Volume 32, Number 3—March 2026
Research
Genetically Similar High-Risk Strains of Carbapenemase-Producing Enterobacterales in Humans and Companion Animals, United States
Cite This Article
Citation for Media
Abstract
To elucidate the zoonotic potential of carbapenemase-producing carbapenem-resistant Enterobacterales (CP-CRE) in US companion animals (i.e., dogs and cats), we queried the National Center for Biotechnology Pathogen Detection database to identify One Health clusters containing CP-CRE isolates from companion animals and humans. The 11 One Health clusters we found included most (69% [169/246]) publicly available CP-CRE sequences from US companion animals and were from 8 internationally disseminated, high-risk sequence types from 3 bacterial species (Escherichia coli, Klebsiella pneumoniae, and Enterobacter cloacae). All clustered isolates had New Delhi metallo-β-lactamase–family carbapenemases, and most (92%) carried the blaNDM-5 allele. The One Health clusters included several closely related subclusters with geographically linked isolates from both humans and companion animals. Those results suggest that CP-CRE is an emerging One Health issue and that direct or indirect transmission of CP-CRE is occurring between humans and companion animals in the United States.
Carbapenem-resistant Enterobacterales (CRE) are among the highest priority antimicrobial-resistant pathogen threats to public health in the United States and globally (1,2). Defined by resistance to the “last resort” carbapenem antibiotics, CRE infections are difficult to treat and associated with high mortality (3). CRE is a major cause of human healthcare-associated infections and have recently emerged as a clinical, and potentially zoonotic, pathogen in companion animals (i.e., dogs and cats) (4).
Enterobacterales are a taxonomic order of gram-negative bacteria that include commensal and pathogenic gastrointestinal tract organisms, such as Escherichia coli, Klebsiella pneumoniae, and Enterobacter spp. Carbapenem resistance in Enterobacterales species can be conferred by several different mechanisms; among those, acquisition of genes that encode carbapenemases (enzymes that inactivate carbapenems and other β-lactam antibiotics) represents the most serious public health threat (5). Because carbapenemase genes are often located on mobile genetic elements, they can spread rapidly through both horizontal transfer and clonal expansion (6,7). The 5 most common and widely disseminated carbapenemase families are K. pneumoniae carbapenemase (KPC), imipenemase metallo-β-lactamase, New Delhi metallo-β-lactamase (NDM), Verona integron-encoded metallo-β-lactamase, and oxacillinase (OXA) 48–like (8,9).
CRE isolates with carbapenemases from each of the 5 major families have been recovered from companion animals across the globe (10). In the United States, the earliest reported carbapenemase-producing CRE (CP-CRE) detections from companion animals were NDM-producing E. coli isolates collected during 2008–2009 (11). CP-CRE from several bacterial species and carbapenemase families have since been isolated from dogs and cats in multiple states (12–18). Although the prevalence of CP-CRE colonization (i.e., asymptomatic carriage in the gastrointestinal tract) in US companion animals was recently estimated to be only 0.2% (16), NDM-producing E. coli has caused several large outbreaks among dogs and cats in veterinary hospitals and animal rescue facilities beginning in 2018 (14,17,18).
Transmission of CP-CRE between humans and animals has been documented only a few times, 1 time in a household in Finland (19) and 2 times in veterinary hospitals in Europe and the United States (20,21). However, CP-CRE outbreaks in companion animals have included strains associated with outbreaks in human healthcare facilities, highlighting the potential for transmission between human and animal hosts (13,14,16,22). Thanks to the widespread adoption of whole-genome sequencing (WGS) for research, surveillance, and outbreak response, thousands of CP-CRE sequences from human and nonhuman sources are now publicly available. We leveraged those data to analyze the relatedness of strains circulating between humans and animals to elucidate the zoonotic potential of CP-CRE in companion animals in the United States. This activity was reviewed by the Centers for Disease Control and Prevention (CDC), deemed research not involving human subjects, and was conducted consistent with applicable federal law and CDC policy (see e.g., 45 C.F.R. part 46; 21 C.F.R. part 56; 42 U.S.C. §241(d), 5 U.S.C. §552a, 44 U.S.C. §3501 et seq.).
