Volume 23, Number 10—October 2017
Dispatch
Macrolide-Resistant Mycoplasma pneumoniae Infection, Japan, 2008–2015
Cite This Article
Citation for Media
Abstract
We evaluated isolates obtained from children with Mycoplasma pneumoniae infection throughout Japan during 2008–2015. The highest prevalence of macrolide-resistant M. pneumoniae was 81.6% in 2012, followed by 59.3% in 2014 and 43.6% in 2015. The prevalence of macrolide-resistant M. pneumoniae among children in Japan has decreased.
Since the early 2000s, macrolide-resistant Mycoplasma pneumoniae isolates have been identified in Japan. We previously reported the results of our national surveillance study that investigated the prevalence of macrolide-resistant M. pneumoniae infection among children in Japan during 2008–2012 (1). An M. pneumoniae pandemic occurred in Japan during 2010–2012, especially among children. Similar pandemics also occurred in other countries (2). However, the prevalence of macrolide-resistant M. pneumoniae infection also gradually increased at the same time in Asia, including Japan (1,2). We investigated the prevalence of macrolide-resistant M. pneumoniae infection after the pandemic.
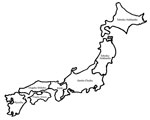
Children with respiratory tract infections who visited 68 medical institutions in Japan were classified according to district: 1) Kyushu (population 14 million); 2) Chugoku-Shikoku (11 million); 3) Kinki (20 million); 4) Kanto-Chubu (62 million); and 5) Tohoku-Hokkaido (14 million) (Figure). Most patients had been enrolled in the Atypical Pathogen Study Group before the 2010–2012 pandemic; however, some did not participate in our previous study reported in 2013 (1). Here, we report the data for January 2008–December 2015.
As previously reported (1), pediatricians collected nasopharyngeal swab samples and sputum samples, when available, from children with respiratory tract infections. Participants’ parents provided informed consent before sample collection. The Ethics Committee of the Kawasaki Medical School (Kurashiki, Japan) approved the study protocol on December 8, 2014 (no. 286–3).
Nasopharyngeal swab specimens were collected with a sterile swab (Nippon Menbo, Saitama, Japan). After collection, each swab was placed into 3 mL of Universal Vial Transport Medium (Becton, Dickinson and Company, Sparks, MD, USA) and transported at room temperature within 2 days to Kawasaki Medical School by a parcel delivery system. Crude DNA extracts were obtained with the following procedure: 300 μL of resuspended transport medium was transferred into a 1.5-mL microtube centrifuged at 4°C, 20,000 × g for 30 min, after which 285 μL of supernatant was discarded; the remainder was transferred into a thin-wall 200-μL PCR tube after resuspension with 85 μL lysis buffer by gentle pipetting. This suspension was incubated at 55°C for 60 min, followed by 100°C for 10 min before cooling to 4°C. The composition of the lysis buffer was Tris-HCl (pH 8.3) 2 mmol/L, KCl 10 mM, MgCl2 0.045 mM, Triton X-100 0.45 o/o, Tween 20 0.45%, and RNA-grade Proteinase K (Thermo Fisher Scientific Inc., Waltham, MA, USA) 0.4 µg/µL. M. pneumoniae DNA was detected by real-time PCR targeting a conserved part of the gene coding for the P1 adhesin (3).
We searched for mutations at sites 2063, 2064, and 2617 in domain V of 23S rRNA of M. pneumoniae using a direct sequencing method with isolates or samples with a positive PCR result, as reported previously (3). For this study, we investigated 1,448 samples obtained from patients in Japan who had respiratory tract infections; we detected M. pneumoniae DNA by real-time PCR and searched for mutations using a direct sequencing method.
The overall prevalence rate of macrolide-resistant M. pneumoniae in Japan was 70.2% (1,016/1,448) and ranged from 43.7% in Kyushu to 89.3% in Kanto-Chubu (Table 1). When divided into 3 time periods (prepandemic, pandemic, and postpandemic), the overall rate of macrolide-resistant M. pneumonia was substantially lower in the post epidemic period than in the pandemic period; macrolide-resistant M. pneumoniae isolates decreased in 4 of the 5 locations.
