Volume 21, Number 12—December 2015
CME ACTIVITY - Dispatch
Life-Threatening Sochi Virus Infections, Russia
Cite This Article
Citation for Media
Introduction

Medscape, LLC is pleased to provide online continuing medical education (CME) for this journal article, allowing clinicians the opportunity to earn CME credit.
This activity has been planned and implemented in accordance with the Essential Areas and policies of the Accreditation Council for Continuing Medical Education through the joint providership of Medscape, LLC and Emerging Infectious Diseases. Medscape, LLC is accredited by the ACCME to provide continuing medical education for physicians.
Medscape, LLC designates this Journal-based CME activity for a maximum of 1.0 AMA PRA Category 1 Credit(s)TM. Physicians should claim only the credit commensurate with the extent of their participation in the activity.
All other clinicians completing this activity will be issued a certificate of participation. To participate in this journal CME activity: (1) review the learning objectives and author disclosures; (2) study the education content; (3) take the post-test with a 75% minimum passing score and complete the evaluation at http://www.medscape.org/journal/eid; (4) view/print certificate.
Release date: November 13, 2015; Expiration date: November 13, 2016
Learning Objectives
Upon completion of this activity, participants will be able to:
• Analyze the demographics of patients infected with the Sochi virus in the current study
• Assess laboratory data available from patients infected with Sochi virus in the current study
• Distinguish the anatomic site of the highest concentration of Sochi virus among infected individuals
• Evaluate the prognosis of infection with Sochi virus.
CME Editor
Karen L. Foster, Technical Writer/Editor, Emerging Infectious Diseases. Disclosure: Karen L. Foster has disclosed no relevant financial relationships.
CME Author
Charles P. Vega, MD, Clinical Professor of Family Medicine, University of California, Irvine. Disclosure: Charles P. Vega, MD, has disclosed the following financial relationships: served as an advisor or consultant for Lundbeck, Inc.; McNeil Pharmaceuticals; Takeda Pharmaceuticals North America, Inc.
Authors
Disclosures: Detlev H. Kruger, MD, PhD; Evgeniy A. Tkachenko, MD, PhD; Vyacheslav G. Morozov, MD; Yulia V. Yunicheva, MD, PhD; Olga M. Pilikova, MD, PhD; Gennadiy Malkin, BS; Aydar A. Ishmukhametov, MD, PhD; Patrick Heinemann, PhD; Peter T. Witkowski, MD; Boris Klempa, PhD; and Tamara K. Dzagurova, MD, PhD, have disclosed no relevant financial relationships.
Abstract
Sochi virus was recently identified as a new hantavirus genotype carried by the Black Sea field mouse, Apodemus ponticus. We evaluated 62 patients in Russia with Sochi virus infection. Most clinical cases were severe, and the case-fatality rate was as high as 14.5%.
Hantaviruses are zoonotic pathogens transmitted from small animals to humans. Hantavirus disease in the Americas is called hantavirus pulmonary syndrome and in Asia and Europe is called hemorrhagic fever with renal syndrome (HFRS). Both syndromes can lead to cardiopulmonary and renal failure (1). Recently we described a new hantavirus, Sochi virus, from the administrative region Krasnodar (including the city of Sochi), southern European Russia, which was isolated in cell culture from a Black Sea field mouse (Apodemus ponticus) and a patient with fulminant hantavirus disease who died of shock and combined kidney and lung failure (2–4). Molecular taxonomical analyses identified Sochi virus as a new genotype within the Dobrava-Belgrade virus (DOBV) species (5). Here we show that HFRS caused by Sochi virus infection occurs in the geographic region where A. ponticus mice are prevalent. For 62 patients infected by this virus during 2000–2013, we evaluated clinical and epidemiologic data.
Serum of patients with suspected acute hantavirus disease from the Krasnodar region were screened for hantavirus antibodies by indirect immunofluorescence assays and ELISA. Sixty-two patients showed clear DOBV IgG seropositivity. During the acute phase of illness, all patients tested positive for DOBV IgM (data not shown). For 26 patients, sufficient volumes of follow-up serum were available for additional focus reduction neutralization assays to specify neutralizing antibodies. All serum samples exhibited substantially higher neutralizing titers toward DOBV than toward Puumala virus, Hantaan virus, and Seoul virus. When the neutralizing effect of DOBV-positive patients’ serum were compared against the different human pathogenic genotypes of DOBV (Dobrava, Kurkino, and Sochi), all serum predominantly reacted with the Sochi genotype (Technical Appendix Table 1).
