Volume 27, Number 12—December 2021
Research
Novel Filoviruses, Hantavirus, and Rhabdovirus in Freshwater Fish, Switzerland, 2017
Cite This Article
Citation for Media
Abstract
European perch (Perca fluviatilis) are increasingly farmed as a human food source. Viral infections of European perch remain largely unexplored, thereby putting farm populations at incalculable risk for devastating fish epizootics and presenting a potential hazard to consumers. To address these concerns, we applied metatranscriptomics to identify disease-associated viruses in European perch farmed in Switzerland. Unexpectedly, in clinically diseased fish we detected novel freshwater fish filoviruses, a novel freshwater fish hantavirus, and a previously unknown rhabdovirus. Hantavirus titers were high, and we demonstrated virus in macrophages and gill endothelial cells by using in situ hybridization. Rhabdovirus titers in organ samples were low, but virus could be isolated on cell culture. Our data add to the hypothesis that filoviruses, hantaviruses, and rhabdoviruses are globally distributed common fish commensals, pathogens, or both. Our findings shed new light on negative-sense RNA virus diversity and evolution.
Aquaculture is the fastest growing sector in food production worldwide (1); innovative intensive production technologies, such as recirculating aquaculture systems (RAS), are becoming increasingly valuable for commercial fish production (2). Attempts are continually made to introduce new types of fish into aquaculture, to reduce overfishing of wild fish populations, and to satisfy the progressing consumer demand for diverse supply (3). One example is the adaptation of European perch (Perca fluviatilis; order Perciformes, family Percidae) to farming and the growing consumer interest in this fish (4). European perch are actinopterygiids that naturally inhabit slow-flowing rivers, lakes, or ponds in Europe and northern Asia. During the 19th century, they were introduced into Australia as angling fish, where they are now considered invasive, competing with native fish for food and space, preying on other fish, and breeding to overpopulation (5).
Although little is known about diseases affecting European perch in the wild, infectious diseases lead to high mortality among these fish in aquaculture, making farming of these fish economically challenging. One of the main threats is infection with perch rhabdovirus (PRV; family Rhabdoviridae, genus Perhabdovirus), leading to the central nervous system (CNS) signs of loss of equilibrium and aberrant swimming behavior and to higher mortality (2,6–10). Lack of investigation of the occurrence and diversity of other pathogenic virus infections of European perch that result in disease impairs the treatment, control, and prevention of disease outbreaks in farm populations. To address this knowledge gap, we applied virus diagnostics, including metatranscriptomics (virus discovery by high-throughput RNA sequencing and bioinformatics), to a set of samples collected from sick juvenile European perch at a perch farm in Switzerland in 2017. Although we did not find PRV in these fish, our investigation led to the discovery of 5 novel negative-sense RNA viruses, belonging to the negarnaviricot families Rhabdoviridae, Filoviridae, and Hantaviridae, that could possibly contribute to disease development.
European Perch Origin
The European perch used in this study were raised in a private pond in Saxony, Germany, and were exported to a farm that uses RAS in Bernese Oberland, Switzerland, at 16 g (≈11,200 fish) and 33 g (≈4,800 fish). Eleven live juvenile European perch were sent to the Centre for Fish and Wildlife Health (FIWI), University of Bern (Bern, Switzerland), where they were euthanized and subjected to microbiological (including parasitologic, bacteriologic, mycologic, and virologic) and pathologic (including histopathologic) examination. Because the remaining fish at the farm exhibited clinical signs of disease, they were kept in quarantine for an additional 2 months and subsequently euthanized.
Cell Culture
We used bluegill (Lepomis macrochirus) fry cells (BF-2) and fathead minnow (Pimephales promelas) epithelioma papulosum cyprini (EPC) cells. These cells were originally obtained from the Friedrich-Loeffler-Institute, Federal Research Institute for Animal Health (Greifswald-Insel Riems, Germany; Collection of Cell Lines in Veterinary Medicine, catalog nos. CCLV-RIE 290 and CCLV-RIE 173 are maintained at FIWI).
