Volume 30, Number 6—June 2024
Synopsis
Deciphering Unexpected Vascular Locations of Scedosporium spp. and Lomentospora prolificans Fungal Infections, France
Cite This Article
Citation for Media
Abstract
Scedosporium spp. and Lomentospora prolificans are emerging non-Aspergillus filamentous fungi. The Scedosporiosis/lomentosporiosis Observational Study we previously conducted reported frequent fungal vascular involvement, including aortitis and peripheral arteritis. For this article, we reviewed 7 cases of Scedosporium spp. and L. prolificans arteritis from the Scedosporiosis/lomentosporiosis Observational Study and 13 cases from published literature. Underlying immunosuppression was reported in 70% (14/20) of case-patients, mainly those who had solid organ transplants (10/14). Osteoarticular localization of infection was observed in 50% (10/20) of cases; infections were frequently (7/10) contiguous with vascular infection sites. Scedosporium spp./Lomentospora prolificans infections were diagnosed in 9 of 20 patients ≈3 months after completing treatment for nonvascular scedosporiosis/lomentosporiosis. Aneurysms were found in 8/11 aortitis and 6/10 peripheral arteritis cases. Invasive fungal disease–related deaths were high (12/18 [67%]). The vascular tropism of Scedosporium spp. and L. prolificans indicates vascular imaging, such as computed tomography angiography, is needed to manage infections, especially for osteoarticular locations.
Scedosporium spp. and Lomentospora prolificans are non-Aspergillus filamentous fungi causing increasingly recognized invasive fungal disease (IFD) in both immunocompromised and immunocompetent patients (1,2). Scedosporium spp. comprise S. apiospermum complex species, which includes S. apiospermum sensu stricto and S. boydii; L. prolificans was previously known as S. prolificans (3). In immunocompetent patients, localized infections have been described, such as mycetomas, osteoarticular infections (4), or central nervous system (CNS) infections after near-drownings. In immunocompromised hosts, scedosporiosis and lomentosporiosis mainly affect the lungs and CNS or are disseminated (5–11). We previously conducted the Scedosporiosis/lomentosporiosis Observational Study (SOS) and reported non-CNS vascular involvement (aorta or peripheral arteries excluding CNS arteries) in 24% of disseminated infections (12). Although CNS vascular involvement during invasive scedosporiosis has been described, a scedosporiosis/lomentosporiosis case series of non-CNS vascular infections has not been reported (5–11,13–17). We describe clinical, imaging, and detailed histopathologic characteristics of all patients with non-CNS vascular scedosporiosis/lomentosporiosis infections identified in the SOS and review cases reported in published literature.
Ethics Approval
The research was approved by the Institut Pasteur Internal Review Board (approval no. 2009–34/IRB) and by the Commission Nationale de l’Informatique et des Libertés in accordance with the laws of France. All clinical data were recorded anonymously.
Study Design
The SOS included all retrospective cases of proven and probable invasive scedosporiosis/lomentosporiosis (IS) characterized at the National Reference Center for Invasive Mycoses and Antifungals, France, during January 2005–March 2017 (12). We performed a medical records review of all patients in the SOS who had non-CNS vascular infections (aortitis or peripheral arteritis) caused by Scedosporium spp. or L. prolificans. In addition, we performed an electronic literature search for case reports in PubMed on October 10, 2021, by using the following search filter: (Scedospori*[Title/Abstract] OR Pseudallescheri*[Title/Abstract] OR Lomentospori*[Title/Abstract]) AND (invasive[Title/Abstract] OR disseminated[Title/Abstract] OR infection[Title/Abstract]) AND (case[Title/Abstract] OR patient[Title/Abstract] OR report[Title/Abstract]). We selected all articles in English, French, or Spanish that reported proven or probable non-CNS vascular infections caused by Scedosporium spp., including Pseudallescheria spp., or L. prolificans, including S. prolificans and S. inflatum. We checked reference lists of selected articles for other relevant studies. We only included published case reports or case series with detailed documentation of clinical history, diagnostic methods, treatment, and outcomes. Data from 1 SOS case (patient 5) had been published previously, and we excluded this case from the literature review.
