Volume 29, Number 10—October 2023
Dispatch
Pseudomonas aeruginosa High-Risk Sequence Type 463 Co-Producing KPC-2 and AFM-1 Carbapenemases, China, 2020–2022
Cite This Article
Citation for Media
Abstract
We report the clonal spread and evolution of high-risk Pseudomonas aeruginosa sequence type 463 co-producing KPC-2 and AFM-1 carbapenemases isolated from hospital patients in China during 2020–2022. Those strains pose a substantial public health threat and surveillance and stricter infection-control measures are essential to prevent further infections.
Carbapenemase-producing Pseudomonas aeruginosa poses a global threat to public health. Epidemics caused by this pathogen are associated with high-risk clones (e.g., sequence type [ST] 235, ST277, ST175, ST233 and ST111), particularly those clones producing metallo-β-lactamases; Verona integron-encoded metallo-β-lactamase and imipenemase are the most prevalent carbapenemase types (1,2). A Klebsiella pneumoniae carbapenemase (KPC)–producing clone, ST463, has emerged and become predominant in carbapenemase-producing P. aeruginosa populations in China (3). Three P. aeruginosa strains co-producing KPC and Alcaligenes faecalis metallo-β-lactamase (AFM) were reported in 2022 and attributed to ST463 (4,5). During 2020–2022, we observed the clonal spread of ST463 carbapenem-resistant P. aeruginosa (CRPA) co-producing KPC-2 and AFM-1 (KPC-2–AFM-1 CRPA) in a hospital in China, which caused infections with high mortality. We report on KPC-2–AFM-1 CRPA emergence and driving forces that caused dissemination.
During September 2020–June 2022, 192 nonduplicated CRPA isolates were collected from 192 patients admitted to a tertiary hospital in Zhejiang, China. Among those isolates, carbapenemase-producing Pseudomonas aeruginosa belonging to 10 different STs reached an overall prevalence of 41.1% (79/192) (Appendix Figure 1). We investigated KPC-2–AFM-1 CRPA strains isolated from 8 patients; 3 strains were colonizers and 5 were associated with infections (Table). The patients (6 men, 2 women) were 45–90 years of age, and all had complicated conditions and a history of intensive care unit admission. Antimicrobial drugs active against KPC-2–AFM-1 CRPA (colistin in monotherapy) were given to 4 patients; 5 infected patients eventually died; the remaining 3 patients, who only had bacterial colonization, were discharged (Table).
All 8 isolates were ST463, had identical resistance genes (Appendix Table 1) and type III secretion system genotype exoU+/exoS+, and differed by 5–30 single-nucleotide polymorphisms, indicating clonal dissemination (Appendix Figure 2). Each strain had 1 chromosome and a plasmid containing the β-lactamase gene blaKPC-2. Alignment of the 8 plasmids showed an identical backbone, but we noted major differences within the insertion region; the conserved core blaKPC-2 genetic platform insertion sequence (IS)Kpn27–blaKPC-2–ISKpn6 in plasmid p94 remained intact, whereas ISKpn6 and several genes immediately upstream (ISKpn6–open reading frame–klcA–open reading frame) were absent in the other 7 plasmids (Appendix Figure 3, panel A), forming a novel genetic context for blaKPC-2. All plasmids were designated as type I plasmids (6) but had a 16-kb deletion of a mobilization-related operon within the backbone (Appendix Figure 3, panel B), supporting their nontransferability, which was confirmed by conjugation assays.
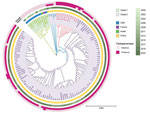
Using phylogenetic analysis of 125 ST463 genomes (68 from the National Center for Biotechnology Information Reference Sequence database [https://www.ncbi.nlm.nih.gov/refseq] and 57 from our collection), we found that those genomes were divided into 2 clades (Figure 1). Collection dates for strains in clade 1 were much earlier than those for clade 2. Clade 1 was primarily represented by clones from the United States, and clade 1 isolates did not harbor blaKPC or blaAFM. The larger clade 2 originated in China and most isolates in this clade carried blaKPC; we also detected blaAFM in clade 2. Independent evolution of ST463 clones in China might be correlated with sequential acquisition of blaKPC and blaAFM. Paired single-nucleotide polymorphism distances for clade 2 strains in a minimum spanning tree were mainly 0–60 (Appendix Figure 2), demonstrating a high degree of relatedness among those isolates and clonal transmission of KPC-producing P. aeruginosa ST463 in China. The 8 KPC-2–AFM-1 CRPAs clustered with 3 blaAFM-carrying strains and formed a separate subclade inside clade 2 that was surrounded by blaAFM-negative, KPC-producing P. aeruginosa. Therefore, we inferred that ST463 KPC-2–AFM-1 CRPA clones probably arose from ST463 KPC-producing P. aeruginosa.
