, Dayanand D, Kundu D, Kamath MS, Kirubakaran R, Varghese GM. The burden of scrub typhus in India: a systematic review. PLoS Negl Trop Dis. 2021;15:
e0009619 . DOIGoogle Scholar - Trowbridge PPD, Premkumar PS, Varghese GM. Prevalence and risk factors for scrub typhus in South India. Trop Med Int Health. 2017;22:576–82. DOIGoogle Scholar
- Devamani CS, Schmidt WP, Ariyoshi K, Anitha A, Kalaimani S, Prakash JAJ. Risk factors for scrub typhus, murine typhus, and spotted fever seropositivity in urban areas, rural plains, and peri-forest hill villages in South India: a cross-sectional study. Am J Trop Med Hyg. 2020;103:238–48. DOIGoogle Scholar
- Schmidt WP, Alexander N, Rose W, Chandramohan D, Cameron M, Abhilash K, et al. Risk factors for scrub typhus infection in South India: population-based cohort study. Epidemiol Infect. 2025;153:
e102 . DOIGoogle Scholar - Devamani C, Alexander N, Chandramohan D, Stenos J, Cameron M, Abhilash KPP, et al. Incidence of scrub typhus in rural South India. N Engl J Med. 2025;392:1089–99. DOIGoogle Scholar
- Agrawal VC. Taxonomic studies on Indian Muridae and Hystricidae. Kolkata: Zoological Survey of India; 2000.
- CERoPath. Community ecology of rodents and their pathogens in southeast Asia. 2024 [cited 2025 Jul 15]. http://www.ceropath.org
- Fernandes S, Kulkarni SM. Studies on the Trombiculid mite fauna of India. Kolkata: Zoological Survey of India; 2003.
- Stekolnikov AA. Leptotrombidium (Acari: Trombiculidae) of the world. Zootaxa. 2013;3728:1–173. DOIGoogle Scholar
- Vercammen-Grandjean PH, Langston R. The chigger mites of the world. Volume III. Leptotrombidium complex. San Francisco: George Williams Hooper Foundation, UCSF Reprographics Department; 1976.
- Jiang J, Chan TC, Temenak JJ, Dasch GA, Ching WM, Richards AL. Development of a quantitative real-time polymerase chain reaction assay specific for Orientia tsutsugamushi. Am J Trop Med Hyg. 2004;70:351–6. DOIGoogle Scholar
- Katholi CR, Toé L, Merriweather A, Unnasch TR. Determining the prevalence of Onchocerca volvulus infection in vector populations by polymerase chain reaction screening of pools of black flies. J Infect Dis. 1995;172:1414–7. DOIGoogle Scholar
- Patterson JE, Ruckstuhl KE. Parasite infection and host group size: a meta-analytical review. Parasitology. 2013;140:803–13. DOIGoogle Scholar
- Liu QY, Fan R, Song WY, Peng PY, Zhao YF, Jin DC, et al. The distribution and host-association of the vector chigger species Leptotrombidium imphalum in southwest China. Insects. 2024;15:504. DOIGoogle Scholar
- Tanskul P, Linthicum KJ. Redescription of Leptotrombidium (Leptotrombidium) imphalum (Acari: Trombiculidae), with observations on bionomics and medical importance in northern Thailand. J Med Entomol. 1999;36:88–91. DOIGoogle Scholar
- Traub R, Wisseman CL Jr, Jones MR, O’Keefe JJ. The acquisition of Rickettsia tsutsugamushi by chiggers (trombiculid mites) during the feeding process. Ann N Y Acad Sci. 1975;266:91–114. DOIGoogle Scholar
- Frances SP, Watcharapichat P, Phulsuksombati D, Tanskul P. Transmission of Orientia tsutsugamushi, the aetiological agent for scrub typhus, to co-feeding mites. Parasitology. 2000;120:601–7. DOIGoogle Scholar
- Takhampunya R, Korkusol A, Promsathaporn S, Tippayachai B, Leepitakrat S, Richards AL, et al. Heterogeneity of Orientia tsutsugamushi genotypes in field-collected trombiculid mites from wild-caught small mammals in Thailand. PLoS Negl Trop Dis. 2018;12:
e0006632 . DOIGoogle Scholar - Frances SP, Watcharapichat P, Phulsuksombati D. Vertical transmission of Orientia tsutsugamushi in two lines of naturally infected Leptotrombidium deliense (Acari: Trombiculidae). J Med Entomol. 2001;38:17–21. DOIGoogle Scholar
- Liu RJ, Guo XG, Zhao CF, Zhao YF, Peng PY, Jin DC. An ecological survey of chiggers (Acariformes: Trombiculidae) associated with small mammals in an epidemic focus of scrub typhus on the China–Myanmar border in southwest China. Insects. 2024;15:812. DOIGoogle Scholar
