Volume 26, Number 8—August 2020
Dispatch
In Vivo Observation of Trombiculosis with Fluorescence–Advanced Videodermatoscopy
Cite This Article
Citation for Media
Abstract
Trombiculosis is a skin infestation by larvae of mites of the Trombiculidae family. We used fluorescence–advanced videodermatoscopy to diagnose trombiculosis in a woman in Italy with targetoid patches. This method might be useful for identifying atypical manifestations of trombiculosis.
Trombiculosis is a skin infestation by mites of the Trombiculidae family. Adult mites live and reproduce on the surface of the soil, whereas the larvae feed on warm-blooded vertebrates, including humans. These mites are also known as harvest mites or chiggers (1–6). Although trombiculid mites are endemic to most of the world, certain soil conditions limit common habitats to grassy fields, forests, parks, gardens, and the moist areas along lakes and streams (1).
Only the larvae (0.15–0.3 mm long), which are 6-legged and orange or bright red, are responsible for chigger bites. Infestation usually occurs when larvae are particularly abundant, throughout late summer and autumn (7). Trombiculid larvae do not burrow into the host’s skin; instead, they use jaw-like structures to attach to hairless areas on the host, secrete digestive enzymes that liquify host epidermal cells, and feed on broken-down tissue and digested cutaneous cells for 2–10 days (1). The host response is mainly caused by sensitization to the injected saliva. Its intensity might vary from a slightly irritable erythema to papules or vesicles with intense itch (3).
Trombiculid mites are also vectors of infectious agents. For example, Leptotrombidium mites transmit scrub typhus in the Far East (3). We report a case of trombiculosis in a woman in Turin, Italy.
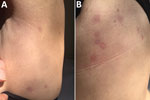
In April 2019, a 48-year-old woman sought care at the Dermatology Clinic University Hospital of Turin, Turin, Italy, for onset of small roundish, targetoid patches, with peripheral erythematous halo and central lightening, on her trunk (Figure 1). The patient was otherwise in good health. A few days before the patches developed, she had gone on an excursion in the countryside near Turin.
We used fluorescence–advanced videodermatoscopy (FAV), an optical electronic system using a monochromatic light‐emitting source with an λ of 405 nm (±5 nm) and a field of view of 340 μm, to examine the patient. FAV uses the ability of endogenous molecules to absorb specific wavelengths and emit fluorescence. The examination is conducted in vivo; the optical device is directly applied to the skin. The images are visualized in real time by using grayscale to indicate the levels of light absorption (i.e., black indicates no fluorescence, and white indicates highest fluorescence) (8).
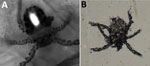
By placing the probe on one of the patient’s skin patches, we visualized an oval-shaped mite with 3 pairs of legs (Figure 2, panel A). We identified this image as the larval stage of a trombiculid mite. We also detected 1 larva on direct examination from a superficial skin scraping (Figure 2, panel B). The patient was treated with topical ivermectin for 7 days, resulting in complete regression of the lesions.
FAV facilitated a real-time diagnosis of trombiculosis in this case. Without this technology, the diagnosis of trombiculosis might have been complicated by the atypical presentation (trombiculosis usually manifests with erythematous homogeneous macules) and seasonality of the case (in Italy, trombiculosis is more prevalent in autumn [7]). To our knowledge, the diagnosis of trombiculosis using videodermatoscopic findings has been reported in only 1 other case (6).
We suspect the agent responsible for this case was the Neotrombicula autumnalis mite, the most frequent causative agent of trombiculosis in Europe (2–5). However, because we could not collect larvae to perform morphologic or molecular studies, we could not definitively identify the mite. At least 4 other species of trombiculid mites might cause trombiculosis in Europe, namely Kepkatrombicula desaleri, Blankaartia acuscutellaris, Trombicula toldti, and N. inopinata (9). In addition, the number of cases caused by species other than N. autumnalis might be underestimated because, in most reports, the identification does not appear to have been attributed with sufficient taxonomic criteria (9).
Dr. Ramondetta is a fourth-year resident in dermatology at the Dermatology Clinic of the University of Turin. Her primary research interests include pediatric dermatology and digital dermoscopy.
References
- McClain D, Dana AN, Goldenberg G. Mite infestations. Dermatol Ther. 2009;22:327–46. DOIPubMedGoogle Scholar
- Gasser R, Wyniger R. Distribution and control of Trombiculidae with special reference to Trombicula autumnalis [in German]. Acta Trop. 1955;12:308–26.PubMedGoogle Scholar
- di Meo N, Fadel M, Trevisan G. Pushing the edge of dermoscopy in new directions: entomodermoscopy of Trombicula autumnalis. Acta Dermatovenerol Alp Panonica Adriat. 2017;26:45–6. DOIPubMedGoogle Scholar
- Brennan JM, Goff ML. Keys to the genera of chiggers of the western hemisphere (acarina: trombiculidae). J Parasitol. 1977;63:554–66. DOIPubMedGoogle Scholar
- Nasca MR, Lacarrubba F, Micali G. Diagnosis of trombiculosis by videodermatoscopy. Emerg Infect Dis. 2014;20:1059–60. DOIPubMedGoogle Scholar
- Leone F, Di Bella A, Vercelli A, Cornegliani L. Feline trombiculosis: a retrospective study in 72 cats. Vet Dermatol. 2013;24:535–e126. DOIPubMedGoogle Scholar
- Sanlorenzo M, Vujic I, De Giorgi V, Tomasini C, Deboli T, Quaglino P, et al. Fluorescence-advanced videodermatoscopy: a new method for in vivo skin evaluation. Br J Dermatol. 2017;177:e209–10. DOIPubMedGoogle Scholar
- Stekolnikov AA, Santibáñez P, Palomar AM, Oteo JA. Neotrombicula inopinata (Acari: Trombiculidae) - a possible causative agent of trombiculiasis in Europe. Parasit Vectors. 2014;7:90. DOIPubMedGoogle Scholar
Figures
Cite This ArticleOriginal Publication Date: July 14, 2020
1These first authors contributed equally to this article.
2These senior authors contributed equally to this article.
Table of Contents – Volume 26, Number 8—August 2020
| EID Search Options |
|---|
|
|
|
|
|
|
Please use the form below to submit correspondence to the authors or contact them at the following address:
Alice Ramondetta, Dermatology Clinic, Department of Medical Sciences, University of Turin, via Cherasco 23 Turin, Italy
Top