Volume 5, Number 6—December 1999
Perspective
Emerging Infectious Diseases and Amphibian Population Declines
Figure 2
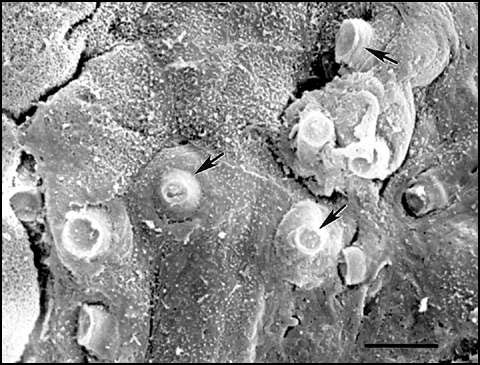
Figure 2. Scanning electron micrograph of digital skin of a wild frog (Litoria lesueuri, from Queensland, Australia) that died of cutaneous chytridiomycosis. Many cells within this area of the superficial layer of the epidermis contain mature sporangia, and unopened discharge tubes are visible protruding through infected cells. The skin was fixed in 2.5% glutaraldehyde, postfixed in 1% osmium tetroxide, dehydrated, critical-point-dried, sputter coated with gold, and examined with a JEOL JSM 840 scanning electron microscope at 5 kV. All specimens are from animals that were naturally infected and died due to chytridiomycosis in montane rain forest regions of Panama and Australia. Bar = 5 µm. Photo courtesy of L. Berger, reprinted with permission from A. Campbell (29).
References
- Daszak P, Cunningham AA, Hyatt AD. Emerging infectious diseases of wildlife—threats to human health and biodiversity. Science. 1999. In press.
- Lyles AM, Dobson AP. Infectious disease and intensive management: population dynamics, threatened hosts, and their parasites. J Zoo Wildl Med. 1993;24:315–26.
- Viggers KL, Lindenmayer DB, Spratt DM. The importance of disease in reintroduction programmes. Wildl Res. 1993;20:687–98. DOIGoogle Scholar
- Cunningham AA. Disease risks of wildlife translocations. Conserv Biol. 1996;10:349–53. DOIGoogle Scholar
- Plowright W. The effects of rinderpest and rinderpest control on wildlife in Africa. Symposia of the Zoological Society of London. 1982;50:1–28.
- Harvell CD, Kim K, Bukholder JM, Colwell RR, Epstein PR, Grimes DJ, Emerging marine diseases—climate links and anthropogenic factors. Science. 1999;285:1505–10. DOIPubMedGoogle Scholar
- Dobson AP, May RM. Disease and conservation. In: Soulé M, editor. Conservation biology: The science of scarcity and diversity. Sunderland (MA): Sinauer Assoc Inc.; 1986. p. 345-65.
- Scott ME. The impact of infection and disease on animal populations: Implications for conservation biology. Conserv Biol. 1988;2:40–56. DOIGoogle Scholar
- Daszak P, Cunningham AA. Wildlife disease biology comes of age. Oryx. 1998;32:238–9. DOIGoogle Scholar
- Anderson RM, May RM. The invasion, persistence and spread of infectious diseases within animal and plant communities. Philos Trans R Soc Lond B Biol Sci. 1986;314:533–70. DOIPubMedGoogle Scholar
- Tompkins DM, Begon M. Parasites can regulate wildlife populations. Parasitol Today. 1999;15:311–3. DOIPubMedGoogle Scholar
- Blaustein AR, Wake DB. Declining amphibian populations: A global phenomenon. Trends Ecol Evol. 1990;5:203–4. DOIGoogle Scholar
- Carey C. Hypothesis concerning the causes of the disappearance of boreal toads from the mountains of Colorado. Conserv Biol. 1993;7:355–62. DOIGoogle Scholar
- Drost CA, Fellers GM. Collapse of a regional frog fauna in the Yosemite area of the California Sierra Nevada, USA. Conserv Biol. 1996;10:414–25. DOIGoogle Scholar
- Cunningham AA, Langton TES, Bennett PM, Drury SEN, Gough RE, Kirkwood JK. Unusual mortality associated with poxvirus-like particles in frogs (Rana temporaria). Vet Rec. 1993;133:141–2.PubMedGoogle Scholar
- Cunningham AA, Langton TES, Bennett PM, Lewin JF, Drury SEN, Gough RE, Investigations into unusual mortalities of the common frog (Rana temporaria) in Britain. In: Proceedings of the 5th International Colloquium on the Pathology of Reptiles and Amphibians; 1995 March 3; The Netherlands. p. 19-27.
- Gupta BK. Declining amphibians. Curr Sci. 1998;75:81–4.