Companion Animal Isolate and One Health Cluster Identification
We queried the National Center for Biotechnology Information (NCBI) Pathogen Detection database (https://www.ncbi.nlm.nih.gov/pathogens) on October 24, 2023, to identify Enterobacterales isolates from US dogs and cats containing any of the 5 major carbapenemase gene families (23) (Figure 1). We designated Pathogen Detection clusters (predefined by NCBI as isolates within <25 allele differences from species-specific, whole-genome multilocus sequence typing schemes) containing CP-CRE collected from companion animal and human sources from the United States as One Health clusters for further analysis. We continued to add isolates to the One Health clusters through February 23, 2024.
Metadata and Epidemiologic Data Collection
We downloaded metadata for isolates belonging to One Health clusters from the Pathogen Detection database (including host species, location, isolation source, etc.). Additional anonymized isolate and patient characteristic data were obtained for analyses, including patient state of residence, isolation source, and specimen type (i.e., colonization or clinical test). When specimen type information was missing, rectal swab samples were categorized as colonization tests, and samples from all other body sites were considered clinical specimens. Those additional data were obtained from the CDC Antimicrobial Resistance Laboratory Network (AR Lab Network), the University of Pennsylvania Veterinary Diagnostic Laboratory, and the Microbiology Laboratory at Texas A&M University Veterinary Medical Teaching Hospital. Linked data were not available or not requested from 7 human-origin sequences and 4 animal-origin sequences; for those sequences only, we used the metadata available in Pathogen Detection. To ensure confidentiality, state of residence for both human and animal patients were classified only by their AR Lab Network region of residence (24).
Bioinformatics Analysis
For each One Health cluster, we downloaded available isolate sequence assemblies from NCBI or generated with SKESA version 3.0.0 (25) with reads downloaded from the NCBI Sequence Read Archive (https://www.ncbi.nlm.nih.gov/sra) for samples without available assemblies. We identified the multilocus sequence types (STs) for all isolates using mlst version 2.23.0 (https://github.com/tseemann/mlst) with PubMLST typing schemes (26). We determined genetic similarity among CP-CRE sequences of human-origin and animal-origin isolates within the same ST by core-genome multilocus sequence typing (cgMLST) to provide a standardized basis of comparison across multiple STs and cluster sizes. We applied publicly available cgMLST schemes for E. coli (2,513 loci) from EnteroBase (27) and K. pneumoniae (2,537 loci) from Institut Pasteur (28) as previously described (29). For Enterobacter cloacae, we constructed an ad hoc cgMLST scheme with 4,229 loci from the 41 CP-CRE isolates within the identified One Health clusters using Roary (https://github.com/rastanton/cgMLST_Scripts; 30). We constructed phylogenetic dendrograms from cgMLST allele differences using the unweighted pair group method with arithmetic mean. We annotated the cgMLST trees with AR Lab Network regions of collection and host species (dog, cat, and human) using iToL version 4.0 (https://github.com/tseemann/mlst). We calculated the cgMLST allele differences within each cluster for different host pairs (e.g., human–human, human–animal, animal–animal) and summarized them using statistics tools from NumPy (31).
Data Validation
We verified isolate host information with epidemiologic data. We excluded isolate sequences if they were from sources other than humans or companion animals, they were duplicate sequences from the same isolate, if an isolate was <3 cgMLST allele differences from another isolate collected from the same patient on the same day, or if the sequence was not from paired-end reads.
Dataset Generation
As of February 23, 2024, a total of 246 CP-CRE isolate sequences from US companion animals were available in the NCBI Pathogen Detection database (Figure 1), 26 isolates from cats (11%) and 220 isolates from dogs (89%). Most isolates harbored blaNDM (236 [96%]). Among the isolates with blaNDM, 56% (n = 131) were E. coli, 31% were E. cloacae (n = 72), and 14% were K. pneumoniae (n = 33). Nine isolates harbored blaKPC; 7 were E. cloacae, 1 was E. coli, and 1 was Klebsiella oxytoca. A single OXA-48–like–producing K. oxytoca isolate from companion animals was also identified.
Among the 246 US isolates from companion animals, 169 (69%) belonged to 11 One Health clusters (Table 1; Appendix 1 Table 1), which included 393 human-origin isolates. All clustered isolates were collected during January 2016–February 2024, a period that marked a rapid increase in the use of WGS; 10 times more US CP-CRE sequences were uploaded to NCBI in 2023 than were uploaded in 2016 (Appendix 2 Figure 1).