We successfully obtained virus genomic large (L) segment sequences from 2 patients (no. 51, specimen no. 6882; no. 59, specimen no. 10752). In the neighborhood of the residence of patient no. 59, mice were trapped, and hantaviral L and small (S) segment regions from 2 A. ponticus animals (specimen nos. 10636, 10645) were amplified. The sequences obtained were deposited in GenBank under accession nos. KM192207–09 and KP878308–10 (L segment) and KP878311–13 (S segment) (Technical Appendix Table 2). Samples from virus-positive mice were phylogenetically characterized by analysis of a 242-bp region of their cytB gene; all of them clustered with those of the previously identified A. ponticus animals (3) (data not shown). In addition, the A. ponticus–derived isolate Sochi/Ap (4), the patient-derived isolate Sochi/hu (5), an S segment sequence from a mouse (GK/Ap) trapped near the home of the previously described Krasnodar patient (4), and sequences originating from 2 A. ponticus mice sampled near the Black Sea coast, 43/Ap and 79/Ap, were included in the molecular analyses of the virus.
The patient-derived sequences 6882/hu, 10752/hu, and Sochi/hu clearly cluster with A. ponticus–derived sequences 43/Ap, 79/Ap, 10636/Ap, 10645/Ap, and Sochi/Ap (Figure 1, panel A). In the analysis of the S segment, we obtained a very similar result; the patient-derived sequences 10752/hu, Krasnodar/hu, and Sochi/hu cluster with A. ponticus–associated sequences 43/Ap, 79/Ap, 10636/Ap, 10645/Ap, GK/Ap, and Sochi/Ap (Figure 1, panel B). In analysis of both L and S segments, the Sochi virus strains form a unique group, clearly distinguishable from all other DOBV genotypes.
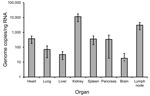
Specimens from different organs of deceased patient no. 59 were analyzed for virus load. The highest concentration was detected in kidney (11,446 copies/ng RNA) and lymph node (3,086 copies/ng RNA), whereas the least virus RNA (10–100 copies/ng RNA) was detected in lung, brain, and liver (Figure 2).
The clinical disease severity of the 62 Sochi virus–infected patients investigated (Table 1) was subdivided into mild, moderate, or severe following the standard Russian criteria (i.e., length of febrile phase, minimal blood pressure in the hypotonic phase, extent of hemorrhagic symptoms, minimal urine production, serum creatinine level, and extent of proteinuria) (Technical Appendix Table 3). The case-fatality rate (CFR) was as high as 14.5% (9/62 patients). Including fatalities, severe disease developed in nearly 60% of patients, whereas the remaining 40% of cases were moderate. The average age of all patients was 33 years. A significantly higher proportion of patients were males (p = 1.05 × 10–9). Moreover, severe disease developed in most affected male patients (66.7%) but in only 35.7% of affected female patients (p = 0.037). The fact that only 2 of 9 fatal cases occurred in female patients (Table 1) underscores this finding.
All 9 patients with fatal infections died of multiorgan failure and shock (Table 2). Postmortem examination showed multiple hemorrhages and edema in internal organs, including kidneys and lungs. The patients died within 8.2 days (range 3–16 days) after disease onset. An extraordinary fulminant course was observed for patient no. 47, who died 3 days after onset and before he could be hospitalized. This 19-year-old man was the son-in-law of patient no. 48, who also died after Sochi virus infection. Both men lived at the same rural address, and rodent contact during work in haystacks was reported.
We have demonstrated the occurrence of human infections by Sochi virus and studied the clinical outcome for 62 patients. This virus is carried by the Black Sea field mouse (A. ponticus), which occurs naturally in the Transcaucasian region between the Black and Caspian Seas, including a part of southern European Russia. In anecdotal field studies in the coast region near Sochi, A. ponticus was the most abundant mouse species (71% of all trapped mice were identified as A. ponticus); moreover, 14% of trapped A. ponticus mice were serologically proven to be DOBV infected (8). This finding indicates that DOBV is the hantavirus indigenous in this geographic area and that A. ponticus mice are highly relevant as a hantavirus reservoir. All evidence from the natural virus reservoir, as well as serologic and molecular diagnostics of patients’ serum, shows that the virus responsible for the infections is the DOBV genotype Sochi.
Most investigated patients found to be infected by Sochi virus exhibited a severe clinical course. With a calculated CFR of 14.5%, Sochi virus might be the most deadly hantavirus outside the Americas, where 35%–50% of hantavirus infections are fatal (1,9). Even Asian Hantaan virus is estimated to be less deadly; recent studies show CFRs of 1%–3% in China and South Korea, where Hantaan virus infections play an important role in HFRS morbidity (10,11). On the other hand, increased awareness in diagnostics, treatment, and prevention by local physicians and public health authorities is expected to improve survival rates for Sochi virus infections.
