Volume 21, Number 12—December 2015
CME ACTIVITY - Dispatch
Life-Threatening Sochi Virus Infections, Russia
Figure 2
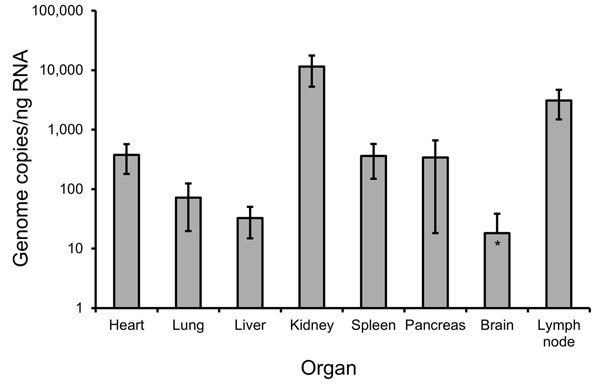
Figure 2. Quantification of hantavirus RNA in tissue biopsies from a 50-year-old Sochi virus–infected man (patient no. 59), Russia. Two independent approaches were performed to extract RNA from each organ. Quantitative reverse transcription PCR previously developed for DOBV (7) was used to measure virus load in the analyzed biopsy samples. Three quantitative reverse transcription PCR estimations were conducted for every RNA extraction, followed by calculation of mean values and SDs. Viral RNA levels are shown as genome copies per nanogram of total RNA isolated from the samples. Error bars indicate SD.
References
- Kruger DH, Figueiredo LTM, Song JW, Klempa B. Hantaviruses—globally emerging pathogens. J Clin Virol. 2015;64:128–36. DOIPubMedGoogle Scholar
- Tkachenko EA, Okulova NM, Yunicheva YV, Morzunov SP, Khaĭbulina SF, Riabova TE, The epizootological and virological characteristics of a natural hantavirus infection focus in the subtropic zone of the Krasnodarsk Territory [in Russian]. Vopr Virusol. 2005;50:14–9 .PubMedGoogle Scholar
- Klempa B, Tkachenko EA, Dzagurova TK, Yunicheva YV, Morozov VG, Okulova NM, Hemorrhagic fever with renal syndrome caused by 2 lineages of Dobrava hantavirus, Russia. Emerg Infect Dis. 2008;14:617–25. DOIPubMedGoogle Scholar
- Dzagurova TK, Witkowski PT, Tkachenko EA, Klempa B, Morozov VG, Auste B, Isolation of Sochi virus from a fatal case of hantavirus disease with fulminant clinical course. Clin Infect Dis. 2012;54:e1–4. DOIPubMedGoogle Scholar
- Klempa B, Avsic-Zupanc T, Clement J, Dzagurova TK, Henttonen H, Heyman P, Complex evolution and epidemiology of Dobrava-Belgrade hantavirus: definition of genotypes and their characteristics. Arch Virol. 2013;158:521–9. DOIPubMedGoogle Scholar
- Tamura K, Stecher G, Peterson D, Filipski A, Kumar S. MEGA6: molecular evolutionary genetics analysis version 6.0. Mol Biol Evol. 2013;30:2725–9. DOIPubMedGoogle Scholar
- Kramski M, Meisel H, Klempa B, Krüger DH, Pauli G, Nitsche A. Detection and typing of human pathogenic hantaviruses by real-time reverse transcription–PCR and pyrosequencing. Clin Chem. 2007;53:1899–905. DOIPubMedGoogle Scholar
- Okulova NM, Khliap LA, Varshavskii AA, Dzagurova TK, Iunicheva IV, Riabova TE, Spatial structure of natural foci of hantavirus on the territory of northwestern Caucasus [in Russian]. Zh Mikrobiol Epidemiol Immunobiol. 2013; (
Sep–Oct ):47–53 .PubMedGoogle Scholar - Mertz GJ, Hjelle B, Crowley M, Iwamoto G, Tomicic V, Vial PA. Diagnosis and treatment of new world hantavirus infections. Curr Opin Infect Dis. 2006;19:437–42. DOIPubMedGoogle Scholar
- Zhang YZ, Zou Y, Fu ZF, Plyusnin A. Hantavirus infections in humans and animals, China. Emerg Infect Dis. 2010;16:1195–203. DOIPubMedGoogle Scholar
- Noh JY, Cheong HJ, Song JY, Kim WJ, Song KJ, Klein TA, Clinical and molecular epidemiological features of hemorrhagic fever with renal syndrome in Korea over a 10-year period. J Clin Virol. 2013;58:11–7. DOIPubMedGoogle Scholar
- Avsic-Zupanc T, Petrovec M, Furlan P, Kaps R, Elgh F, Lundkvist A. Hemorrhagic fever with renal syndrome in the Dolenjska region of Slovenia—a 10-year survey. Clin Infect Dis. 1999;28:860–5. DOIPubMedGoogle Scholar
- Papa A, Antoniadis A. Hantavirus infections in Greece—an update. Eur J Epidemiol. 2001;17:189–94 . DOIPubMedGoogle Scholar
- Dzagurova TK, Klempa B, Tkachenko EA, Slyusareva GP, Morozov VG, Auste B, Molecular diagnostics of hemorrhagic fever with renal syndrome during a Dobrava virus infection outbreak in the European part of Russia. J Clin Microbiol. 2009;47:4029–36 . DOIPubMedGoogle Scholar
Page created: November 13, 2015
Page updated: November 13, 2015
Page reviewed: November 13, 2015
The conclusions, findings, and opinions expressed by authors contributing to this journal do not necessarily reflect the official position of the U.S. Department of Health and Human Services, the Public Health Service, the Centers for Disease Control and Prevention, or the authors' affiliated institutions. Use of trade names is for identification only and does not imply endorsement by any of the groups named above.