We could not map the genome sequences of clade 1 strains to the reference plasmid p94 (0% coverage), indicating the absence of type I plasmids in ST463 clones outside of China. Using BLAST (https://blast.ncbi.nlm.nih.gov), we retrieved 40 homologous plasmids that had >50% coverage and >95% identity to p94; all were type I plasmids carried by 14 different P. aeruginosa STs (Appendix Figure 4), suggesting an extensive horizontal transfer of type I plasmids within a narrow host range. Plasmid phylogeny revealed that all blaKPC-2–encoding plasmids clustered together in an independent branch, and separation of that branch from closely related blaKPC-2–negative plasmids might be associated with acquisition of blaKPC-2 (Appendix Figure 5). The presence of multiple copies of IS26 in the blaKPC-2–adjacent region beyond the core platform indicated a critical role for IS26 elements in remodeling and resistance evolution of type I plasmids in the ST463 lineage (Appendix Figure 4) (7). We deduced an underlying evolutionary pathway: an ST463 P. aeruginosa progenitor initially acquired a highly transferable type I plasmid, which subsequently evolved into a resistance plasmid through IS26-mediated insertion events involving a blaKPC-2–carrying region, then by chromosomal integration of blaAFM-1 to form KPC-2–AFM-1 CRPA. To further verify this hypothesis, we conducted a genomic comparison between ZY94 and the 1755 strain, which carried neither blaKPC nor blaAFM but was phylogenetically closest to the KPC-2–AFM-1 CRPA cluster (Figure 1). The genome of 1755 was also composed of 1 chromosome and 1 plasmid (p1755). Plasmid p94 was highly homologous (coverage 81%, identity 99.76%) to p1755 and might have evolved from p1755 via IS26-mediated intermolecular transposition of a blaKPC-2–carrying translocatable unit that targeted an existing copy of IS26 (Appendix Figure 6, panel A). Comparisons between their chromosomes identified a redundant blaAFM-1–containing, multidrug-resistance fragment bracketed by IS5564 in the same orientation, which was flanked by two 6-bp (GCTAGA) target site duplications (Appendix Figure 6, panel B), indicating a site-specific insertion event.
All 8 isolates were resistant to β-lactams and β-lactam/β-lactamase inhibitors, including carbapenems, ceftazidime/avibactam, fluoroquinolones, and gentamicin, and were only susceptible to amikacin and intermediately susceptible to colistin. We observed synergistic inhibitory effects against all 8 strains when we used combinations of ceftazidime/avibactam and aztreonam (Appendix Table 1). Compared with P. aeruginosa strain ATCC27853, all strains showed 4-fold higher MICs for chlorhexidine, a commonly used medical disinfectant, suggesting chlorhexidine tolerance (Appendix Table 1) (8).
All 8 strains exhibited small colony variant phenotypes together with strong biofilm formation capacities (Appendix Table 2). To assess their stability, desiccation resilience, and virulence, we selected strains ZY94 (infecting strain) and ZY1214 (colonizing strain) for further experiments. After we subcultured in antimicrobial drug–free Luria-Bertani broth for 10 days, ZY94 and ZY1214 retained stable small colony variant phenotypes; their blaKPC-2–carrying plasmid and blaAFM-1 gene were both stably inherited (stability was 93% for ZY94 and 95% for ZY1214). Both strains survived for 8 days on a dry polystyrene surface at higher rates than strains PAO1 and NDTH6412 (Figure 2, panel A). Furthermore, in mouse intraperitoneal challenge models (Appendix), ZY94 and ZY1214 strains had significantly higher (≈50%) lethality than ATCC9027 (p<0.05), but were less virulent than the PA14 strain (Figure 2, panel B).