The peak rate of macrolide-resistant M. pneumoniae infection was 81.6% in 2012 (493/604) (Table 2). Rates of macrolide-resistant M. pneumoniae infection gradually decreased as follows: 65.8% (25/38) in 2013, 59.3% (16/27) in 2014, and 43.6% (65/149) in 2015. The most frequent mutation was A2063G mutation (95.8%), followed by A2063T (3.1%), A2064G (0.6%), A2063C (0.3%), C2617G (0.2%), and C2617T (0.1%).
The prevalence of macrolide-resistant M. pneumoniae infection was high during 2008–2012 but gradually decreased throughout Japan during 2013–2015. One reason for this decrease might be the 2011 publication of guidelines for treating M. pneumoniae pneumonia (4). Because of the higher prevalence of macrolide-resistant M. pneumoniae infection in children than in adults (5), respiratory fluoroquinolone/tosufloxacin was recommended for use in patients in whom M. pneumoniae pneumonia responded poorly to macrolide treatment in these guidelines. After the pandemic, the guideline committee addressed the concerns of further accumulation of macrolide-resistant M. pneumoniae in children resulting from constant macrolide use and teeth damage from tetracycline use in children <8 years of age (4). Tosufloxacin was approved for use in children in 2010 in Japan and has been used to treat M. pneumoniae infection. Because the guidelines recommend tosufloxacin as a second-line drug, pediatricians in Japan may be using the appropriate antimicrobial drugs for M. pneumoniae infection in accordance with these guidelines, which might have led to the decrease in incidence. Unfortunately, to our knowledge, no reports have been published to support these hypotheses. However, prescriptions for oral antimicrobial drugs in Japan comprise most of the prescriptions for antimicrobial drugs (6); therefore, we believe that the rate of macrolide-resistant M. pneumoniae might be affected by changes in the use of oral macrolide agents.
In addition, the prevalence of macrolide-resistant M. pneumoniae infection varies among countries: for example, 13.2% in the United States (7), 8.3% in France (8), and 3.1% in Germany (9). These variations might be attributed to differences in the number of prescription macrolide agents among countries. Although accurately assuming the number of prescription macrolide agents in each country is difficult, we can estimate the macrolide resistance rate of Streptococcus pneumoniae among those countries. A recent report supported the hypothesis that antimicrobial selection pressure results in clonal expansion of existing resistant strains (10). In Japan and China, which have a high prevalence of macrolide-resistant M. pneumoniae (3,5), the rates of macrolide-resistant S. pneumoniae also are very high (11,12). In countries with low rates of macrolide-resistant M. pneumoniae, such as the United States, France, and Germany (7,8,9), the prevalence rates of macrolide-resistant S. pneumoniae are low (13,14). However, the mechanisms of macrolide resistance are difficult to compare between Streptococcus and Mycoplasma (15). Therefore, we also interviewed the pediatricians who collected the samples for this study, and analysis of their responses is ongoing. Upon completion of these interviews, we will be able to report patients’ background characteristics, such as previous use of macrolides and medical examination histories; this information may provide further insight into the decreased prevalence of macrolide-resistant M. pneumoniae infection.
In summary, the prevalence of macrolide-resistant M. pneumoniae infection in children in Japan was high and increased between 2008 and 2012 but declined thereafter. Careful continuous monitoring of macrolide-resistant M. pneumoniae infection rates in Japan and other countries is needed.
Dr. Tanaka is a senior lecturer in the Department of Pediatrics at Kawasaki Medical School. His primary research interest is vaccines for travelers.
Acknowledgment
We thank Reiji Kimura and Mika Suwa for their technical assistance and all the clinicians who participated by taking samples in the Atypical Pathogen Study Group.
