Volume 10, Number 2—February 2004
THEME ISSUE
2004 SARS Edition
Laboratory Study
Interferon-β 1a and SARS Coronavirus Replication
Cite This Article
Citation for Media
Abstract
A global outbreak of severe acute respiratory syndrome (SARS) caused by a novel coronavirus began in March 2003. The rapid emergence of SARS and the substantial illness and death it caused have made it a critical public health issue. Because no effective treatments are available, an intensive effort is under way to identify and test promising antiviral drugs. Here, we report that recombinant human interferon (IFN)-β 1a potently inhibits SARS coronavirus replication in vitro.
The recent global outbreak of severe acute respiratory syndrome (SARS) has quickly gained notoriety as a newly emerging infectious disease. The etiologic agent was identified as a coronavirus (SARS-CoV) that is not closely related to any of the previously characterized coronaviruses (1,2). As of September 26, 2003, a total of 8,098 probable cases of SARS have occurred with 774 deaths.
No antiviral treatments are currently available against SARS-CoV. SARS cases have been treated symptomatically according to the severity of the illness. A treatment protocol consisting of antibacterial agents and a combination of ribavirin and methylprednisolone was recently proposed. However, the therapeutic value of ribavirin remains uncertain because it has no activity against SARS-CoV in vitro. Molecular modeling studies suggest that rhinovirus 3Cpro inhibitors may be useful for SARS therapy, but results of recent in vitro testing of the lead molecule, AG7088, were negative (3).
Previous studies showed that some coronaviruses, including avian infectious bronchitis virus, murine hepatitis virus, and human coronavirus 229E, are susceptible to type I interferons in vitro or in vivo (4–7). Therefore, we evaluated the in vitro efficacy of a recombinant human type I interferon (IFN), IFN-β 1a (Serono International, Geneva, Switzerland) against three different isolates of SARS-CoV (Tor2 and Tor7 and Urbani) using yield reduction assays. The IFN-β 1a preparation employed in this study was selected because it is currently used as part of the most effective treatment regimen for relapsing forms of multiple sclerosis (8), and more importantly, because it was shown to have antiviral activity (as measured in a vesicular stomatitis virus cytopathic assay system) 14 times greater than the currently available treatment using IFN-β 1b (9).
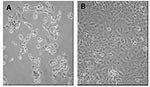
In the current study, Vero E-6 cells were treated with concentrations (5,000 to 500,000 IU/mL) of IFN-β 1a either 24 h before or 1 h after inoculation with the SARS-CoV (m.o.i. 0.1 PFU/cell), and monitored for cytopathic effect and production of infectious SARS-CoV at 24, 48, and 72 h postinfection. Inhibition of the SARS-CoVs by IFN-β 1a was dependent on both time of drug administration and time of culture sampling after SARS-CoV infection. Production of infectious SARS-CoV was potently inhibited (≥99.5% or 2.00 log10 PFU/mL) at 24 h postinfection. by pretreatment of Vero E-6 cells with IFN-β 1a at all concentrations tested (Figure 1). By 72 h postinfection, inhibition of SARS-CoV production by IFN-β 1a had declined for all three SARS-CoVs, with inhibition (>70%) being detected in the Tor7 (Figure 1) and Urbani isolates (data not shown). IFN-β 1a was somewhat less effective at inhibiting SARS-CoV replication when employed after infection of cultures (Figure 1). Nonetheless, production of infectious SARS-CoVs was considerably reduced (≥90% or 1.00 log10 PFU/mL) at 24 and 48 h postinfection. Protection of Vero E-6 monolayers against SARS-CoV–induced cytopathic effects by preinfection or postinfection treatment with IFN-β 1a was dramatic, even at 72 h postinfection (Figure 2). Additional concentrations of IFN-β 1a (0.5–5,000 IU/mL) were tested to determine the 50% inhibitory concentration (IC50). Pretreatment of Vero E-6 cells with concentrations as low as 50 IU/mL, or posttreatment of cells with concentrations at 500 IU/mL, provided a 50% reduction with the Tor2 isolate at 24 h postinfection.
