Volume 28, Number 12—December 2022
Dispatch
Hepatitis E Virus Infections in Free-Ranging and Captive Cetaceans, Spain, 2011–2022
Cite This Article
Citation for Media
Abstract
Epidemiologic surveillance of hepatitis E virus in over 300 free-ranging and captive cetaceans in waters off Spain revealed extensive exposure to this pathogen. We suggest the persistent and widespread presence of hepatitis E in the marine environment off the coast of Spain may be driven by terrestrial sources of contamination.
Paslahepevirus balayani (previously known as hepatitis E virus [HEV]; family Hepeviridae) is the leading cause of acute viral hepatitis in humans (1,2). Although 8 different genotypes of HEV have been identified, HEV-3 is the genotype with the broadest geographic distribution, including Europe, where the number of hepatitis E cases has sharply increased in the past decade (3). The main reservoirs of this genotype are suids, but a wide range of other land mammals has been shown to be susceptible to this emerging genotype (2). Although echinoderms and several bivalve shellfish species from coastal waters have tested positive for HEV, the susceptibility of other marine animals, such as cetaceans, to HEV has been unknown, as has their possible role in the epidemiology of this family of viruses (4). We conducted a large-scale study to determine the seroprevalence and prevalence of HEV in cetacean populations, both free-ranging and captive, in Spain, and to assess the dynamics of seropositivity in marine animals sampled longitudinally during the study period.
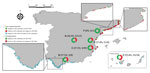
We collected blood and liver samples from 304 cetaceans belonging to 13 different species in Spain during 2011–2022 (Table 1; Figure 1). We based our study on 240 free-ranging animals found stranded on the Atlantic and Mediterranean coasts of Spain and 64 cetaceans kept in captivity at 6 aquatic parks (deemed A–F) in Spain. We performed longitudinal surveillance on 30 of the 64 animals kept in aquatic parks during the study period.
We assessed the presence of HEV antibodies in serum or plasma using a commercial multispecies ELISA (MP Biomedicals, https://www.mpbio.com) and, whenever possible, further investigated seropositivity by Western blot analysis (Appendix). We determined the presence of HEV RNA by using 2 broad-spectrum reverse transcription PCR (RT-PCR) assays in parallel (Appendix) (5,6). We analyzed associations between the presence of HEV antibodies and explanatory variables using Pearson χ2 test or Fisher exact test and further included variables with p<0.05 in the bivariate analysis (except habitat status) in a generalized estimating equation model.
We identified 69 (50.7%, 95% CI 42.3%–59.1%) of 136 cetaceans as harboring anti-HEV antibodies (Table 1; Figures 1, 2; Appendix Table 1). We confirmed antibodies against HEV-3 in 5 of the 7 ELISA-positive animals analyzed by Western blot analysis: a free-ranging striped dolphin, a free-ranging Cuvier’s beaked whale, a free-ranging Risso’s dolphin, and 2 captive bottlenose dolphins. We found none (0.0%; 95% CI 0.0%–1.2%) of the 302 animals analyzed to be positive for HEV RNA (Figure 2).
We noted seroprevalence to be significantly higher in free-ranging animals (44/72; 61.1%; 95% CI 49.9%–72.4%) than in those kept in captivity (25/64; 39.1%; 95% CI 27.1%–51.0%) (relative risk = 2.5, 95% CI 1.2%–4.9%; p = 0.008). We found seropositivity in adult free-ranging cetaceans (33/45; 73.3%) to be significantly higher than that in young animals (11/27; 40.7%; odds ratio 4.0, 95% CI 1.4–11.0; p = 0.006).
Our testing revealed seropositive animals in 5 of the 6 aquatic facilities sampled; within-zoo seroprevalence ranged from 27.8% in aquatic park D to 55.6% in aquatic park E (Table 2; Figure 2). Of the 30 longitudinally sampled animals, 21 remained seronegative, and 6 animals showed seropositivity at all samplings during the study period (Appendix Table 2). Two bottlenose dolphins seroconverted, 1 in 2013 and another in 2017. Seroreversions were detected in 2 animals (1 a dolphin that had shown seroconversion); 1 incident occurred 1 year after the first positive sampling, the other 5 years.
Our survey reveals high exposure to HEV in free-ranging and captive populations of cetaceans in Spain. The detection of HEV antibodies in Atlantic spotted, common, Risso’s, and striped dolphins, as well as in Cuvier’s beaked and killer whales, demonstrates an increase in the number of cetartiodactyls susceptible to this virus (2).