We documented the persistent clonal spread of ST463 KPC-2–AFM-1 CRPAs in a hospital in China and provided insights into a potential evolutionary pathway for KPC-2–AFM-1 CRPA formation. Extensive drug resistance, disinfectant and desiccation resilience, strong biofilm formation, and high stability constituted strategies for those strains to defend against host and clinical challenges, thereby driving persistent transmission of this high-risk clone. Infections caused by such pathogens might lead to high death rates, especially in immunocompromised or critically ill patients, highlighting the urgent need for effective infection prevention and control policies. ST463 KPC-2–AFM-1 CRPA strains pose a substantial public health threat because of their extensive drug resistance, considerable pathogenicity, and ability to persist in the environment. Targeted surveillance and stricter infection-control measures are essential to prevent further infection outbreaks.
Dr. Zhang is a resident doctor at the First Affiliated Hospital of Zhejiang University School of Medicine, in Hangzhou, Zhejiang, China. Her primary research interests are antimicrobial drug resistance and Pseudomonas aeruginosa epidemiology.
Acknowledgments
This study was supported by the Natural Science Foundation of Zhejiang Province (grant no. TGY23H010005) and Scientific Research Fund of Zhejiang Provincial Education Department (grant no. Y202146838).
This study was approved by the institutional review board of the First Affiliated Hospital of Zhejiang University School of Medicine in China (approval no. IIT20210120B).
References
- Del Barrio-Tofiño E, López-Causapé C, Oliver A. Pseudomonas aeruginosa epidemic high-risk clones and their association with horizontally-acquired β-lactamases: 2020 update. Int J Antimicrob Agents. 2020;56:
106196 . DOIPubMedGoogle Scholar - Reyes J, Komarow L, Chen L, Ge L, Hanson BM, Cober E, et al.; Antibacterial Resistance Leadership Group and Multi-Drug Resistant Organism Network Investigators. Global epidemiology and clinical outcomes of carbapenem-resistant Pseudomonas aeruginosa and associated carbapenemases (POP): a prospective cohort study. Lancet Microbe. 2023;4:e159–70. DOIPubMedGoogle Scholar
- Hu Y, Liu C, Wang Q, Zeng Y, Sun Q, Shu L, et al. Emergence and expansion of a carbapenem-resistant Pseudomonas aeruginosa clone are associated with plasmid-borne bla KPC-2 and virulence-related genes. mSystems. 2021;6:e00154–21. DOIPubMedGoogle Scholar
- Li Y, Zhu Y, Zhou W, Chen Z, Moran RA, Ke H, et al. Alcaligenes faecalis metallo-β-lactamase in extensively drug-resistant Pseudomonas aeruginosa isolates. Clin Microbiol Infect. 2022;28:880.e1–8. DOIPubMedGoogle Scholar
- Zhang P, Wang J, Shi W, Wang N, Jiang Y, Chen H, et al. In vivo acquisition of blaKPC-2 with low biological cost in blaAFM-1-harboring ST463 hypervirulent Pseudomonas aeruginosa from a patient with hematologic malignancy. J Glob Antimicrob Resist. 2022;31:189–95. DOIPubMedGoogle Scholar
- Zhu Y, Chen J, Shen H, Chen Z, Yang QW, Zhu J, et al. Emergence of ceftazidime- and avibactam-resistant Klebsiella pneumoniae carbapenemase-producing Pseudomonas aeruginosa in China. mSystems. 2021;6:
e0078721 . DOIPubMedGoogle Scholar - He S, Hickman AB, Varani AM, Siguier P, Chandler M, Dekker JP, et al. Insertion sequence IS26 reorganizes plasmids in clinically isolated multidrug-resistant bacteria by replicative transposition. MBio. 2015;6:
e00762 . DOIPubMedGoogle Scholar - Sahin S, Mogulkoc MN, Kürekci C. Disinfectant and heavy metal resistance profiles in extended spectrum β-lactamase (ESBL) producing Escherichia coli isolates from chicken meat samples. Int J Food Microbiol. 2022;377:
109831 . DOIPubMedGoogle Scholar
Figures
Table
Cite This ArticleOriginal Publication Date: September 15, 2023
1These authors contributed equally to this article.
2These senior authors contributed equally to this article.
Table of Contents – Volume 29, Number 10—October 2023
| EID Search Options |
|---|
|
|
|
|
|
|

Please use the form below to submit correspondence to the authors or contact them at the following address:
Tingting Qu, The First Affiliated Hospital of Zhejiang University School of Medicine, No.79 Qingchun Rd, Hangzhou 310000, China
Top