- Chaisiri K, Gill AC, Stekolnikov AA, Hinjoy S, McGarry JW, Darby AC, et al. Ecological and microbiological diversity of chigger mites, including vectors of scrub typhus, on small mammals across stratified habitats in Thailand. Anim Microbiome. 2019;1:18. DOIGoogle Scholar
- Wulandhari SA, Paladsing Y, Saesim W, Charoennitiwat V, Sonthayanon P, Kumlert R, et al. High prevalence and low diversity of chigger infestation in small mammals found in Bangkok metropolitan parks. Med Vet Entomol. 2021;35:534–46. DOIGoogle Scholar
- Govindarajan R, Sankar SG, Kumar MS, Rajamannar V, Krishnamoorthi R, Anand AAP, et al. Molecular detection of Orientia tsutsugamushi in ectoparasites & their small mammal hosts captured from scrub typhus endemic areas in Madurai district, India. Indian J Med Res. 2024;159:180–92. DOIGoogle Scholar
- Paulraj PS, Renu G, Ranganathan K, Veeramanoharan R, Kumar A. Ectoparasites diversity on rodents and shrews at scrub typhus endemic Vellore district of Tamil Nadu, India. J Arthropod Borne Dis. 2022;16:51–60. DOIGoogle Scholar
- Prakash JAJ, Kamarasu K, Samuel PP, Govindarajan R, Govindasamy P, Johnson LA, et al. Detection of Orientia tsutsugamushi in novel trombiculid mite species in northern Tamil Nadu, India: use of targeting the multicopy traD gene. J Med Entomol. 2022;59:693–9. DOIGoogle Scholar
- Rajasegaran P, Tan KK, Khoo JJ, Mansor MS, Ahmad Khusaini MKS, AbuBakar S, et al. Molecular species delimitation analysis of Leptotrombidium spp. and other chigger species parasitizing birds in Malaysia. J Med Entomol. 2025;62:1175–91. DOIGoogle Scholar
- Rose W, Kang G, Verghese VP, Candassamy S, Samuel P, Prakash JJA, et al. Risk factors for acquisition of scrub typhus in children admitted to a tertiary centre and its surrounding districts in South India: a case–control study. BMC Infect Dis. 2019;19:665. DOIGoogle Scholar
- Sadanandane C, Elango A, Panneer D, Mary KA, Kumar NP, Paily KP, et al. Seasonal abundance of Leptotrombidium deliense, the vector of scrub typhus, in areas reporting acute encephalitis syndrome in Gorakhpur district, Uttar Pradesh, India. Exp Appl Acarol. 2021;84:795–808. DOIGoogle Scholar
- Candasamy S, Ayyanar E, Paily K, Karthikeyan PA, Sundararajan A, Purushothaman J. Abundance & distribution of trombiculid mites & Orientia tsutsugamushi, the vectors & pathogen of scrub typhus in rodents & shrews collected from Puducherry & Tamil Nadu, India. Indian J Med Res. 2016;144:893–900. DOIGoogle Scholar
- Elliott I, Thangnimitchok N, Chaisiri K, Wangrangsimakul T, Jaiboon P, Day NPJ, et al. Orientia tsutsugamushi dynamics in vectors and hosts: ecology and risk factors for foci of scrub typhus transmission in northern Thailand. Parasit Vectors. 2021;14:540. DOIGoogle Scholar
- Nakamoto A, Nakanishi N. Home range, habitat selection, and activity of male Asian house shrews, Suncus murinus, on Okinawa-Jima Island. Mammal Study. 2013;38:147–53. DOIGoogle Scholar
- Jones BM. A method for studying the distribution and bionomics of trombiculid mites (Acarina: Trombidiidae). Parasitology. 1950;40:1–13. DOIGoogle Scholar
- Munawar N, Mahmood T, Akrim F, Fatima H, Farooq M, Irshad N, et al. Small rodent communities and their associated damage to wheat-groundnut agriculture systems. Braz J Biol. 2024;84:
e254445 . DOIGoogle Scholar - Kumlert R, Chaisiri K, Anantatat T, Stekolnikov AA, Morand S, Prasartvit A, et al. Autofluorescence microscopy for paired-matched morphological and molecular identification of individual chigger mites (Acari: Trombiculidae), the vectors of scrub typhus. PLoS One. 2018;13:
e0193163 . DOIGoogle Scholar
Figures
Tables
Cite This ArticleTable of Contents – Volume 32, Number 2—February 2026
| EID Search Options |
|---|
|
|
|
|
|
|




Please use the form below to submit correspondence to the authors or contact them at the following address:
Wolf-Peter Schmidt, Department for Disease Control, London School of Hygiene and Tropical Medicine, Keppel St, WC1E 7HT London, UK
Top