We inoculated the cells with small pieces of pooled CNS or spleen, kidney, heart, and pyloric ceca tissue from 5 of the 11 euthanized European perch (in total <5 g) and incubated at 15°C. We monitored the cell cultures for CPE daily for 7 days by using light microscopy and then monitored subcultures for another 7 days. We harvested supernatants from cell cultures showing CPE and tested by reverse transcription PCR (RT-PCR) for the common European perch pathogen PRV, targeting the glycoprotein (G) gene of PRV isolate 9574.1 (GenBank accession no. JF502613), according to a previously published protocol, by using the primer pair oPVP116/118 and oPVP126/Rha2 (11).
High-Throughput Sequencing and Bioinformatics
We extracted total RNA from fresh-frozen pooled visceral organs and CNS tissue, originally taken for cell culture inoculation by using TRI Reagent (Sigma Life Sciences, https://www.sigmaaldrich.com), according to the manufacturer’s instructions. We then prepared a high-throughput sequencing (HTS) library with the TruSeq Stranded Total RNA kit (Illumina, https://www.illumina.com) and performed HTS on a HiSeq 3000 machine (Illumina), generating paired-end reads of 2 × 150 bp. We performed bioinformatic analysis as described previously (12) (Appendix).
Reverse Transcription PCR, Rapid Amplification of cDNA Ends, and Sanger Sequencing
To fill gaps between HTS scaffolds, we reverse-transcribed extracted RNA to cDNA, performed PCRs, and subjected the amplicons to Sanger sequencing (Appendix). We performed 3′ and 5′ rapid amplification of cDNA ends (RACE), as described previously, on RNA extracted from pooled organs and CNS tissue as well as cell culture supernatants (13) (Appendix).
Taxonomic Analyses
We performed taxonomic analyses by using protein or nucleic acid sequences, following precedents established by the International Committee on Taxonomy of Viruses (ICTV) Filoviridae (14,15), Hantaviridae (16), and Rhabdoviridae (17) Study Groups. New filovirus-like genome sequences were analyzed by using PAirwise Sequence Comparison (PASC) (18) and maximum-likelihood phylogenetics. Filovirus phylogenetic estimations were inferred in FastTree version 2.1 (19) by using a general time-reversible model with 20 gamma-rate categories, 5,000 bootstrap replicates, and exhaustive search parameters (-slow) and pseudocounts (-pseudo). The new rhabdovirus-like sequence was taxonomically placed via analysis of its full-length large protein gene (L) sequence using maximum-likelihood phylogenetics. We applied the maximum-likelihood method in MEGA X (20) with 1,000 bootstraps for rhabdovirus and hantavirus phylogenetic estimations.
Histopathology and In Situ Hybridization
During a complete necropsy of the 11 fish, we used 3 whole perch for histologic examination. We fixed these fish in 10% buffered formalin for 24 hours and embedded cut sections of the gills, longitudinal head sections, and longitudinal cut sections of the body cavity in paraffin. We prepared 3-μm sections and stained them with hematoxylin and eosin according to standard protocols. We conducted chromogenic in situ hybridization on all formalin-fixed paraffin-embedded (FFPE) tissues used for histopathology. We performed staining with the RNAscope system (Advanced Cell Diagnostics, https://acdbio.com) (Appendix).
Clinical and Pathologic Findings
In December 2016, a total of 16,000 juvenile European perch were imported from Saxony, Germany, to a farm using RAS in Bernese Oberland, Switzerland. After arrival, the fish were routinely quarantined. Shortly after arrival, because of a high death rate of 1% per day (reference range 0.01%–0.03% per day), 11 randomly selected live fish were sent to FIWI for microbiological and pathologic examination. Clinical signs included anorexia, lethargy, skin ulcerations, multifocal hemorrhages, and eroded tail fins. Culture and PCR indicated that skin ulcerations were caused by oomycete (Saprolegnia parasitica) infection (21). Histopathology revealed mild to moderate gill epithelial proliferation and epithelial cell hypertrophy. Additional findings included necrotizing dermatitis with hemorrhage and intralesional oomycete hyphi and bacterial colonies. The newly arrived fish were treated with flubendazole, formalin, and peracetic acid, but deaths increased to 1.8%–4.2% per day. Two months after importation, deaths for the quarantined fish reached 22%–27% in total. All remaining fish were euthanized and discarded, thereby preventing these fish from entering the food chain.