Isolate and Patient Characteristics
Among the 562 isolates in One Health CP-CRE clusters, E. coli was the most common species (88%, n = 493), followed by E. cloacae (7%, n = 41) and K. pneumoniae (5%, n = 28) (Table 2). All isolates harbored NDM-family carbapenemases; 92% (n = 519) had blaNDM-5 and 8% (n = 43) had blaNDM-7. Seven isolates from humans, all from the largest E. coli One Health cluster (ST167 cluster 3) (Table 2), also carried carbapenemases from different families (4 with OXA-48–like and 3 with KPC genes) (Appendix 1 Table 2) in addition to blaNDM-5.
Among isolates with available data, 75% (126/168) of companion animal-origin isolate sequences were collected for colonization screening, compared with 5% (21/388) of the human isolates (Table 1). Seventy-two percent of human clinical isolates were collected from urine (261/365), whereas those from companion animals were divided roughly equally among the respiratory tract (tracheal wash or bronchoalveolar lavage samples, 33%, 14/42), wounds (29%, 12/42), and urine (26%, 11/42).
The One Health cluster isolates were from 386 unique human patients and 158 companion animal patients. Compared with the broad geographic distribution of human-origin isolates, companion animal isolates were concentrated in 2 neighboring regions; 77% (119/154) were from the Mid-Atlantic or Northeast.
Cluster Characteristics and Genetic Analyses
Of the 11 One Health clusters, 8 were from E. coli. The 11 clusters were composed of 8 unique STs; 2 E. coli STs were associated with multiple clusters (ST162 [2 clusters] and ST167 [3 clusters]) (Table 2). The size of the clusters varied from 2 isolates (E. coli ST162 cluster 1) to 285 isolates (E. coli ST167 cluster 3) (Table 2). Seven clusters contained isolates collected from dogs, cats, and humans, whereas 4 contained isolates collected from humans and dogs only. The fraction of human isolates within clusters varied from 6% (K. pneumoniae ST11 [1/18]) to 96% (E. coli ST167 cluster 3 [275/285]). Of the CP-CRE isolates from companion animals that were not part of One Health clusters, 59% (49/77) were from the same STs as the One Health clusters (E. coli ST162 and ST167, K. pneumoniae ST307, and E. cloacae ST171) (Appendix 1 Table 1).
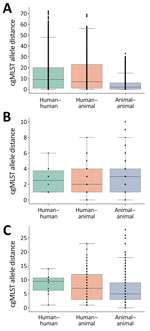
To investigate whether the isolates from humans and companion animals within One Health clusters were genetically distinct from one another, we compared cgMLST allele differences between human–human, human–animal, and animal–animal isolate pairs (examples in Figure 2). The interquartile ranges overlapped across all 3 pairwise categories. The median allele difference for human–animal pairs was lower than that for human–human pairs across all 3 CP-CRE species. Plots of the relative frequencies of within-cluster pairwise allele differences by CP-CRE species also showed overlapping human–human and human–animal pair distributions (Appendix 2 Figure 2).
Eight of the One Health clusters (representing each of the 3 species and all STs) contained human–animal isolate pairs that were 0–1 cgMLST allele differences apart (Appendix 1 Table 2). Seven of those clusters included human–animal pairs that were related within 0–1 cgMLST allele differences and collected from the same region (Figure 3).
Our analysis of CP-CRE sequences in the NCBI Pathogen Detection database found >240 isolates collected from US companion animals; nearly 70% clustered with isolates from humans. Those One Health clusters included isolates from 3 different bacterial species and 8 unique STs, and all harbored NDM-family carbapenemase genes. All cluster isolates were collected during a period that coincided with the rapid emergence of the NDM family of carbapenemase genes in US human patients (32,33). The One Health clusters included very closely genetically related isolate pairs from human and companion animals and many geographically linked genetic subclusters. Those findings support that emerging CP-CRE populations carried by companion animals are not genetically distinct from those isolated from humans and that strains are likely being shared among hosts.
Each of the STs identified in this analysis have been recognized as globally disseminated, high-risk strains (i.e., known to disseminate antimicrobial resistance genes) and have previously been isolated from companion animals (13,16,34–38). E. coli with blaNDM-5 was the most frequent species and carbapenemase allele combination in One Health clusters. Of the 5 E. coli STs (ST167, ST410, ST361, and ST617) identified, 4 are also among the most common NDM-5–producing human strains worldwide and were recently linked to community associated NDM-producing CRE cases in the United States (39–41). The most frequently identified of those, NDM-5–producing E. coli ST167, has caused outbreaks among companion animals at a veterinary hospital and an animal rescue facility in the United States and has been implicated in transmission between humans and companion animals in Europe (19,20).