- Weygoldt P. Changes in the composition of mountain stream frog communities in the Atlantic Mountains of Brazil: Frogs as indicators of environmental deteriorations? Stud Neotrop Fauna Environ. 1989;24:249–55. DOIGoogle Scholar
- Mahony M. The decline of the green and golden bell frog Litoria aurea viewed in the context of declines and disappearances of other Australian frogs. Aust Zool. 1996;30:237–47.
- Richards SJ, McDonald KR, Alford RA. Declines in populations of Australia's endemic tropical forest frogs. Pac Conserv Biol. 1993;1:66–77.
- Williams SE, Hero J-M. Rainforest frogs of the Australian Wet Tropics: Guild classification and the ecological similarity of declining species. Proc R Soc Lond B Biol Sci. 1998;265:597–602. DOIGoogle Scholar
- Pounds JA, Fogden MPL, Savage JM, Gorman GC. Tests of null models for amphibian declines on a tropical mountain. Conserv Biol. 1997;11:1307–22. DOIGoogle Scholar
- Lips KR. Mass mortality and population declines of anurans at an upland site in Western Panama. Conserv Biol. 1999;13:117–25. DOIGoogle Scholar
- Cunningham AA, Langton TES, Bennett PM, Lewin JF, Drury SEN, Gough RE, Pathological and microbiological findings from incidents of unusual mortality of the common frog (Rana temporaria). Philos Trans R Soc Lond B Biol Sci. 1996;351:1529–57. DOIPubMedGoogle Scholar
- Jancovich JK, Davidson EW, Morado JF, Jacobs BL, Collins JP. Isolation of a lethal virus from the endangered tiger salamander Ambystoma tigrinum stebbinsi. Dis Aquat Organ. 1997;31:161–7. DOIGoogle Scholar
- Berger L, Speare R, Daszak P, Green DE, Cunningham AA, Goggin CL, Chytridiomycosis causes amphibian mortality associated with population declines in the rainforests of Australia and Central America. Proc Natl Acad Sci U S A. 1998;95:9031–6. DOIPubMedGoogle Scholar
- Berger L, Speare R, Hyatt AD. Chytrid fungi and amphibian declines: Overview, implications and future directions. In: Campbell A, editor. Declines and disapparances of Australian frogs. Environmental Australia, Canberra, Australia: Environmental Australia; 2000. p.21-31.
- Daszak P, Cunningham AA. Extinction by infection. Trends Ecol Evol. 1999;14:279. DOIPubMedGoogle Scholar
- Carey C, Cohen N, Rollins-Smith L. Amphibian declines: an immunological perspective. Dev Comp Immunol. 1999;23:459–72. DOIPubMedGoogle Scholar
- Drury SEN, Gough RE, Cunningham AA. Isolation of an iridovirus-like agent from common frogs (Rana temporaria). Vet Rec. 1995;137:72–3.PubMedGoogle Scholar
- Hyatt AD, Gould AR, Zupanovic Z, Cunningham AA, Hengstberger S, Whittington RJ, Characterisation of piscine and amphibian iridoviruses. Arch Virol. 2000. In press.PubMedGoogle Scholar
- Docherty D, Chinchar VG, Meteyer C, Brannian R, Hansen W, Wang J, A diagnostic evaluation of three salamander mortality events associated with an iridovirus and subsequent genomic comparison of the virus isolates obtained. In: Proceedings of the 48th Annual Wildlife Disease Association Conference; 1999 Aug 8-12; Athens, Georgia.
- Bollinger TK, Mao J, Schock D, Brigham RM, Chinchar VG. Pathology, isolation and preliminary molecular characterization of a novel iridovirus from tiger salamanders in Saskatchewan. J Wildl Dis. 1999;35:413–29.PubMedGoogle Scholar
- Pessier AP, Nichols DK, Longcore JE, Fuller MS. Cutaneous chytridiomycosis in poison dart frogs (Dendrobates spp.) and White's tree frogs (Litoria caerulea). J Vet Diagn Invest. 1999;11:194–9.PubMedGoogle Scholar
- Milius S. Fatal skin fungus found in U.S. frogs. Science News 1998;July 4th:7.