Virus Isolation
For routine virus investigation, we exposed standard fish cell cultures (BF-2 and EPC) to suspensions of pooled perch CNS and pooled visceral organs. We selected these cell lines because of their high susceptibility to diverse fish viruses (22). CPE developed 10 days after inoculation of CNS suspension into BF-2 cells and 13 days after inoculation into EPC cells. Affected cell supernatants were harvested and tested preliminarily positive for PRV infection by RT-PCR. The sequence of the detected amplicon was, however, only 78% identical to the sequence of the perhabdovirus lake trout rhabdovirus (LTRV; GenBank accession no. AF434991), indicating the presence of a perhabdovirus distinct from PRV/ LTRV.
Novel Perhabdovirus
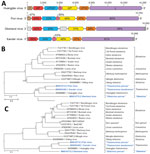
To further characterize the putative novel perhabdovirus, we performed metatranscriptomics by using HTS and bioinformatic analysis of pooled CNS tissue and visceral organ RNA extracts of 5 fish. We found 16 sequence scaffolds 247–1,454-nt long with mean k-mer coverages of 1.0–5.9 and nucleotide sequence identities of 62%–98% to perhabdovirus genomes (Appendix Table 1). We mapped these scaffolds to LTRV (GenBank accession no. AF434991) and PRV (GenBank accession no. JX679246) as references and closed sequence gaps by RT-PCR and RACE followed by Sanger sequencing. The resulting complete genome of the novel virus was 11,595-nt long and had the characteristic genomic organization of (pe)rhabdoviruses (Figure 1, panel A). Each of the open reading frames (ORFs) is flanked by conserved transcriptional initiation (3′-UUGUUC) and termination/polyadenylation (3′-AURC[U]7) signals with inverse complementarity of 13 nt of the 3′ and 5′ terminal genome sequences.
Phylogenetic comparison of the L gene confirmed a close relationship of the new virus to the 2 members of the species Sea trout perhabdovirus (i.e., LTRV and Swedish sea trout virus; GenBank accession no. AF434992) (Figure 1, panel B) and a recently described and thus far unclassified virus from a percid (sea trout rhabdovirus isolate 18/203; GenBank accession no. MN963997). This relationship is also reflected in phylogenetic comparisons of the nucleoprotein (N), phosphoprotein (P), matrix protein (M), and G genes (Appendix Figure 1). The current perhabdovirus sequence-based species demarcation criterion is a minimum divergence of 15% in the L gene (17). The L gene sequence of the novel virus is most closely related to that of the isolate 18/203 (divergence 10%); L genes of both viruses are ≈19% divergent from the next closest related perhabdovirus, sea trout perhabdovirus isolate R6146 (GenBank accession no. MN963999), thus indicating that we discovered a novel perhabdovirus that should be assigned to a new species along with isolate 18/203. We named the new virus Egli (EGLV) virus after a local Swiss-German word for European perch and deposited the complete viral genome sequence into GenBank (accession no. MN510774). Detection of EGLV in European perch FFPE tissue sections by RNA in situ hybridization was unsuccessful.
Four Novel Filoviruses
In addition to scaffolds that ultimately led us to identify EGLV, we found 41 scaffolds 240–4,726-nt long with low k-mer coverage (0.8–4.8×). The deduced amino acid sequences were 28%–30% identical to proteins of Huángjiāo virus (HUJV), a recently identified virus of marine greenfin horse‐faced filefish (Thamnaconus septentrionalis) captured in the East China Sea (23) (Appendix Table 1). Alignment of these scaffolds to the HUJV genome (GenBank accession no. MG599981) and the deduced amino acid sequences to those of HUJV-encoded proteins revealed a complex scenario suggesting the presence of several distinct thamnoviral genomes, however with numerous gaps between the scaffolds. To obtain complete or coding-complete viral genome sequences, we resequenced the HTS library to generate ≈10 times more paired-end reads (2,051,046,671) than during the initial HTS run, reassembled the sequences, and performed RT-PCR and Sanger sequencing to bridge sequence gaps. This effort resulted in 3 long scaffolds of 13,764 nt (k-mer coverage 24×), 14,593 nt (k-mer coverage 15×), and 13,066 nt (k-mer coverage 5×), corresponding to 3 novel viruses.
