Volume 32, Number 3—March 2026
Research Letter
Mycobacterium nanjing sp. nov. Isolated from Cutaneous Infection, China
Cite This Article
Citation for Media
Abstract
We report a case of a cutaneous infection in an immunocompetent person in China caused by an uncharacterized Mycobacterium strain. The patient isolate was identified as a novel species by whole-genome sequencing. We propose Mycobacterium nanjing sp. nov. as the name for this new species.
Mycobacterial infections are a major public health concern and pose a continued threat to human health. The incidence and prevalence of nontuberculosis mycobacteria (NTM) infections are on the rise in certain regions and might surpass the rates of tuberculosis (1). In southeastern coastal China, the Mycobacterium avium complex, particularly M. intracellulare, predominates, followed by rapidly growing mycobacteria such as the M. abscessus complex. M. kansasii has been reported in some coastal cities (2). The relatively high NTM case count might be associated with warm climates, urban water systems, and improved laboratory detection (3–5). We report a case of cutaneous infection in Jiangsu Province, China, caused by an uncharacterized Mycobacterium strain, for which we propose the name Mycobacterium nanjing sp. nov.
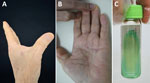
In 2025, an 86-year-old man was admitted to the outpatient department with a subcutaneous nodule on his left palm. The patient reported the nodule appeared 1 month earlier after a minor penetrating trauma to the left palm caused by a wooden splinter during carpentry work. Physical examination revealed 2 red subcutaneous nodules on the left hand, 1 in the palm and 1 on the back of the hand, each measuring 0.5 cm (Figure). Laboratory tests revealed a reduced erythrocyte count of 3.82 × 109 cells/L (reference range 4.3–5.8 × 1012 cells/L) and hemoglobin of 117 g/L (reference range 130–175 g/L). Serologic tests for syphilis, HIV, hepatitis B and C viruses, and tuberculosis were all negative.
Color doppler ultrasound revealed 3 hypoechoic masses in the dermis and subcutaneous fat layer of the left palm and back of hand lesions. The larger mass was in the palm, with uneven echo in the inner part, irregular shape, approximately 2.25 × 1.17 cm in size, approximately 0.93 cm in thickness, unclear boundary, no capsule, and enhanced echo in the surrounding soft tissue. Abundant blood flow was seen in and around the masses.
Histopathologic examination revealed hyperplasia of the epidermis and lymphocyte-dominated inflammatory cell infiltration in the superficial dermis. We conducted PCR testing on skin specimens by using common mycobacterial primers and cultured the samples on Löwenstein–Jensen slants. All PCRs included a no-template negative control that remained amplification-free. Direct PCR results were inconclusive. After 15 days of incubation at 37°C, we observed light yellow colonies on modified Löwenstein–Jensen medium slants (Figure). The organism was photochromogenic with a smooth appearance and slow growth rate.
We extracted DNA from the colonies for PCR analysis and aligned the sequences by using BLAST (https://blast.ncbi.nlm.nih.gov/Blast.cgi) for species identification. The partial sequence of the hsp65 gene (736-bp) shared greatest similarity with Mycolicibacterium gilvum Spyr1 (95.18%), and the rpoB gene (435-bp) shared greatest similarity with Mycolicibacterium vanbaalenii strain L1I3 (97.30%). On the basis of those results, we propose this strain might be a new member of the Mycobacterium genus.
For precise pathogen species identification, we subjected the patient isolate, ZZG, to whole-genome sequencing on the DNBSEQ platform (MGI, https://mgi-tech.eu) at the Beijing Genomics Institute (Shenzhen, China), yielding 1,177 Mb of data at 195× depth. Whole-genome sequencing yielded a genome assembly of 5.75 Mb (6,029,915 bp) for isolate ZZG, with an overall GC content of 67.87%. We submitted the whole genome to the type strain genome server (https://tygs.dsmz.de) to evaluate digital DNA-DNA hybridization with all available mycobacterial genomes (Table). The result suggests this strain is closest to M. vaccae (ATCC no. 15483). The digital DNA-DNA hybridization value is 28.9% and was calculated by using the genome-to-genome distance calculator formula, which is far from the threshold value for species delineation (70%). For phylogenetic analysis, the 5 species most closely related to isolate ZZG identified by the Type Strain Genome Server were involved; M. marinum (ATCC no. 927) was the outgroup. We constructed a phylogenetic tree from core genes by using RAxML (https://github.com/amkozlov/raxml-ng) under the general time-reversible plus invariable site plus discrete Gamma model with 1,000 bootstrap replicates, which demonstrated that strain ZZG was a distinct monophyletic clade, independent of its closest relatives (Appendix Figure, http://wwwnc.cdc.gov/EID/article/32/3/25-2001-App1.pdf). We propose Mycobacterium nanjing sp. nov. as the name for this potential new species.
