Volume 32, Number 5—May 2026
Research Letter
Borrelia turicatae in Ticks from Animals in a Public Park, Aguascalientes, Mexico
Cite This Article
Citation for Media
Abstract
We obtained 5 isolates of Borrelia turicatae from ticks captured in a public park in Aguascalientes, Mexico. A serologic survey in resident fauna showed antibodies against B. turicatae. Relapsing fever borrelias are present in Ornithodoros turicata ticks and circulate in a zoonotic cycle, posing a risk for human infection.
Ornithodoros turicata ticks were originally described in Mexico by Alfredo Dugès at the end of the 19th Century. In 1936, it was implicated as the vector of tick-borne relapsing fever (TBRF) when febrile patients were first described in the city of Aguascalientes, in central Mexico (1). Borrelia turicatae is the species of TBRF spirochete transmitted by O. turicata, an argasid tick has been found in multiple regions of the United States and Mexico (2,3). We isolated B. turicatae from O. turicata ticks captured in the northern state of Sinaloa and used the diagnostic recombinant glycerophosphodiester phosphodiesterase (rGlpQ) antigen to detect circulating antibodies in clinical patients (4,5). Those findings indicated that B. turicatae and its vector are endemic in regions of Mexico, with spillover into human populations. However, TBRF in Mexico and in many other regions worldwide is a neglected disease that is often misdiagnosed because its symptoms are frequently confused with those of other diseases, such as malaria and brucellosis (4).
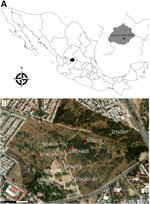
We obtained 5 isolates of B. turicatae from O. turicata ticks collected at La Pona Park in Aguascalientes, Mexico, in January and April 2023 (Figure 1). We examined tick specimens morphologically and molecularly by taxonomically analyzing a fragment of the mitochondrial genome, as described (6). Using a mouse model (either C57BL/6 or DBA/2J) (5), we evaluated the ticks by feeding them on the animals and assessing murine infection. We performed bacterial isolation as previously described by culturing blood samples from mice with spirochetemia in liquid, modified, and supplemented Barbour-Stoenner-Kelly II media supplemented with 10 µg/mL rifampin, 4 µg/mL phosphomycin, and 0.5 µg/mL amphotericin B (5). We amplified the 16S rRNA genes from each isolate by PCR, then sequenced and taxonomically analyzed them to confirm that all 5 isolates were B. turicatae; we named the isolates AGU1–AGU5. Of the 5 isolates, we sequenced genomic DNA from isolates AGU1–4 using NovaSeq X (Illumina, https://www.illumina.com); we used MinION for AGU1–3 and PromethION P2 Solo for AGU4 (Oxford Nanopore Technologies, https://nanoporetech.com). We base-called nanopore sequencing data using Dorado version 7.4.12 with the version 4.3.0 (https://github.com/nanoporetech/dorado) super-accurate base-calling model. We generated Illumina data using the Illumina DNA library prep kit 2 × 150 bp. We assembled chromosome-resolved and plasmid-resolved genome assemblies from Oxford Nanopore and Illumina data as previously described (7), with some modifications. We inferred a maximum-likelihood species tree as previously described; the tree demonstrated that AGU1–AGU4 clustered with B. turicatae 91E135, BTE5EL, and BTCAM1 strains (Appendix Figure). Both 91E135 and BTE5EL originated in Texas, USA, whereas BTCAM1 originated in Sinaloa, Mexico. AGU1–AGU5 are the southernmost B. turicatae isolates identified as of April 2026.
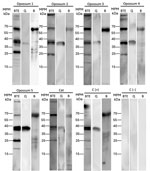
As part of our investigation, we captured wild fauna in La Pona public park to determine whether local animals were exposed to Borrelia spp. We collected blood samples in accordance with animal welfare guidelines (8). We captured 5 opossums (Didelphis virginiana), 1 deer mouse (Peromyscus sp.), and 1 feral cat (Felis silvestris catus) and tested serum samples by immunoblotting to detect antibodies against B. turicatae protein lysates, rGlpQ, and rBipA, as described previously (4,5). Results showed that the opossum and cat serum samples were reactive with B. turicatae protein lysates rGlpQ, and rBipA (Figure 2). None of the opossum samples were negative for the immunoblotting test, which might be a limitation of our study; however, opossum and cat serum samples from other states tested negative. Our results suggest that B. turicatae circulates in a tick–opossum–feral cat infectious cycle.
Our findings support efforts to understand the mechanisms maintaining B. turicatae in nature, the distribution of infected O. turicata ticks, and the public health impact. Although we identified endemic foci of infected ticks, we did not determine the prevalence of B. turicatae in these populations. Understanding prevalence is relevant to public health because B. turicatae can be vertically transmitted from female ticks to their offspring at rates as high as 40% (9). Public parks and human dwellings have been shown to be a source for infected soft ticks in the southern United States and likely in human infections (10), but that relationship has been scarcely studied in Mexico. Given that unhoused and underserved persons reside in La Pona public park, they are at risk for exposure to O. turicata ticks and infection with B. turicatae. Our future work will focus on defining the tick–vertebrate infectious cycle of B. turicatae and assessing its effect on human populations living in or around La Pona Park. Clinicians should be aware of this potential zoonotic risk to humans in the area.