Although our results provide evidence that exchange of CP-CRE between humans and companion animals is occurring in the United States, no established thresholds of relatedness (i.e., cgMLST allele differences) can be interpreted as absolute evidence of direct or indirect transmission (e.g., by exposure to a shared contaminated environment) or directionality (i.e., whether transmission occurred from humans to animals or vice versa) in the absence of clear epidemiologic links. The data do suggest that the emergence of CP-CRE among humans and companion animals in the United States is primarily driven by clonal expansion of strains that might be better suited for community spread, instead of horizontal transfer of carbapenemase genes into otherwise unrelated strains.
Most (75%) animal CP-CRE isolates in this study were found through colonization screening; those tests are used to identify persons or animals that might be asymptomatically shedding the organisms, usually to contain outbreaks or prevent introducing CP-CRE into healthcare facilities or veterinary hospitals (42). That finding confirms other reports that companion animals can silently carry zoonotic CP-CRE, which might accelerate spread of such organisms in community settings (43,44). Our findings of shared strains between companion animals and humans, as well as reports of outbreaks in veterinary facilities, highlight the potential risks of transmission to other companion animals, pet owners, and veterinary staff. Although the frequency of transmission is unknown, a study in Switzerland found 2 separate instances of veterinary hospital employees colonized with the same strain that had been identified in animals in their respective veterinary hospitals (20). That finding reinforces the importance of adhering to routine infection prevention and control measures to prevent spread within veterinary hospitals, among animal patients, and between animal patients and veterinary staff (14,18,20,37,45,46).
The first limitation of our study is that we used a convenience sample of publicly available WGS data, which are not representative of the true burden or characteristics of CP-CRE in companion animals or humans and might be skewed by the overrepresentation of closely related sequences associated with outbreaks (e.g., the E. coli sequences included dozens of sequences from 2 known companion animal outbreaks). In addition, most companion animal samples were from only 2 regions, the Mid-Atlantic and Northeast, the same regions in which CP-CRE outbreaks in US veterinary hospitals have been reported (14,18). Therefore, the results might underestimate the diversity and distribution of CP-CRE in companion animals across the United States.
Our results demonstrate that CP-CRE in companion animals and humans are genetically very similar and include many diverse, high-risk sequence types commonly associated with infections and outbreaks in human healthcare settings. That finding suggests that both companion animals and humans serve as reservoirs for high-risk CP-CRE strains; community reservoirs of historically healthcare-associated pathogens have the potential to increase CP-CRE infections in otherwise healthy humans and pets. Coordinated efforts between human and animal health sectors are warranted to mitigate further spread of such highly antimicrobial-resistant bacteria.
Dr. Xiaoli completed this work during her Molecular Epidemiology Fellowship at the Centers for Disease Control and Prevention. Her research interests include pathogen genomics, microbiology, and pathogenesis, with a focus on integrating multidisciplinary data to advance outbreak responses and disease surveillance.
Acknowledgments
We thank Sara Lawhon for her assistance in providing epidemiological information for a companion animal patient.
S.D.C. has received honoraria for speaking engagements and consultations for Idexx Laboratories and bioMérieux. He has also received travel support for his role as advisor and working group chairholder for the Clinical and Laboratory Standards Institute and for being a guest speaker for the Council of State and Territorial Epidemiologists.