- Laurance WF, McDonald KR, Speare R. Epidemic disease and the catastrophic decline of Australian rain forest frogs. Conserv Biol. 1996;10:406–13. DOIGoogle Scholar
- Alford RA, Richards SJ. Lack of evidence for epidemic disease as an agent in the catastrophic decline of Australian rain forest frogs. Conserv Biol. 1997;11:1026–9. DOIGoogle Scholar
- Powell MJ. Looking at mycology with a Janus face: A glimpse at chytridiomycetes active in the environment. Mycologia. 1993;85:1–20. DOIGoogle Scholar
- Longcore JE, Pessier AP, Nichols DK. Batrachochytrium dendrobatidis gen. et sp. nov., a chytrid pathogenic to amphibians. Mycologia. 1999;91:219–27. DOIGoogle Scholar
- Garnett GP, Homes EC. The ecology of emergent infectious disease. Bioscience. 1996;46:127–35. DOIGoogle Scholar
- Laurance WF, McDonald KR, Speare R. In defense of the epidemic disease hypothesis. Conserv Biol. 1997;11:1030–4. DOIGoogle Scholar
- McCallum H, Dobson A. Detecting disease and parasite threats to endangered species and ecosystems. Trends Ecol Evol. 1995;10:190–4. DOIGoogle Scholar
- Dobson AP, Carper ER. Infectious diseases and human population history. Bioscience. 1996;46:115–26. DOIGoogle Scholar
- Burdon JJ. Fungal pathogens as selective forces in plant populations and communities. Aust J Ecol. 1991;16:423–32. DOIGoogle Scholar
- Vitousek PM, D'Antonio CM, Loope LL, Westbrooks R. Biological invasions as global environmental change. Am Sci. 1996;84:468–78.
- Scott NJ. Postmetamorphic death syndrome. Froglog. 1993;7:1–2.
- Taylor SK, Williams ES, Thorne ET, Mills KW, Withers DI, Pier AC. Causes of mortality of the Wyoming toad. J Wildl Dis. 1999;35:49–57.PubMedGoogle Scholar
- Groff JM, Mughannam A, McDowell TS, Wong A, Dykstra MJ, Frye FL, An epizootic of cutaneous zygomycosis in cultured dwarf African clawed frogs (Hymenochirus curtipes) due to Basidiobolus ranarum. J Med Vet Mycol. 1991;29:215–23. DOIPubMedGoogle Scholar
- Richard A, Cory J, Speight M, Williams T. Foraging in a pathogen reservoir can lead to local host population extinction: A case study of lepidoptera-virus interaction. Oecologia. 1999;118:29–38. DOIPubMedGoogle Scholar
- Cox RA. Coccidioidomycosis. In: Fungal infections and immune responses. Murphy JW, Friedman H, Bendinelli M, editors. New York: Plenum Press; 1993. p. 173-211.
- Godfray HCJ, Briggs CJ, Barlow ND, O'Callaghan M, Glare TR, Jackson TA. A model of insect-pathogen dynamics in which a pathogenic bacterium can also reproduce saprophytically. Proc R Soc Lond B Biol Sci. 1999;266:233–40. DOIGoogle Scholar
- Bennett PM, Owens IPF. Variation in extinction risk among birds: Chance or evolutionary predisposition? Proc R Soc Lond B Biol Sci. 1997;264:401–8. DOIGoogle Scholar
- Pounds JA, Fogden MPL, Campbell JH. Biological response to climate change on a tropical mountain. Nature. 1999;398:611–5. DOIGoogle Scholar
- Kiesecker JM, Blaustein AR. Synergism between UV-B radiation and a pathogen magnifies amphibian embryo mortality in nature. Proc Natl Acad Sci U S A. 1995;92:11049–52. DOIPubMedGoogle Scholar
- Madronich S, de Gruijl FR. Skin cancer and UV radiation. Nature. 1993;366:23. DOIPubMedGoogle Scholar
- Granoff A, Came PE, Rafferty KA. The isolation and properties of viruses from Rana pipiens: Their possible relationship to the renal adenocarcinoma of the leopard frog. Ann N Y Acad Sci. 1965;126:237–55. DOIPubMedGoogle Scholar
- Clark HF, Brennan JC, Zeigel RF. Karzon. Isolation and characterization of viruses from the kidneys of Rana pipiens with renal adenocarcinoma before and after passage in red eft (Triturus viridescens). J Virol. 1968;2:629–40.PubMedGoogle Scholar
- Wolf K, Bullock GL, Dunbar CE, Quimby MC. Tadpole edema virus: A viscerotrophic pathogen for anuran amphibians. J Infect Dis. 1968;118:253–62.PubMedGoogle Scholar
- Fijan N, Matasin Z, Petrinec Z, Valpotic I, Zwillenberg LO. Isolation of an iridovirus-like agent from the green frog Rana esculenta L. Veterinarski Archiv Zagreb. 1991;61:151–8.