We named these viruses Fiwi virus (FIWIV; GenBank accession no. MN510772), after FIWI; Oberland virus (OBLV; GenBank accession no. MN510773), after Bernese Oberland; and Kander virus (KNDV; GenBank accession no. MW093492), after the Kander River, which flows through Bernese Oberland. Whereas the FIWIV genome appears to be coding complete, the sequences of OBLV are coding incomplete at the 5′ terminus and of KNDV at both the 5′ and the 3′ termini. All attempts to determine the authentic 3′ and 5′ termini by RACE were unsuccessful, most likely because of low viral RNA loads. However, all 3 sequences have the genomic features of HUJV, encoding the filovirus-typical proteins nucleoprotein (NP), polymerase cofactor (VP35), glycoprotein (GP1,2), transcriptional activator (VP30), and large protein (L) containing an RNA-directed RNA polymerase (RdRp) domain, as well as 1–2 novel proteins (14,23,24) (Figure 2, panel A). Phylogenetic comparison of the FIWIV, OBLV, and KNDV genomic sequences (Figure 2, panel B) and L gene sequences with those of representative classified viruses of the family Filoviridae (Figure 2, panel C) confirmed the genetic relationship of all 3 viruses to HUJV. The current demarcation criteria for filovirus sequence-based genus and species are >55% and >23% sequence divergence over complete genome sequences determined by using PASC (14,15). We found a pairwise divergence of 49% compared with HUJV by using the FIWIV genome sequence (Table), indicating that FIWIV is a member of a new thamnovirus species (“Thamnovirus percae”). The available KNDV genome sequence is 49% divergent from HUJV and 37% divergent from FIWIV (Table), suggesting that KNDV represents yet another novel thamnovirus species (“Thamnovirus kanderense”). In contrast, OBLV was >62% divergent from HUJV, FIWIV, and KNDV viruses and thus represents a new species (“Oblavirus percae”) within a new genus (“Oblavirus”). All attempts to detect FIWIV or OBLV viruses in 3 European perch FFPE tissue sections were unsuccessful.
In addition to FIWIV, OBLV, and KNDV, we found 4 shorter scaffolds (573–3,259 nt; Appendix Figure 2) similar to various HUJV genes, but attempts to demonstrate a physical linkage of these sequences by RT-PCR were not successful. Still, the presence of these scaffolds is indicative of at least 1 additional novel filovirus, which we were not able to further characterize.
Novel Hantavirus
In the same RNA extract of pooled organs, we found 3 scaffolds, of which the deduced amino acid sequences were 25%–35% similar to those of the large (L), medium (M), and small (S) segments of Wēnlǐng minipizza batfish virus (WEMBV) and Wēnlǐng red spikefish virus (WERSV). WEMBV was recently identified in apparently healthy minipizza batfish (Halieutaea stellate) and WERSV in red spikefish (Triacanthodes anomalus) captured in the East China Sea (Appendix Table 1) (23). The mean k-mer coverage of these scaffolds ranged from 3 × 103 to 1.5 × 104, indicating a high viral RNA load. Using Sanger sequencing and RACE, we determined the complete sequences of the genomic L (GenBank accession no. MN510769), M (GenBank accession no. MN510770), and S (GenBank accession no. MN510771) segments of a novel virus, here named Bern perch virus (BPV). Similar to WEMBV and WERSV, the BPV L segment (6,372 nt) was deduced to encode the L protein including an RdRp domain, the M segment (3,804 nt) was deduced to encode the glycoprotein precursor, and the S segment (2,435 nt) was deduced to encode the nucleocapsid protein (Figure 3, panel A). The S and M segments contain 2 additional ORFs encoding putative proteins not represented in current protein databases in antisense (S segment) and sense (M segment) orientation. Alignment of the 3′ and 5′ sequences of all 3 segments revealed that the 8 terminal nucleotides are complementary within and conserved among segments (Figure 3, panel B), a known feature of members of the order Bunyavirales (25). However, these terminal sequences differ from those of members of the genus Orthohantavirus and are similar to those of members of the genus Orthobunyavirus (Appendix Table 1). Phylogenetic analysis of the L protein confirmed the close relationship of BPV to all currently classified actinoviruses (Figure 3, panel C; Appendix Figure 3) but indicated the need for a novel species to accommodate this virus. This need was confirmed by DEmARC (Diversity Partitioning by Hierarchical Clustering) analysis (26); on the basis of this evidence, the ICTV officially established this species as Perch actinovirus in 2021 (27,28).