References
- Centers for Disease Control and Prevention. Antibiotic resistance threats in the United States 2019 [cited 2025 Mar 10]. https://www.cdc.gov/antimicrobial-resistance/media/pdfs/2019-ar-threats-report-508.pdf
- World Health Organization. WHO bacterial priority pathogens list, 2024 [cited 2025 Mar 10]. https://iris.who.int/server/api/core/bitstreams/1a41ef7e-dd24-4ce6-a9a6-1573562e7f37/content
- Zhou R, Fang X, Zhang J, Zheng X, Shangguan S, Chen S, et al. Impact of carbapenem resistance on mortality in patients infected with Enterobacteriaceae: a systematic review and meta-analysis. BMJ Open. 2021;11:
e054971 . DOIPubMedGoogle Scholar - Silva JMD, Menezes J, Marques C, Pomba CF. Companion animals—an overlooked and misdiagnosed reservoir of carbapenem resistance. Antibiotics (Basel). 2022;11:533. DOIPubMedGoogle Scholar
- Gupta N, Limbago BM, Patel JB, Kallen AJ. Carbapenem-resistant Enterobacteriaceae: epidemiology and prevention. Clin Infect Dis. 2011;53:60–7. DOIPubMedGoogle Scholar
- Naas T, Cuzon G, Villegas MV, Lartigue MF, Quinn JP, Nordmann P. Genetic structures at the origin of acquisition of the β-lactamase bla KPC gene. Antimicrob Agents Chemother. 2008;52:1257–63. DOIPubMedGoogle Scholar
- Mathers AJ, Cox HL, Kitchel B, Bonatti H, Brassinga AK, Carroll J, et al. Molecular dissection of an outbreak of carbapenem-resistant enterobacteriaceae reveals Intergenus KPC carbapenemase transmission through a promiscuous plasmid. mBio. 2011;2:e00204–11. DOIPubMedGoogle Scholar
- Nordmann P, Poirel L. Epidemiology and diagnostics of carbapenem resistance in gram-negative bacteria. Clin Infect Dis. 2019;69(Suppl 7):S521–8. DOIPubMedGoogle Scholar
- Wise MG, Karlowsky JA, Mohamed N, Hermsen ED, Kamat S, Townsend A, et al. Global trends in carbapenem- and difficult-to-treat-resistance among World Health Organization priority bacterial pathogens: ATLAS surveillance program 2018-2022. J Glob Antimicrob Resist. 2024;37:168–75. DOIPubMedGoogle Scholar
- Sellera FP, Da Silva LCBA, Lincopan N. Rapid spread of critical priority carbapenemase-producing pathogens in companion animals: a One Health challenge for a post-pandemic world. J Antimicrob Chemother. 2021;76:2225–9. DOIPubMedGoogle Scholar
- Shaheen BW, Nayak R, Boothe DM. Emergence of a New Delhi metallo-β-lactamase (NDM-1)-encoding gene in clinical Escherichia coli isolates recovered from companion animals in the United States. Antimicrob Agents Chemother. 2013;57:2902–3. DOIPubMedGoogle Scholar
- Liu X, Thungrat K, Boothe DM. Occurrence of OXA-48 carbapenemase and other β-lactamase genes in ESBL-producing multidrug resistant Escherichia coli from dogs and cats in the United States, 2009–2013. Front Microbiol. 2016;7:1057. DOIPubMedGoogle Scholar
- Daniels JB, Chen L, Grooters SV, Mollenkopf DF, Mathys DA, Pancholi P, et al. Enterobacter cloacae complex sequence type 171 isolates expressing KPC-4 carbapenemase recovered from canine patients in Ohio. Antimicrob Agents Chemother. 2018;62:e01161–18. DOIPubMedGoogle Scholar
- Cole SD, Peak L, Tyson GH, Reimschuessel R, Ceric O, Rankin SC. New Delhi metallo-β-lactamase-5-producing Escherichia coli in companion animals, United States. Emerg Infect Dis. 2020;26:381–3. DOIPubMedGoogle Scholar
- Lavigne SH, Cole SD, Daidone C, Rankin SC. Risk factors for the acquisition of a blaNDM-5 carbapenem-resistant Escherichia coli in a veterinary hospital. J Am Anim Hosp Assoc. 2021;57:101–5. DOIPubMedGoogle Scholar
- Dietrich J, LeCuyer TE, Hendrix GK, Burbick CR, Jacob ME, Byrne BA, et al. Prevalence and molecular epidemiology of carbapenemase-producing Enterobacterales isolated from dog and cat faeces submitted to veterinary laboratories in the USA. Zoonoses Public Health. 2024;71:538–48. DOIPubMedGoogle Scholar