Ingestion of contaminated food is considered to be one of the main transmission routes of HEV in humans and has also been suggested for other mammal species, including dolphins (4). The seropositive species detected in our study feed on a wide variety of resources, including fish and cephalopods. The presence of HEV in these food resources has not yet been assessed, but the virus has been frequently detected in such other aquatic animals as sea urchins and bivalve shellfish in different areas of Europe (2,7), which provides evidence that HEV does abide in marine ecosystems. Of note, the virus is shed primarily in the feces of infected species, which can lead to viral contamination of the environment, and HEV has been shown to be highly resistant to even high concentrations of salt (8). Contaminated water has been considered a potential source of zoonotic HEV (9), because drinking tap water or water from private wells or nearby rivers has been suggested as a risk factor for acquiring HEV infection in humans (10). This hypothesis is supported by a study conducted in captive cetaceans all sharing the same tanks, which revealed the detection of seropositivity and active HEV infection (4).
The significantly higher seroprevalence we found in adult free-ranging animals compared with young animals likely reflects the increased cumulative exposure to HEV in these species. Our additional discovery of HEV antibodies in 4 free-ranging yearlings in 2011, 2019, and 2021 could suggest endemic circulation of HEV in cetaceans living in Spanish waters during the study period. Free-ranging cetaceans had a 2.5-times higher risk of being exposed to HEV than those kept in captivity, which might be explained by differences in diet or longer exposure to environmental contamination. Human- and swine-related HEV-3 strains have been detected in sewage and slurry in Spain (11) and in rivers and coastal waters in Italy (12). The high census of some susceptible domestic and wildlife species (13,14), combined with high coastal urbanization and insufficient control of urban sewage in some regions of our study area (15), might be contributing factors in the higher seropositivity we noted in free-ranging cetaceans. By contrast, cetaceans in zoological parks, including those analyzed in our study, live in large water tanks that are frequently decontaminated with ozone, ultraviolet radiation, brine, or chlorine, some of which deactivates HEV (9). Nonetheless, the high seroprevalence we observed in the 5 zoos with seropositive animals indicates a wide circulation of the virus in these more controlled environments.
The 2 seroconversions we noted in captive bottlenose dolphins support the hypothesis of HEV circulation in zoos during our study period. However, 4 of the longitudinally surveyed cetaceans remained seropositive at all samplings. This finding might be due to the long-lived persistence of anti-HEV antibodies in cetaceans, which is supported by the significantly higher seroprevalence we detected in older, free-ranging cetaceans. There is no known information about the long-term persistence of HEV antibodies in these species. Thus, possible loss of antibodies and reexposure in some of the persistently seropositive cetaceans during the study period cannot be ruled out, as evidenced by the seroreversions we observed in 2 bottlenose dolphins 1 and 5 years after the first seropositive sampling was detected.
In conclusion, the seropositivity noted in our study indicates widespread circulation of HEV in both free-ranging and captive cetacean populations in southwestern Europe. Additional molecular and serologic studies are warranted to determine the role of cetaceans in the epidemiology of HEV and to elucidate the sources of HEV infection, particularly in the free-ranging cetacean population.
Dr. Javier Caballero-Gómez is a postdoctoral researcher at the Clinical Virology and Zoonoses Group at the Maimonides Biomedical Research Institute of Cordoba and the Animal Health and Zoonosis Research Group (GISAZ) at the University of Cordoba. His research interests are focused on the epidemiology of hepeviruses and other zoonotic emerging diseases.
Acknowledgments
This study did not involve the purposeful killing of animals. Samples from live cetaceans were collected from serum banks or animals in health programs or undergoing routine medical check-ups, and those from dead individuals were collected by veterinarians and animal keepers following routine procedures in compliance with Ethical Principles in Animal Research. Ethics approval by an Institutional Animal Care and Use Committee was not therefore deemed necessary.
We gratefully acknowledge all veterinarians, animal trainers, and other professionals involved in the caring and sampling of the animals. We thank Zoo Madrid, Selwo Marina, Mundomar Benidorm, Oceanogràfic València, Aquopolis Costa Dorada, and Loro Parque for providing the valuable samples and also to Centro de Gestión del Medio Marino Andaluz (CEGMA), which belongs to the Consejería de Agricultura, Ganadería, Pesca y Desarrollo Sostenible de la Junta de Andalucía. We are grateful for the support by the German Centre of Infection Research (DZIF), TTU “Emerging infections.” We would like to thank Dörte Kaufmann and Ina Linzke for their support and Barbara Kubickova for providing recombinant HEV antigens.
This research was supported by CIBER Consorcio Centro de Investigación Biomédica en Red (CB 2021), Instituto de Salud Carlos III, Ministerio de Ciencia e Innovación and Unión Europea–NextGenerationEU. This work was also supported by the Ministerio de Sanidad (RD12/0017/0012) integrated in the Plan Nacional de I+D+I and cofinanced by the ISCIII-Subdirección General de Evaluación and the Fondo Europeo de Desarrollo Regional (FEDER), Fundación para la Investigación en Salud (FIS) del Instituto Carlos III (PI19/00864 and PI21/00793). J.C.-G. was supported by an FPU grant of the Spanish Ministry of Science, Innovation, and Universities (FPU17/01319). A.R.-J. is the recipient of a Miguel Servet Research Contract by the Spanish Ministry of Science, Innovation, and Universities (CP18/00111). The funders did not play any role in the design, conclusions, or interpretation of this study.