For each case, we reported demographic conditions, underlying diseases/conditions, vascular impairment sites, signs and symptoms, other infected sites, microbiologic results, antifungal treatments, surgical therapy, and clinical outcomes by using a standard anonymous case report form. Each SOS case was reviewed by 4 investigators (C.V., J.E., F.L., and D.B.); each literature review case was reviewed by 2 investigators (C.V. and D.B.).
Definitions
We defined non-CNS vascular infection as either imaging of structural abnormalities of the arterial wall suggestive of aortitis/arteritis (18) associated with a concomitant IS diagnosis (Appendix), a positive culture from an intraoperative arterial wall sample, or anatomopathologic evidence of arterial wall fungal infection with a concomitant IS diagnosis. We excluded cases of arterial thrombosis without evidence of parietal impairment. We defined proven and probable IS according to the 2019 European Organization for Research and Treatment of Cancer/Mycoses Study Group criteria (19), modified by including trauma and near-drowning as risk factors.
We considered 6 main underlying conditions for IFD, but only 1 condition was assigned to each patient as follows: malignancies; solid organ transplantation; systemic inflammatory disease; contamination by traumatic penetration, injury, surgery, or near-drowning (trauma/inoculation); other, for miscellaneous underlying medical conditions, including neutropenia, chronic renal or respiratory insufficiency, chronic respiratory disease, diabetes mellitus, HIV infection, or corticosteroids administration; and no risk factor in the absence of all previous factors.
We considered infections to be disseminated when they involved >2 noncontiguous sites or were associated with fungemia. We defined breakthrough infections as those occurring in patients who received antifungal therapy for >7 days within 30 days before IS diagnosis and defined prior colonization as a Scedosporium/Lomentospora culture within a nonsterile clinical sample without signs of infection before IS diagnosis. We defined follow-up as the period from IS diagnosis to last patient contact or death if vascular infection was diagnosed at the time of IS diagnosis or within 3 months of diagnosis; otherwise, we defined it as the period from vascular infection diagnosis to last patient contact or death. We considered IFD-related death as death supposedly caused by IFD. We defined radical surgery as complete resection of the vascular infection site, including vascular prothesis removal, whether or not it was associated with vascular reconstruction. For analyses, we described quantitative variables as medians with interquartile ranges and categorical variables as numbers and percentages.