- McNamara K, Habrun C, Wilson W, Kollmann L, Stapleton G, Stanton R, et al. New Delhi metallo-β-lactamase–producing Escherichia coli among dogs at an animal rescue facility—Wisconsin, 2022. Antimicrob Steward Health Epidemiol. 2023;3:s90. DOIGoogle Scholar
- DeStefano I, Fellman CL, Bergeron AC, Golato LM, Doron S, Cumming M, et al. Effective mitigation of an outbreak of New Delhi metallo-β-lactamase-producing Escherichia coli infections in a small animal veterinary teaching hospital. J Am Vet Med Assoc. 2025;263:1–10. DOIPubMedGoogle Scholar
- Grönthal T, Österblad M, Eklund M, Jalava J, Nykäsenoja S, Pekkanen K, et al. Sharing more than friendship - transmission of NDM-5 ST167 and CTX-M-9 ST69 Escherichia coli between dogs and humans in a family, Finland, 2015. Euro Surveill. 2018;23:
1700497 . DOIPubMedGoogle Scholar - Endimiani A, Brilhante M, Bernasconi OJ, Perreten V, Schmidt JS, Dazio V, et al. Employees of Swiss veterinary clinics colonized with epidemic clones of carbapenemase-producing Escherichia coli. J Antimicrob Chemother. 2020;75:766–8. DOIPubMedGoogle Scholar
- DeStefano IM, Fellman CL, Vagnone PMS, Cumming MA, Dale JL, Ruhland A, et al. Human cases of carbapenemase-producing Escherichia coli linked to spread between animals and the environment in a veterinary facility—Massachusetts, USA, 2023. Clin Infect Dis. 2025;ciaf541. DOIGoogle Scholar
- Campos-Madueno EI, Moser AI, Jost G, Maffioli C, Bodmer T, Perreten V, et al. Carbapenemase-producing Klebsiella pneumoniae strains in Switzerland: human and non-human settings may share high-risk clones. J Glob Antimicrob Resist. 2022;28:206–15. DOIPubMedGoogle Scholar
- National Center for Biotechnology Information. Pathogen detection help document [cited 2025 May 2]. https://www.ncbi.nlm.nih.gov/pathogens/pathogens_help
- Centers for Disease Control and Prevention. Antimicrobial Resistance Laboratory Network [cited 2025 May 2]. https://www.cdc.gov/antimicrobial-resistance-laboratory-networks/php/about/domestic.html
- Souvorov A, Agarwala R, Lipman DJ. SKESA: strategic k-mer extension for scrupulous assemblies. Genome Biol. 2018;19:153. DOIPubMedGoogle Scholar
- Jolley KA, Bray JE, Maiden MCJ. Open-access bacterial population genomics: BIGSdb software, the PubMLST.org website and their applications. Wellcome Open Res. 2018;3:124. DOIPubMedGoogle Scholar
- Dyer NP, Päuker B, Baxter L, Gupta A, Bunk B, Overmann J, et al. EnteroBase in 2025: exploring the genomic epidemiology of bacterial pathogens. Nucleic Acids Res. 2025;53(D1):D757–62. DOIPubMedGoogle Scholar
- Bialek-Davenet S, Criscuolo A, Ailloud F, Passet V, Jones L, Delannoy-Vieillard A-S, et al. Genomic definition of hypervirulent and multidrug-resistant Klebsiella pneumoniae clonal groups. Emerg Infect Dis. 2014;20:1812–20. DOIPubMedGoogle Scholar
- Stanton RA, McAllister G, Daniels JB, Breaker E, Vlachos N, Gable P, et al. Development and application of a core genome multilocus sequence typing scheme for the health care-associated pathogen Pseudomonas aeruginosa. J Clin Microbiol. 2020;58:e00214–20. DOIPubMedGoogle Scholar
- Page AJ, Cummins CA, Hunt M, Wong VK, Reuter S, Holden MT, et al. Roary: rapid large-scale prokaryote pan genome analysis. Bioinformatics. 2015;31:3691–3. DOIPubMedGoogle Scholar
- Harris CR, Millman KJ, van der Walt SJ, Gommers R, Virtanen P, Cournapeau D, et al. Array programming with NumPy. Nature. 2020;585:357–62. DOIPubMedGoogle Scholar
- Sader HS, Mendes RE, Carvalhaes CG, Kimbrough JH, Castanheira M. Changing epidemiology of carbapenemases among carbapenem-resistant Enterobacterales from United States hospitals and the activity of aztreonam-avibactam against contemporary Enterobacterales (2019–2021). Open Forum Infect Dis. 2023;10:ofad046. DOIGoogle Scholar
- Devinney K, Burton N, Alroy KA, Crawley A, Da Costa-Carter CA, Kratz MM, et al. Notes from the field: increase in New Delhi metallo-β-lactamase-producing carbapenem-resistant Enterobacterales—New York City, 2019–2024. MMWR Morb Mortal Wkly Rep. 2025;74:401–3. DOIPubMedGoogle Scholar