References
- Purdy MA, Drexler JF, Meng XJ, Norder H, Okamoto H, Van der Poel WHM, et al. ICTV virus taxonomy profile: Hepeviridae 2022. J Gen Virol. 2022;103:103. DOIPubMedGoogle Scholar
- Kenney SP. The current host range of hepatitis E viruses. Viruses. 2019;11:52. DOIPubMedGoogle Scholar
- Aspinall EJ, Couturier E, Faber M, Said B, Ijaz S, Tavoschi L, et al.; The Country Experts. Hepatitis E virus infection in Europe: surveillance and descriptive epidemiology of confirmed cases, 2005 to 2015. Euro Surveill. 2017;22:30561. DOIPubMedGoogle Scholar
- Montalvo Villalba MC, Cruz Martínez D, Ahmad I, Rodriguez Lay LA, Bello Corredor M, Guevara March C, et al. Hepatitis E virus in bottlenose dolphins Tursiops truncatus. Dis Aquat Organ. 2017;123:13–8. DOIPubMedGoogle Scholar
- Frías M, López-López P, Zafra I, Caballero-Gómez J, Machuca I, Camacho Á, et al. Development and clinical validation of a pangenotypic PCR-based assay for the detection and quantification of hepatitis E virus (Orthohepevirus A genus). J Clin Microbiol. 2021;59:e02075–20. DOIPubMedGoogle Scholar
- Johne R, Plenge-Bönig A, Hess M, Ulrich RG, Reetz J, Schielke A. Detection of a novel hepatitis E-like virus in faeces of wild rats using a nested broad-spectrum RT-PCR. J Gen Virol. 2010;91:750–8. DOIPubMedGoogle Scholar
- Santos-Ferreira N, Mesquita JR, Rivadulla E, Inácio ÂS, Martins da Costa P, Romalde JL, et al. Hepatitis E virus genotype 3 in echinoderms: First report of sea urchin (Paracentrotus lividus) contamination. Food Microbiol. 2020;89:
103415 . DOIPubMedGoogle Scholar - Wolff A, Günther T, Albert T, Johne R. Effect of sodium chloride, sodium nitrite and sodium nitrate on the infectivity of hepatitis E virus. Food Environ Virol. 2020;12:350–4. DOIPubMedGoogle Scholar
- Fenaux H, Chassaing M, Berger S, Gantzer C, Bertrand I, Schvoerer E. Transmission of hepatitis E virus by water: An issue still pending in industrialized countries. Water Res. 2019;151:144–57. DOIPubMedGoogle Scholar
- Mansuy JM, Gallian P, Dimeglio C, Saune K, Arnaud C, Pelletier B, et al. A nationwide survey of hepatitis E viral infection in French blood donors. Hepatology. 2016;63:1145–54. DOIPubMedGoogle Scholar
- Clemente-Casares P, Rodriguez-Manzano J, Girones R. Hepatitis E virus genotype 3 and sporadically also genotype 1 circulate in the population of Catalonia, Spain. J Water Health. 2009;7:664–73. DOIPubMedGoogle Scholar
- La Rosa G, Proroga YTR, De Medici D, Capuano F, Iaconelli M, Della Libera S, et al. First detection of hepatitis E virus in shellfish and in seawater from production areas in Southern Italy. Food Environ Virol. 2018;10:127–31. DOIPubMedGoogle Scholar
- Bosch J, Peris S, Fonseca C, Martinez M, De la Torre A, Iglesias I, et al. Distribution, abundance and density of the wild boar on the Iberian Peninsula, based on the CORINE program and hunting statistics. Folia Zool (Brno). 2012;61:138–51. DOIGoogle Scholar
- Ministerio de Agricultura. Pesca y alimentación. El sector de la carne de cerdo en cifras 2021. 2022 Spanish Report [cited 2022 Nov 15]. https://www.mapa.gob.es/es/agricultura/estadisticas/indicadoressectorporcino2021_tcm30-564427.pdf
- European Commission. Urban waste water: The Commission decided today to refer SPAIN to the Court of Justice of the European Union for breach of the Urban Waste Water Treatment Directive [cited 2022 Apr 6]. https://ec.europa.eu/commission/presscorner/detail/es/ip_22_1923
Figures
Tables
Cite This ArticleOriginal Publication Date: November 18, 2022
Table of Contents – Volume 28, Number 12—December 2022
| EID Search Options |
|---|
|
|
|
|
|
|
Please use the form below to submit correspondence to the authors or contact them at the following address:
Antonio Rivero-Juarez, Grupo de Virología Clínica y Zoonosis, Unidad de Enfermedades Infecciosas, Instituto Maimónides de Investigación Biomédica de Córdoba (IMIBIC), Hospital Universitario Reina Sofía, Universidad de Córdoba, 14004 Córdoba, Spain
Top