References
- Kawai Y, Miyashita N, Kubo M, Akaike H, Kato A, Nishizawa Y, et al. Nationwide surveillance of macrolide-resistant Mycoplasma pneumoniae infection in pediatric patients. Antimicrob Agents Chemother. 2013;57:4046–9. DOIPubMedGoogle Scholar
- Zhao F, Liu G, Wu J, Cao B, Tao X, He L, et al. Surveillance of macrolide-resistant Mycoplasma pneumoniae in Beijing, China, from 2008 to 2012. Antimicrob Agents Chemother. 2013;57:1521–3. DOIPubMedGoogle Scholar
- Miyashita N, Kawai Y, Yamaguchi T, Ouchi K, Oka M; Atypical Pathogen Study Group. Clinical potential of diagnostic methods for the rapid diagnosis of Mycoplasma pneumoniae pneumonia in adults. Eur J Clin Microbiol Infect Dis. 2011;30:439–46. DOIPubMedGoogle Scholar
- Joint Committee of Japanese Society of Pediatric Infectious Disease and Japanese Society of Pediatric Pulmonology. Pneumonia. In: Ouchi K, Kurosaki T, Okada K, editors. Guidelines for management of respiratory infectious diseases in children in Japan 2011 [in Japanese]. Tokyo: Kyowa Kikaku Ltd.; 2011. p. 44
- Miyashita N, Kawai Y, Akaike H, Ouchi K, Hayashi T, Kurihara T, et al.; Atypical Pathogen Study Group. Macrolide-resistant Mycoplasma pneumoniae in adolescents with community-acquired pneumonia. BMC Infect Dis. 2012;12:126. DOIPubMedGoogle Scholar
- Muraki Y, Yagi T, Tsuji Y, Nishimura N, Tanabe M, Niwa T, et al. Japanese antimicrobial consumption surveillance: First report on oral and parenteral antimicrobial consumption in Japan (2009-2013). J Glob Antimicrob Resist. 2016;7:19–23. DOIPubMedGoogle Scholar
- Zheng X, Lee S, Selvarangan R, Qin X, Tang YW, Stiles J, et al. Macrolide-Resistant Mycoplasma pneumoniae, United States. Emerg Infect Dis. 2015;21:1470–2. DOIPubMedGoogle Scholar
- Pereyre S, Touati A, Petitjean-Lecherbonnier J, Charron A, Vabret A, Bébéar C. The increased incidence of Mycoplasma pneumoniae in France in 2011 was polyclonal, mainly involving M. pneumoniae type 1 strains. Clin Microbiol Infect. 2013;19:E212–7. DOIPubMedGoogle Scholar
- Dumke R, Schnee C, Pletz MW, Rupp J, Jacobs E, Sachse K, et al.; Capnetz Study Group. Mycoplasma pneumoniae and Chlamydia spp. infection in community-acquired pneumonia, Germany, 2011-2012. Emerg Infect Dis. 2015;21:426–34. DOIPubMedGoogle Scholar
- Keenan JD, Klugman KP, McGee L, Vidal JE, Chochua S, Hawkins P, et al. Evidence for clonal expansion after antibiotic selection pressure: pneumococcal multilocus sequence types before and after mass azithromycin treatments. J Infect Dis. 2015;211:988–94. DOIPubMedGoogle Scholar
- Okade H, Funatsu T, Eto M, Furuya Y, Mizunaga S, Nomura N, et al. Impact of the pneumococcal conjugate vaccine on serotype distribution and susceptibility trends of pediatric non-invasive Streptococcus pneumoniae isolates in Tokai, Japan over a 5-year period. J Infect Chemother. 2014;20:423–8. DOIPubMedGoogle Scholar
- Liu WK, Liu Q, Chen DH, Liang HX, Chen XK, Chen MX, et al. Epidemiology of acute respiratory infections in children in Guangzhou: a three-year study. PLoS One. 2014;9:e96674. DOIPubMedGoogle Scholar
- Jenkins SG, Farrell DJ. Increase in pneumococcus macrolide resistance, United States. Emerg Infect Dis. 2009;15:1260–4. DOIPubMedGoogle Scholar
- Farrell DJ, Couturier C, Hryniewicz W. Distribution and antibacterial susceptibility of macrolide resistance genotypes in Streptococcus pneumoniae: PROTEKT Year 5 (2003-2004). Int J Antimicrob Agents. 2008;31:245–9. DOIPubMedGoogle Scholar
- Bley C, van der Linden M, Reinert RR. mef(A) is the predominant macrolide resistance determinant in Streptococcus pneumoniae and Streptococcus pyogenes in Germany. Int J Antimicrob Agents. 2011;37:425–31. DOIPubMedGoogle Scholar
Figure
Tables
Cite This ArticleNo table matches the in-text citation "Table 1". Please supply the missing table or delete the citation.
Table of Contents – Volume 23, Number 10—October 2017
| EID Search Options |
|---|
|
|
|
|
|
|
Please use the form below to submit correspondence to the authors or contact them at the following address:
Tomohiro Oishi, Kawasaki Medical School—Pediatrics, 577, Matsushima, Kurashiki Kurashiki Okayama 701-0192, Japan
Top