Among the related viruses of the DOBV species, Sochi virus seems to have the highest level of virulence, similar to Dobrava virus (carried by A. flavicollis mice), which has a CFR of up to 10%–12% (12,13). As shown in larger studies, disease caused by infection with the related Kurkino genotype (carried by the western lineage of A. agrarius mice) is associated with a CFR of only 0.3%–0.9% (3,14). These phylogenetically related viruses exert a quite different pathogenicity in humans.
Dr. Kruger is the head of the Institute of Medical Virology, Charité–University Medicine Berlin. His research focuses on the molecular epidemiology and clinical relevance of emerging virus infections.
Acknowledgments
We thank Brita Auste for careful execution of the molecular diagnostics.
This work was supported by Russian Science Foundation (grant no. 14-15-00619), Robert Koch Institute on behalf of the German Ministry of Public Health (grant no. 1369-382), and Deutsche Forschungsgemeinschaft (Priority Program 1596 “Ecology and species barriers in emerging viral diseases,” grant no. KR1293/13-1).
References
- Kruger DH, Figueiredo LTM, Song JW, Klempa B. Hantaviruses—globally emerging pathogens. J Clin Virol. 2015;64:128–36. DOIPubMedGoogle Scholar
- Tkachenko EA, Okulova NM, Yunicheva YV, Morzunov SP, Khaĭbulina SF, Riabova TE, The epizootological and virological characteristics of a natural hantavirus infection focus in the subtropic zone of the Krasnodarsk Territory [in Russian]. Vopr Virusol. 2005;50:14–9 .PubMedGoogle Scholar
- Klempa B, Tkachenko EA, Dzagurova TK, Yunicheva YV, Morozov VG, Okulova NM, Hemorrhagic fever with renal syndrome caused by 2 lineages of Dobrava hantavirus, Russia. Emerg Infect Dis. 2008;14:617–25. DOIPubMedGoogle Scholar
- Dzagurova TK, Witkowski PT, Tkachenko EA, Klempa B, Morozov VG, Auste B, Isolation of Sochi virus from a fatal case of hantavirus disease with fulminant clinical course. Clin Infect Dis. 2012;54:e1–4. DOIPubMedGoogle Scholar
- Klempa B, Avsic-Zupanc T, Clement J, Dzagurova TK, Henttonen H, Heyman P, Complex evolution and epidemiology of Dobrava-Belgrade hantavirus: definition of genotypes and their characteristics. Arch Virol. 2013;158:521–9. DOIPubMedGoogle Scholar
- Tamura K, Stecher G, Peterson D, Filipski A, Kumar S. MEGA6: molecular evolutionary genetics analysis version 6.0. Mol Biol Evol. 2013;30:2725–9. DOIPubMedGoogle Scholar
- Kramski M, Meisel H, Klempa B, Krüger DH, Pauli G, Nitsche A. Detection and typing of human pathogenic hantaviruses by real-time reverse transcription–PCR and pyrosequencing. Clin Chem. 2007;53:1899–905. DOIPubMedGoogle Scholar
- Okulova NM, Khliap LA, Varshavskii AA, Dzagurova TK, Iunicheva IV, Riabova TE, Spatial structure of natural foci of hantavirus on the territory of northwestern Caucasus [in Russian]. Zh Mikrobiol Epidemiol Immunobiol. 2013; (
Sep–Oct ):47–53 .PubMedGoogle Scholar - Mertz GJ, Hjelle B, Crowley M, Iwamoto G, Tomicic V, Vial PA. Diagnosis and treatment of new world hantavirus infections. Curr Opin Infect Dis. 2006;19:437–42. DOIPubMedGoogle Scholar
- Zhang YZ, Zou Y, Fu ZF, Plyusnin A. Hantavirus infections in humans and animals, China. Emerg Infect Dis. 2010;16:1195–203. DOIPubMedGoogle Scholar
- Noh JY, Cheong HJ, Song JY, Kim WJ, Song KJ, Klein TA, Clinical and molecular epidemiological features of hemorrhagic fever with renal syndrome in Korea over a 10-year period. J Clin Virol. 2013;58:11–7. DOIPubMedGoogle Scholar
- Avsic-Zupanc T, Petrovec M, Furlan P, Kaps R, Elgh F, Lundkvist A. Hemorrhagic fever with renal syndrome in the Dolenjska region of Slovenia—a 10-year survey. Clin Infect Dis. 1999;28:860–5. DOIPubMedGoogle Scholar
- Papa A, Antoniadis A. Hantavirus infections in Greece—an update. Eur J Epidemiol. 2001;17:189–94 . DOIPubMedGoogle Scholar
- Dzagurova TK, Klempa B, Tkachenko EA, Slyusareva GP, Morozov VG, Auste B, Molecular diagnostics of hemorrhagic fever with renal syndrome during a Dobrava virus infection outbreak in the European part of Russia. J Clin Microbiol. 2009;47:4029–36 . DOIPubMedGoogle Scholar
Figures
Tables
Follow Up
Earning CME Credit