Dr. Vázquez-Guerrero is a physician, pediatrician, and infectious disease specialist in Mexico. His areas of interest include vectorborne infectious diseases and microbiology.
Acknowledgments
We thank the authorities of the Aguascalientes city municipality for permission to collect ticks and capture animals in La Pona. We also thank our research groups at Laboratorio de Genética Microbiana and Baylor University.
The animal protocol followed in this study was approved by our Research Ethics Committee at ENCB Instituto Politécnico Nacional (no. ZOO-001-2022e1). Capture of wild fauna was approved by Secretaría de Medio Ambiente y Recursos Naturales, Government of Mexico (no. SGPA/DGVS/0564/21).
Borrelia turicatae sequencing data and genome assemblies were submitted to GenBank associated with BioProject PRJNA1338334.
Secretaría de Investigación y Posgrado IPN provided financial support for this study (project nos. 20250266, 20253924, and 20260917).
References
- Dugès A. Turicata of Guanajuato. Artículo en el periódico “El Repertorio” de Guanajuato. 1876;Sect. 11–12.
- Donaldson TG, Pèrez de León AA, Li AY, Castro-Arellano I, Wozniak E, Boyle WK, et al. Assessment of the geographic distribution of Ornithodoros turicata (Argasidae): climate variation and host diversity. PLoS Negl Trop Dis. 2016;10:
e0004383 . DOIPubMedGoogle Scholar - Guzmán-Cornejo C, Herrera-Mares A, Robbins RG, Rebollo-Hernández A. The soft ticks (Parasitiformes: Ixodida: Argasidae) of Mexico: species, hosts, and geographical distribution. Zootaxa. 2019;4623:3. DOIPubMedGoogle Scholar
- Vázquez-Guerrero E, Gordillo-Pérez G, Ríos-Sarabia N, Lopez JE, Ibarra JA. Case report: exposure to relapsing fever group borreliae in patients with undifferentiated febrile illness in Mexico. Am J Trop Med Hyg. 2023;108:510–2. DOIPubMedGoogle Scholar
- Vázquez-Guerrero E, Kneubehl AR, Pellegrini-Hernández P, González-Quiroz JL, Domínguez-López ML, Krishnavajhala A, et al. Borrelia turicatae from ticks in peridomestic setting, Camayeca, Mexico. Emerg Infect Dis. 2024;30:380–3. DOIPubMedGoogle Scholar
- Vázquez-Guerrero E, González-Quiroz JL, Domínguez-López ML, Kneubehl AR, Krishnavajhala A, Curtis MW, et al. New records of Ornithodoros turicata (Ixodida: Argasidae) in rural and urban sites in the Mexican states of Aguascalientes and Zacatecas indicate the potential for tick-borne relapsing fever. Exp Appl Acarol. 2023;91:99–110. DOIPubMedGoogle Scholar
- Kneubehl AR, Krishnavajhala A, Leal SM, Replogle AJ, Kingry LC, Bermúdez SE, et al. Comparative genomics of the Western Hemisphere soft tick-borne relapsing fever borreliae highlights extensive plasmid diversity. BMC Genomics. 2022;23:410. DOIPubMedGoogle Scholar
- Sikes RS; Animal Care and Use Committee of the American Society of Mammalogists. 2016 Guidelines of the American Society of Mammalogists for the use of wild mammals in research and education. J Mammal. 2016;97:663–88. DOIPubMedGoogle Scholar
- Filatov S, Krishnavajhala A, Lopez JE. Autogenous reproduction by Ornithodoros turicata (Ixodida: Argasidae) females and vertical transmission of the tick-borne pathogen Borrelia turicatae (Spirochaetales: Borreliaceae). Appl Environ Microbiol. 2023;89:
e0103223 . DOIPubMedGoogle Scholar - Bissett JD, Ledet S, Krishnavajhala A, Armstrong BA, Klioueva A, Sexton C, et al. Detection of tickborne relapsing fever spirochete, Austin, Texas, USA. Emerg Infect Dis. 2018;24:2003–9. DOIPubMedGoogle Scholar
Figures
Cite This ArticleOriginal Publication Date: April 28, 2026
1These authors contributed equally to this article.
Table of Contents – Volume 32, Number 5—May 2026
| EID Search Options |
|---|
|
|
|
|
|
|
Please use the form below to submit correspondence to the authors or contact them at the following address:
J. Antonio Ibarra, Instituto Politecnico Nacional, Microbiología Prol. Carpio y Plan de Ayala SN Col. Santo Tomas, Ciudad de México, 11340, Mexico;jibarrag@ipn.mx
Top