- Hengstberger SG, Hyatt AD, Speare R, Coupar BEH. Comparison of epizootic haematopoietic necrosis and Bohle iridoviruses, recently isolated Australian iridoviruses. Dis Aquat Organ. 1993;15:93–107. DOIGoogle Scholar
- Zupanovic Z, Musso C, Lopez G, Louriero C-L, Hyatt AD, Hengstberger S, Isolation and characterization of iridoviruses from the giant toad Bufo marinus in Venezuela. Dis Aquat Organ. 1998;33:1–9. DOIPubMedGoogle Scholar
- Mao J, Green DE, Fellers G, Chinchar VG. Molecular characterization of iridoviruses isolated from sympatric amphibians and fish. Virus Res. 1999;63:45–52. DOIPubMedGoogle Scholar
- Mao J, Hedrick RP, Chinchar VG. Molecular characterization, sequence analysis and taxonomic position of newly isolated fish iridoviruses. Virology. 1997;229:212–20. DOIPubMedGoogle Scholar
- Zhang QY, Li ZQ, Gui JF. Studies on morphogenesis and cellular interactions of Rana grylio virus in an infected fish cell line. Aquaculture. 1999;175:185–97. DOIGoogle Scholar
- Westhouse RA, Jacobsen ER, Harris RK, Winter KR, Homer BL. Respiratory and pharyngo-esophageal iridovirus infection in a gopher tortoise (Gopherus polyphemus). J Wildl Dis. 1996;32:682–6.PubMedGoogle Scholar
- Chen Z-X, Zheng J-C, Jiang Y-I. A new iridovirus from soft-shelled turtle. Virus Res. 1999;63:147–51. DOIPubMedGoogle Scholar
- Murphy FA, Fauquet CM, Bishop DHL, Ghabrial SA, Jarvis AW, Martelli P, Iridoviridae. In: Murphy FA, Fauquet CM, Bishop DHL, Ghabrial SA, Jarvis AW, Martelli P, et al., editors. Virus taxonomy: The classification and nomenclature of viruses. The sixth report of the international committee on the taxonomy of viruses. New York, Wien: Springer-Verlag; 1995. p. 95-9.
- Hyatt AD, Parkes H, Zupanovic Z. Identification, characterisation and assessment of Venezuelan viruses for potential use as biological control agents against the cane toad (Bufo marinus) in Australia. Report to the Australian Federal Government and Environment Australia. 1998.
- Ahne W, Bremont M, Hedrick RP, Hyatt AD, Whittington RJ. Iridoviruses associated with epizootic haematopoietic necrosis (EHN) in aquaculture. World J Microbiol Biotechnol. 1997;13:367–73. DOIGoogle Scholar
- Hyatt AD, Whittington RJ. National diagnostic tests for the detection of epizootic haematopoietic necrosis virus (EHNV) and certification of EHNV-free fish. Final report (project 92/66). Fish Research Development Council of Australia; 1997.
- Langdon JS. Experimental transmission and pathogenicity of epizootic haematopoietic necrosis virus (EHNV) in redfin perch, Perca fluviatilis L., and 11 other teleosts. J Fish Dis. 1989;12:295–310. DOIGoogle Scholar
- Whittington RJ, Kearns C, Hyatt AD, Hengstberger S, Rutzou T. Spread of epizootic haematopoietic necrosis virus (EHNV) in redfin perch (Perca fluviatilis) in Southern Australia. Aust Vet J. 1996;73:112–4. DOIPubMedGoogle Scholar
- Whittington RJ, Hyatt AD. Diagnosis and prevention of epizootic haematopoietic necrosis virus infection. Proceedings of the National Research Institute of Aquaculture International Workshop: New approaches to viral diseases of aquatic animals; 1997 Jan 21-24; Kyoto, Japan. National Research Institute of Aquaculture; 1997.
- Zupanovic Z, Lopez B, Hyatt AD, Green B, Bartran G, Parkes H, Giant toads Bufo marinus in Australia and Venezuela have antibodies against "ranaviruses". Dis Aquat Organ. 1998;32:1–8. DOIPubMedGoogle Scholar
- Speare R, Smith JR. An iridovirus-like agent isolated from the ornate burrowing frog Limnodynastes ornatus in northern Australia. Dis Aquat Organ. 1992;14:51–7. DOIGoogle Scholar
- Cullen BR, Owens L, Whittington RJ. Experimental infection of Australian anurans (Limnodynastes terraereginae and Litoria latopalmata) with Bohle iridovirus. Dis Aquat Organ. 1995;23:83–92. DOIGoogle Scholar
- Moody NJC, Owens L. Experimental demonstration of the pathogenicity of a frog virus, Bohle iridovirus, for a fish species, barramundi Lates calcarifer. Dis Aquat Organ. 1994;18:95–102. DOIGoogle Scholar
- Lampo M, De Leo GA. The invasion ecology of the toad Bufo marinus: From South America to Australia. Ecol Appl. 1998;8:388–96.
- Nichols DK, Pessier AP, Longcore JE. Cutaneous chytridiomycosis: an emerging disease? Proceedings American Association of Zoo Veterinarians 1998:269-71.
- Lessios HA. Mass mortality of Diadema antillarum in the Caribbean: What have we learned? Annu Rev Ecol Syst. 1988;19:371–93.
- Burton TM, Likens GE. Salamander populations and biomass in the Hubbard Brook Experimental Forest, New Hampshire. Copeia. 1975;3:541–6. DOIGoogle Scholar