We initially treated the patient with moxifloxacin (400 mg 1×/d), and clarithromycin (250 mg 2×/d) for 14 days. Antimicrobial drug susceptibility testing then revealed susceptibility to meropenem, linezolid, ciprofloxacin, moxifloxacin, tobramycin, minocycline, trimethoprim/sulfamethoxazole, doxycycline, amikacin, and rifabutin but resistance to rifampin and amoxicillin. We prescribed a roxithromycin regimen (150 mg 2×/d) for the patient on the basis of those results.
In summary, we isolated a distinct Mycobacterium species from a patient with a cutaneous infection. We propose the name Mycobacterium nanjing sp. nov. for this species. M. nanjing can cause disease in immunocompetent patients and shows susceptibility to multiple antimicrobial drugs. Despite the advantages of direct molecular detection in sensitivity and convenience, culture remains the standard and an indispensable component of a complementary diagnostic strategy, particularly for emerging mycobacterial species. The combination of surgical resolutions with antimicrobial therapy could be a good option for patients with Mycobacterium–caused cutaneous infections.
Ms. Zou is a medical student at the Institute of Dermatology, Chinese Academy of Medical Sciences and Peking Union Medical College in Nanjing, China. Her primary research interest is mycobacterial infectious diseases.
Acknowledgments
This whole-genome project has been deposited at the DNA Data Bank of Japan, the European Nucleotide Archive, and GenBank (accession nos. JBTBFV000000000 and JBTBFV010000000).
This study was supported by the Jiangsu Provincial Medical Key Laboratory, Jiangsu Province Capability Improvement Project through Science, Technology and Education (grant no. ZDXYS202204), National Natural Science Foundation of China (grant nos. 81972950, 82173431, 82103748), the Nanjing Incubation Program for National Clinical Research Center (grant no. 2019060001), and the Chinese Academy of Medical Sciences Innovation Fund for Medical Science (grant no. 2022-I2M-C&T-B-094).
References
- Shenjie T, Liang L, Xiaofeng Y, Meiying W. Guidelines for the diagnosis and treatment of nontuberculous mycobacteriosis (2020 edition). Chinese Journal of Tuberculosis and Respiratory. 2020;43:918–46. DOIGoogle Scholar
- Xu X, Lei Y, Zheng L. Non-tuberculous mycobacterial infections in mainland China and Taiwan: a systematic review and meta-analysis of epidemiology, species distribution, and drug resistance (2013-2024). Front Public Health. 2025;13:
1676715 . DOIPubMedGoogle Scholar - Faverio P, Del Castello L, Spotti M, Rebora P, Maggioni EC, Salerno R, et al.; METEO-NTM study group. How do climate conditions and urbanisation impact nontuberculous mycobacterial infections? An Italian nationwide case-control study. ERJ Open Res. 2025;11:00044–02025. DOIPubMedGoogle Scholar
- Dowdell KS, Potgieter SC, Olsen K, Lee S, Vedrin M, Caverly LJ, et al. Source-to-tap investigation of the occurrence of nontuberculous mycobacteria in a full-scale chloraminated drinking water system. Appl Environ Microbiol. 2024;90:
e0060924 . DOIPubMedGoogle Scholar - Wang L, Chen Y, Wang Q, Pan J, Bao R, Jin W, et al. Comparison of molecular testing methods for diagnosing non-tuberculous mycobacterial infections. Eur J Clin Microbiol Infect Dis. 2025;44:109–16. DOIPubMedGoogle Scholar
Figure
Table
Cite This ArticleOriginal Publication Date: March 10, 2026
1These first authors contributed equally to this article.
Table of Contents – Volume 32, Number 3—March 2026
| EID Search Options |
|---|
|
|
|
|
|
|
Please use the form below to submit correspondence to the authors or contact them at the following address:
Hongsheng Wang, Chinese Academy of Medical Sciences and Peking Union Medical College, St. 12 Jiangwangmiao, Nanjing, Jiangsu, China
Top