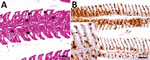
Using in situ hybridization on FFPE tissue sections, we were able to detect BPV genomic RNA in gills with histopathologic lesions of 2 fish (Figure 4, panels A, B; Appendix Figure 4) and in a granuloma in the perivisceral fat tissue of 1 of these animals. Morphologically, we identified the affected cells in the gills and the perivisceral fat tissue as putative macrophages. In addition, putative endothelial cells were labeled positively in the gills (Figure 4, panel B).
The diversity of fish viruses, in particular that of RNA viruses, remains poorly understood (29). Recent initial studies indicate that this diversity is enormous and that many viral taxa that have been established for pathogens of humans and other mammals need to be redefined (23,30–32). Husbandry conditions in huge tanks used on farms may favor the emergence and rapid intraspecies and interspecies transmission of fish viruses, potentially resulting in high economic loss for the fish industry. Also of concern is the introduction of novel viruses from fish farms into native fauna, which could have disastrous ecologic consequences. In addition, viruses of unknown pathogenicity in food animals may have zoonotic potential of yet unpredictable importance.
In this study, we identified 1 novel rhabdovirus, 4 novel filoviruses (3 confirmed and 1 likely), and 1 novel hantavirus in morbid farmed European perch. The discovery of a novel rhabdovirus was not surprising and adds to the role of rhabdoviruses in fish health; rhabdoviruses, in particular those of the genera Novirhabdovirus, Perhabdovirus, Sprivivirus, and Vesiculovirus, are notorious marine and freshwater fish pathogens, causing diseases characterized by high lethality (33,34). Thus, these viruses pose a considerable threat to aquaculture. The European perch examined in this study exhibited signs compatible with rhabdovirus infection (7). Using HTS and exposing cell cultures to CNS tissue suspensions, we discovered a novel perhabdovirus, Egli virus. Although the viral RNA loads in tissues were low and we could not detect RNA by in situ hybridization in tissue sections, we were able to isolate the virus from brain tissue. Rhabdoviruses in perch are usually associated with disease and not known as commensals. The host range of Egli virus is unknown, but genetically it is more closely related to viruses identified in trout than to those infecting perch, suggesting the possibility of cross-species transmission, highlighting a concern for farms that raise fish other than perch and for native fish populations. Discovery of a lake trout rhabdovirus as a probable cause of disease in European perch in Ireland (7) supports the hypothesis of high potential for interspecies transmission of these viruses.
Unexpectedly, we were able to assemble near-complete viral genomes of 3 novel filoviruses (Fiwi, Oberland, and Kander viruses) and detected a likely fourth filovirus in the diseased perch. Until recently, filoviruses, notorious for causing disease in humans with extremely high lethality (35), were thought to exclusively infect mammals. This view changed with the discovery of Xīlǎng virus (the only member of genus Striavirus) in striated frogfish (Antennarius striatus ) and HUJV in greenfin horse‐faced filefish captured in the East China Sea (23) as well as apparent thamnoviruses John Dory filovirus in John Dory (Zeus faber) and blue spotted goatfish filovirus in blue spotted goatfish (Upeneichthys lineatus) purchased at a fish market in Sydney, New South Wales, Australia (31). However, currently, <300 nt contigs are known from John Dory fish and blue spotted goatfish filoviruses; hence, their true taxonomic affiliation remains to be determined. In contrast to Xīlǎng virus, HUJV, John Dory filovirus, and blue spotted goatfish filovirus (all of which were found in marine fish in China and Australia), FIWIV, OBLV, and KNDV apparently infect freshwater fish in Europe. This geographic and ecologic distribution indicates that filoviruses are broadly dispersed fish commensals or potential pathogens that probably number in the hundreds or thousands. Although on the basis of our data we cannot attribute filovirus infection to individual fish, a possible scenario includes co-infection with FIWIV, OBLV, and KNDV, EGLV, or BPV, or any combination of these viruses. Thamnovirus abundance was low in the sampled European perch, and detection of thamnoviral RNA proved impossible in FFPE tissues. This low abundance, together with the unsuccessful attempt to demonstrate viral RNA in the tissue, suggests that infection in the investigated fish was subclinical rather than the cause of the observed clinical signs.