Faced with a burgeoning epidemic of SARS cases and a lack of effective treatment options, identifying compounds with antiviral activity that could be potential therapeutics has become a high priority. Our report suggests that IFN-β 1a may be effective as a treatment for SARS-CoV infections. As noted above, IFN-β 1a is currently being used for a variety of clinical indications, including multiple sclerosis, and has shown dose-dependent efficacy in several clinical trials. Importantly, IFN-β 1a exhibited potent antiviral activity at doses that have already been shown to have acceptable safety profiles in animals (10). Thus, we report the identification of a compound that may be suitable for rapid development as a treatment for SARS-CoV infection.
Dr. Hensley is a staff microbiologist in the Virology Division, United States Army Medical Research Institute of Infections Diseases. She specializes in the pathogenesis and immunobiology of high-hazard virus infections in animal models for vaccine and antiviral drug development.
Acknowledgments
We thank Dr. Heinz Feldman for his kind provision of Tor2 and Tor7 isolates.
This study was supported, in part, by NIH grant R21 AI053539.
References
- Peiris JSM, Lai ST, Poon LLM, Guan Y, Yam LY, Lim W, Coronavirus as a possible cause of severe acute respiratory syndrome. Lancet. 2003;361:1319–25. DOIPubMedGoogle Scholar
- Marra MA, Jones SJM, Astell CR, Holt RA, Brooks-Wilson A, Butterfield YS, The genome sequence of the SARS-associated coronavirus. Science. 2003;300:1399–404. DOIPubMedGoogle Scholar
- Anand K, Ziebuhr J, Wadhwani P, Mesters JR, Hilgenfeld R. Coronavirus main proteinase (3CLpro) structure: basis for design of anti-SARS drugs. Science. 2003;300:1763–7. DOIPubMedGoogle Scholar
- Sperber SJ, Hayden FG. Comparative susceptibility of respiratory viruses to recombinant interferons-alpha 2b and -beta. J Interferon Res. 1989;9:285–93.PubMedGoogle Scholar
- Pei J, Sekellick MJ, Marcus PI, Choi IS, Collisson EW. Chicken interferon type I inhibits infectious bronchitis virus replication and associated respiratory illness. J Interferon Cytokine Res. 2001;21:1071–7. DOIPubMedGoogle Scholar
- Vassao RC, de Franco MT, Hartz D, Modolell M, Sippel AE, Pereira CA. Down-regulation of Bgp1(a) viral receptor by interferon-gamma is related to the antiviral state and resistance to mouse hepatitis virus 3 infection. Virology. 2000;274:278–83. DOIPubMedGoogle Scholar
- Minagawa H, Takenaka A, Mohri S, Mori R. Protective effect of recombinant murine interferon beta against mouse hepatitis virus infection. Antiviral Res. 1987;8:85–95. DOIPubMedGoogle Scholar
- Panitch H, Goodin DS, Francis G, Chang P, Coyle PK, O’Connor P, Randomized, comparative study of interferon beta-1a treatment regimens in MS: The EVIDENCE trial. Neurology. 2002;59:1496–506.PubMedGoogle Scholar
- Antonetti F, Finocchiaro O, Mascia M, Terlizzese MG, Jaber A. A comparison of the biologic activity of two recombinant IFN-beta preparations used in the treatment of relapsing-remitting multiple sclerosis. J Interferon Cytokine Res. 2002;22:1181–4. DOIPubMedGoogle Scholar
- Mager DE, Neuteboom B, Efthymiopoulos C, Munafo A, Jusko WJ. Receptor-mediated pharmacokinetics and pharmacodynamics of interferon-{beta}1a following intravenous and subcutaneous dosing in monkeys. J Pharmacol Exp Ther. 2003;306:262–70. DOIPubMedGoogle Scholar
Figures
Cite This ArticleTable of Contents – Volume 10, Number 2—February 2004
| EID Search Options |
|---|
|
|
|
|
|
|

Please use the form below to submit correspondence to the authors or contact them at the following address:
Peter B. Jahrling, USAMRIID, Attn: MCMR-UIZ, 1425 Porter Street, Fort Detrick, MD 21702-5011, USA; fax: 301-619-4625
Top