- Caddey B, Fisher S, Barkema HW, Nobrega DB. Companions in antimicrobial resistance: examining transmission of common antimicrobial-resistant organisms between people and their dogs, cats, and horses. Clin Microbiol Rev. 2025;38:
e0014622 . DOIPubMedGoogle Scholar - de Mendieta JM, Argüello A, Menocal MA, Rapoport M, Albornoz E, Más J, et al. Emergence of NDM-producing Enterobacterales infections in companion animals from Argentina. BMC Vet Res. 2024;20:174. DOIPubMedGoogle Scholar
- Brookshire C, Robinson D, Seo K. Carbapenemase-producing ST307 Klebsiella pneumoniae in dogs. Am J Med Sci. 2023;365:S313–4. DOIGoogle Scholar
- Brilhante M, Gobeli Brawand S, Endimiani A, Rohrbach H, Kittl S, Willi B, et al. Two high-risk clones of carbapenemase-producing Klebsiella pneumoniae that cause infections in pets and are present in the environment of a veterinary referral hospital. J Antimicrob Chemother. 2021;76:1140–9. DOIPubMedGoogle Scholar
- Garcia-Fierro R, Drapeau A, Dazas M, Saras E, Rodrigues C, Brisse S, et al. Comparative phylogenomics of ESBL-, AmpC- and carbapenemase-producing Klebsiella pneumoniae originating from companion animals and humans. J Antimicrob Chemother. 2022;77:1263–71. DOIPubMedGoogle Scholar
- Linkevicius M, Bonnin RA, Alm E, Svartström O, Apfalter P, Hartl R, et al. Rapid cross-border emergence of NDM-5-producing Escherichia coli in the European Union/European Economic Area, 2012 to June 2022. Euro Surveill. 2023;28:
2300209 . DOIPubMedGoogle Scholar - Jones S, Stanton R, D’Angeli M, Brezak A, Sinkevitch J, Sredl M, et al. Community-associated New Delhi metallo-beta-lactamase-producing carbapenem-resistant Enterobacterales: multiple states, from September 2021 through September 2022. Infect Control Hosp Epidemiol. 2025;46:1–4. DOIPubMedGoogle Scholar
- Xia C, Yan R, Liu C, Zhai J, Zheng J, Chen W, et al. Epidemiological and genomic characteristics of global blaNDM-carrying Escherichia coli. Ann Clin Microbiol Antimicrob. 2024;23:58. DOIPubMedGoogle Scholar
- Centers for Disease Control and Prevention. MDRO prevention strategies [cited 2025 Aug 12]. https://www.cdc.gov/healthcare-associated-infections/php/preventing-mdros/mdro-prevention-strategies.html
- Dazio V, Nigg A, Schmidt JS, Brilhante M, Campos-Madueno EI, Mauri N, et al. Duration of carriage of multidrug-resistant bacteria in dogs and cats in veterinary care and co-carriage with their owners. One Health. 2021;13:
100322 . DOIPubMedGoogle Scholar - Nigg A, Brilhante M, Dazio V, Clément M, Collaud A, Gobeli Brawand S, et al. Shedding of OXA-181 carbapenemase-producing Escherichia coli from companion animals after hospitalisation in Switzerland: an outbreak in 2018. Euro Surveill. 2019;24:
1900071 . DOIPubMedGoogle Scholar - Schmidt JS, Kuster SP, Nigg A, Dazio V, Brilhante M, Rohrbach H, et al. Poor infection prevention and control standards are associated with environmental contamination with carbapenemase-producing Enterobacterales and other multidrug-resistant bacteria in Swiss companion animal clinics. Antimicrob Resist Infect Control. 2020;9:93. DOIPubMedGoogle Scholar
- Haenni M, Boulouis HJ, Lagrée AC, Drapeau A, Va F, Billet M, et al. Enterobacterales high-risk clones and plasmids spreading blaESBL/AmpC and blaOXA-48 genes within and between hospitalized dogs and their environment. J Antimicrob Chemother. 2022;77:2754–62. DOIPubMedGoogle Scholar
Figures
Tables
Cite This ArticleOriginal Publication Date: February 26, 2026
Table of Contents – Volume 32, Number 3—March 2026
| EID Search Options |
|---|
|
|
|
|
|
|


Please use the form below to submit correspondence to the authors or contact them at the following address:
Allison E. James, Centers for Disease Control and Prevention, 1600 Clifton Rd NE, Mailstop H20-3, Atlanta, GA 30329-4018, USA
Top