To obtain credit, you should first read the journal article. After reading the article, you should be able to answer the following, related, multiple-choice questions. To complete the questions (with a minimum 75% passing score) and earn continuing medical education (CME) credit, please go to http://www.medscape.org/journal/eid. Credit cannot be obtained for tests completed on paper, although you may use the worksheet below to keep a record of your answers. You must be a registered user on Medscape.org. If you are not registered on Medscape.org, please click on the “Register” link on the right hand side of the website to register. Only one answer is correct for each question. Once you successfully answer all post-test questions you will be able to view and/or print your certificate. For questions regarding the content of this activity, contact the accredited provider, CME@medscape.net. For technical assistance, contact CME@webmd.net. American Medical Association’s Physician’s Recognition Award (AMA PRA) credits are accepted in the US as evidence of participation in CME activities. For further information on this award, please refer to http://www.ama-assn.org/ama/pub/about-ama/awards/ama-physicians-recognition-award.page. The AMA has determined that physicians not licensed in the US who participate in this CME activity are eligible for AMA PRA Category 1 Credits™. Through agreements that the AMA has made with agencies in some countries, AMA PRA credit may be acceptable as evidence of participation in CME activities. If you are not licensed in the US, please complete the questions online, print the certificate and present it to your national medical association for review.
Article Title: Life-Threatening Sochi Virus Infections, Russia
CME Questions
ou are seeing a 33-year-old man in the emergency department for a 2-day history of fever, malaise, and headache. His temperature is 39.4°C, and his pulse is 120 bpm. He is admitted, and a thorough workup is initiated, along with intravenous antibiotics and supportive care. Infection with hantavirus is in the differential diagnosis. Which one of the following statements regarding the demographics of patients infected with the Sochi virus in the current study is most accurate?
A. Patients were generally younger women
B. Patients were generally older men
C. Patients were generally older women
D. Patients were generally younger men
2. Which one of the following statements regarding laboratory testing of patients with Sochi virus in the current study is most accurate?
A. Only half of the cases demonstrated clear DOBV immunoglobulin G (IgG) positivity
B. 100% of patients developed anti-DOBV IgM in the acute phase
C. Patients' serologic reaction was stronger against other types of hantavirus compared with the Sochi virus
D. The genetic signature of the Sochi virus could not be distinguished from that of other hantaviruses
3. The patient is diagnosed with infection with the Sochi virus. Which one of the following anatomic sites had the highest concentration of Sochi virus among patients in the current study?
A. Lung
B. Kidney
C. Liver
D. Brain
4. Which one of the statements regarding the prognosis of patients infected with Sochi virus in the current study is most accurate?
A. The case-fatality rate was 1.5%
B. The case-fatality rate was 14.5%
C. Approximately half of the patients developed mild disease only
D. Disease was more severe among women vs men
Activity Evaluation
|
1. The activity supported the learning objectives. |
||||
|
Strongly Disagree |
|
|
|
Strongly Agree |
|
1 |
2 |
3 |
4 |
5 |
|
2. The material was organized clearly for learning to occur. |
||||
|
Strongly Disagree |
|
|
|
Strongly Agree |
|
1 |
2 |
3 |
4 |
5 |
|
3. The content learned from this activity will impact my practice. |
||||
|
Strongly Disagree |
|
|
|
Strongly Agree |
|
1 |
2 |
3 |
4 |
5 |
|
4. The activity was presented objectively and free of commercial bias. |
||||
|
Strongly Disagree |
|
|
|
Strongly Agree |
|
1 |
2 |
3 |
4 |
5 |
Related Links
Table of Contents – Volume 21, Number 12—December 2015
| EID Search Options |
|---|
|
|
|
|
|
|

Please use the form below to submit correspondence to the authors or contact them at the following address:
Detlev H. Kruger, Institute of Medical Virology, Helmut-Ruska-Haus, Charité–University Medicine Berlin, Charitéplatz 1, D-10117 Berlin, Germany
Top