We also identified a novel hantavirus, Bern perch virus. Hantaviruses are best known as rodent-borne viruses of the mammantavirin genus Orthohantavirus, which cause hemorrhagic fever with renal syndrome or hantavirus pulmonary syndrome in humans (36) but have also been found in bats and eulipotyphla. Reptile hantaviruses (family Repantavirinae, genus Reptillovirus) and fish hantaviruses (family Actantavirinae, genus Actinovirus, and family Agantavirinae, genus Agnathovirus) (32) have only recently been discovered. The fish viruses include the actinoviruses WEMBV, WERSV (23), and Wēnlǐng yellow goosefish virus (detected in yellow goosefish [Lophius litulon] captured in China [23]); the likely actinovirus Aronnax virus, found in pygmy goby (Eviota zebrina) purchased at a fish market in Sydney (31); and the agnathovirus Wēnlǐng hagfish virus, detected in inshore hagfish (Eptatretus burgeri) captured in China (23). Similar to our filovirus findings, the discovery of BPV is remarkable because this actinovirus was found in freshwater fish from Europe rather than in marine fish from China or Australia. Actinoviruses have not yet been associated with disease in fish. Using in situ hybridization, we demonstrated, however, high concentrations of BPV RNA in macrophages and endothelial cells in the gills as well as in macrophages in the perivisceral fat tissue of morbid European perch. Other cell types tested negative. Human pathogenic orthohantaviruses predominantly infect macrophages and microvascular endothelial cells of a variety of organs, which leads to increased vascular permeability and severe disease (11). It is therefore tempting to speculate that the pathology observed in the gills of the European perch may have resulted in dyspnea, contributing to elevated mortality. In conclusion, our identification of new rhabdoviruses, filoviruses, and hantaviruses in farmed European perch in Switzerland raises concerns about the global distribution, host spectrum, and risks to human and animal health for these viruses.
Dr. Hierweger works as a postdoctoral researcher at the Division of Neurological Sciences, Vetsuisse Faculty, University of Bern. Her main research interest is the discovery of emerging viruses and their involvement in disease.
Acknowledgments
We thank Pamela Nicholson and the HTS platform, University of Bern, for performing the HTS runs, Elsbeth Keller-Gautschi and Regula Hirschi for excellent technical assistance, and Laurent Bigarré for scientific exchange and advice. We are grateful to Anya Crane for editing the manuscript.
This work was supported by the Swiss National Science Foundation (grant no. 31003A_163438 to R.B. and T.S.), the Swiss Food Safety and Veterinary Office (grant no. MON-108 to T.S.), and the Swiss Innovation Agency (Innossuisse, grant no. 25178.1 PFLS-LS to H.S.-P). This work was supported in part through Laulima Government Solutions, LLC, prime contract with the US National Institute of Allergy and Infectious Diseases under contract no. HHSN272201800013C. J.H.K. performed this work as an employee of Tunnell Government Services, a subcontractor of Laulima Government Solutions, LLC, under contract no. HHSN272201800013C.
HTS raw data from this study have been deposited into the Sequence Read Archive (https://www.ncbi.nlm.nih.gov/sra) as SRR12586223.
References
- Food and Agriculture Organization of the United Nations. The state of world fisheries and aquaculture 2018. Meeting the sustainable development goals [cited 2021 Jul 10]. https://www.fao.org/3/i9540en/I9540EN.pdf
- Martins CIM, Eding EH, Verdegem MCJ, Heinsbroek LTN, Schneider O, Blancheton JP, et al. New developments in recirculating aquaculture systems in Europe: a perspective on environmental sustainability. Aquacult Eng. 2010;43:83–93. DOIGoogle Scholar
- Naylor RL, Hardy RW, Buschmann AH, Bush SR, Cao L, Klinger DH, et al. A 20-year retrospective review of global aquaculture. Nature. 2021;591:551–63. DOIPubMedGoogle Scholar
- Policar T, Schaefer FJ, Panana E, Meyer S, Teerlinck S, Toner D, et al. Recent progress in European percid fish culture production technology—tackling bottlenecks. Aquacult Int. 2019;27:1151–74. DOIGoogle Scholar
- Morgan DL, Gill HS, Maddern MG, Beatty SJ. Distribution and impacts of introduced freshwater fishes in Western Australia. N J Mar Freshwater Res. 2004;38:511–23. DOIGoogle Scholar
- Caruso C, Gustinelli A, Pastorino P, Acutis PL, Prato R, Masoero L, et al. Mortality outbreak by perch rhabdovirus in European perch (Perca fluviatilis) farmed in Italy: Clinical presentation and phylogenetic analysis. J Fish Dis. 2019;42:773–6. DOIPubMedGoogle Scholar
- Ruane NM, Rodger HD, McCarthy LJ, Swords D, Dodge M, Kerr RC, et al. Genetic diversity and associated pathology of rhabdovirus infections in farmed and wild perch Perca fluviatilis in Ireland. Dis Aquat Organ. 2014;112:121–30. DOIPubMedGoogle Scholar
- Rupp M, Knüsel R, Sindilariu P-D, Schmidt-Posthaus H. Identification of important pathogens in European perch (Perca fluviatilis) culture in recirculating aquaculture systems. Aquacult Int. 2019;27:1045–53. DOIGoogle Scholar
- Dorson M, Torchy C, Chilmonczyk S, Kinkelin P, Michel C. A rhabdovirus pathogenic for perch, Perca fluviatilis L.: isolation and preliminary study. J Fish Dis. 1984;7:241–5. DOIGoogle Scholar
- Dannevig BH, Olesen NJ, Jentoft S, Kvellestad A, Taksdal T, Håstein T. The first isolation of a rhabdovirus from perch (Perca fluviatilis) in Norway. Bull Eur Assoc Fish Pathol. 2001;21:186–94.
- Talbi C, Cabon J, Baud M, Bourjaily M, de Boisséson C, Castric J, et al. Genetic diversity of perch rhabdoviruses isolates based on the nucleoprotein and glycoprotein genes. Arch Virol. 2011:156:2133–44. DOIGoogle Scholar
- Kauer RV, Koch MC, Hierweger MM, Werder S, Boujon CL, Seuberlich T. Discovery of novel astrovirus genotype species in small ruminants. PeerJ. 2019;7:
e7338 . DOIPubMedGoogle Scholar - Hierweger MM, Werder S, Seuberlich T. Parainfluenza virus 5 infection in neurological disease and encephalitis of cattle. Int J Mol Sci. 2020;21:498. DOIPubMedGoogle Scholar
- Kuhn JH, Amarasinghe GK, Basler CF, Bavari S, Bukreyev A, Chandran K, et al.; Ictv Report Consortium. ICTV virus taxonomy profile: Filoviridae. J Gen Virol. 2019;100:911–2. DOIPubMedGoogle Scholar
- Bào Y, Amarasinghe GK, Basler CF, Bavari S, Bukreyev A, Chandran K, et al. Implementation of objective PASC-derived taxon demarcation criteria for official classification of filoviruses. Viruses. 2017;9:106. DOIPubMedGoogle Scholar
- Laenen L, Vergote V, Calisher CH, Klempa B, Klingström J, Kuhn JH, et al. Hantaviridae: current classification and future perspectives. Viruses. 2019;11:788. DOIPubMedGoogle Scholar
- Walker PJ, Blasdell KR, Calisher CH, Dietzgen RG, Kondo H, Kurath G, et al.; Ictv Report Consortium. ICTV virus taxonomy profile: Rhabdoviridae. J Gen Virol. 2018;99:447–8. DOIPubMedGoogle Scholar
- National Center for Biotechnology Information. Pairwise Sequence Comparison (PASC) [cited 2021 Jul 10]. https://www.ncbi.nlm.nih.gov/sutils/pasc/viridty.cgi?textpage=overview
- Price MN, Dehal PS, Arkin AP. FastTree 2—approximately maximum-likelihood trees for large alignments. PLoS One. 2010;5:
e9490 . DOIPubMedGoogle Scholar - Kumar S, Stecher G, Li M, Knyaz C, Tamura K. MEGA X: Molecular Evolutionary Genetics Analysis across computing platforms. Mol Biol Evol. 2018;35:1547–9. DOIPubMedGoogle Scholar
- Ravasi D, De Respinis S, Wahli T. Multilocus sequence typing reveals clonality in Saprolegnia parasitica outbreaks. J Fish Dis. 2018;41:1653–65. DOIPubMedGoogle Scholar
- World Organisation for Animal Health. Aquatic Manual online access [cited 2021 Jul 10]. https://www.oie.int/standard-setting/aquatic-manual/access-online
- Shi M, Lin X-D, Chen X, Tian J-H, Chen L-J, Li K, et al. The evolutionary history of vertebrate RNA viruses. Nature. 2018;556:197–202. DOIPubMedGoogle Scholar
- Hume AJ, Mühlberger E. Distinct genome replication and transcription strategies within the growing filovirus family. J Mol Biol. 2019;431:4290–320. DOIPubMedGoogle Scholar
- Barr JN, Weber F, Schmaljohn CS. Bunyavirales: the viruses and their replication. In: Howley PM, Knipe DM, Whelan SPJ, editors. Fields virology. 7th ed. Philadelphia: Wolters Kluwer/Lippincott Williams & Wilkins; 2020. p. 706–49.
- Lauber C, Gorbalenya AE. Partitioning the genetic diversity of a virus family: approach and evaluation through a case study of picornaviruses. J Virol. 2012;86:3890–904. DOIPubMedGoogle Scholar
- International Committee on Taxonomy of Viruses [cited 2021 Jul 10]. https://talk.ictvonline.org/taxonomy
- Walker PJ, Siddell SG, Lefkowitz EJ, Mushegian AR, Adriaenssens EM, Alfenas-Zerbini P, et al. Changes to virus taxonomy and to the International Code of Virus Classification and Nomenclature ratified by the International Committee on Taxonomy of Viruses (2021). Arch Virol. 2021;166:2633–48. DOIPubMedGoogle Scholar
- Kibenge FSB, Godoy MG, editors. Aquaculture virology. Amsterdam: Elsevier; 2016. p. 568.
- Geoghegan JL, Di Giallonardo F, Cousins K, Shi M, Williamson JE, Holmes EC. Hidden diversity and evolution of viruses in market fish. Virus Evol. 2018;4:
vey031 . DOIPubMedGoogle Scholar - Geoghegan JL, Di Giallonardo F, Wille M, Ortiz-Baez AS, Costa VA, Ghaly T, et al. Virome composition in marine fish revealed by meta-transcriptomics. Virus Evol. 2021;7:veab005.
- Kuhn JH, Adkins S, Agwanda BR, Al Kubrusli R, Alkhovsky SV, Amarasinghe GK, et al. 2021 Taxonomic update of phylum Negarnaviricota (Riboviria: Orthornavirae), including the large orders Bunyavirales and Mononegavirales. Arch Virol. 2021;2021:31; Epub ahead of print. DOIPubMedGoogle Scholar
- Kurath G, Stone D. Fish rhabdoviruses (Rhabdoviridae). In: Bamford D, Zuckerman M, editors. Encyclopedia of Virology. 4th ed. Amsterdam: Elsevier; 2021. p. 324–31.
- LaPatra S, Misk E, al-Hussinee L, Lumsden JS. Rhabdoviruses of fish. In: Kibenge FSB, Godoy MG, editors. Aquaculture virology. Amsterdam: Elsevier; 2016. p. 276–97.
- Kuhn JH, Amarasinghe GK, Perry DL. Filoviridae. In: Howley PM, Knipe DM, Whelan SPJ, editors. Fields virology. 7th ed. Philadelphia: Wolters Kluwer/Lippincott Williams & Wilkins; 2020. p. 449–503.
- Kuhn JH, Charrel RN. Arthropod-borne and rodent-borne virus infections. In: Jameson JL, Fauci AS, Kasper DL, Hauser SL, Longo DL, Loscalzo J, editors. Harrison’s principles of internal medicine. 20th ed. Columbus (OH): McGraw-Hill Education; 2018. p. 1489–509.
Figures
Table
Cite This ArticleOriginal Publication Date: November 10, 2021
1These authors contributed equally to this article.
Table of Contents – Volume 27, Number 12—December 2021
| EID Search Options |
|---|
|
|
|
|
|
|


Please use the form below to submit correspondence to the authors or contact them at the following address:
Torsten Seuberlich, Divison of Neurological Sciences, Vetsuisse Faculty, University of Bern, Bremgartenstrasse 109a, CH-3012 Bern, Switzerland
Top