SOS Case-Patients
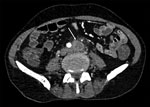
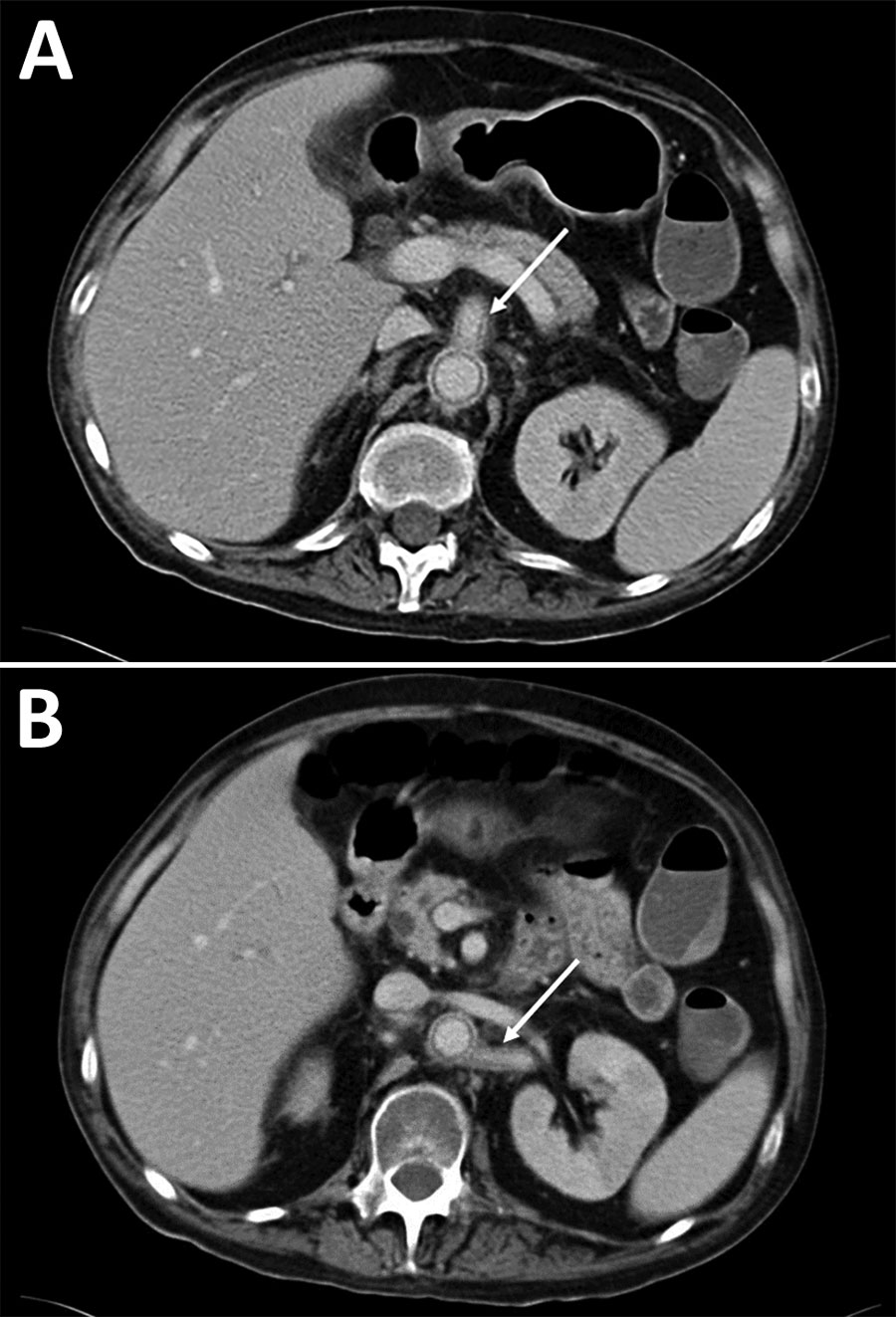
Case-patient 1 was a 52-year-old man who had liver and kidney transplants because of alcoholic cirrhosis and IgA nephropathy. He had dermohypodermitis of the left lower limb caused by S. apiospermum and was treated with voriconazole for 18 months. Despite initial clinical success, S. apiospermum oligoarthritis developed 3 months after the patient stopped antifungal therapy; infection was documented in cultures of left knee joint liquid. He received a combination of voriconazole, caspofungin, and terbinafine and reduction of his immunosuppressive regimen. No evidence of endocarditis was found on an echocardiogram. Blood culture results were negative. Because fever persisted during treatment, positron emission tomography (PET)/computed tomography (CT) and abdominal/pelvic CT were performed, which identified multiple osteoarticular localizations (ankles, knees, left sacroiliac joint, left hip, T8 spondylitis, L5–S1 spondylodiscitis), left primitive iliac arteritis (thrombosis with perivascular and soft tissue contrast) (Figure 1) and abdominal bifurcation aortitis (perivascular and soft tissue contrast associated with focal hypermetabolism). The patient died 2 months later from multiorgan failure accompanied by acute left lower limb ischemia and possible mesenteric ischemia.
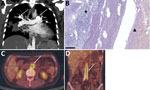
Case-patient 2 was a 31-year-old woman recovering from a severe road accident in muddy water. She was hospitalized 2 months after the accident for pneumonia and bilateral pulmonary embolisms. She was treated empirically with antimicrobial drugs and anticoagulation therapy, which was stopped a few days later because of massive hemoptysis. She experienced persistent fever and epileptic seizures; an echocardiogram and CT scan revealed tricuspid endocarditis associated with cerebral abscess and bilateral aneurysms of pulmonary arteries rapidly growing and partially thrombosed (Figure 2, panel A). At that time, S. apiospermum was identified from blood cultures. She received long-term therapy with voriconazole and caspofungin, which was successful. However, chronic pulmonary arterial hypertension developed, requiring cardiopulmonary transplantation while she was undergoing antifungal treatment for >2 years. She died the day of transplantation because of a pulmonary hilum twist. Postmortem examination showed highly altered pulmonary arteries with major intimal fibrosis and a thrombus containing multiple fungal septate hyphae (Figure 2, panel B).
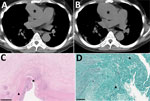
Case-patient 3 was a 32-year-old man who had macrophage activation syndrome, acute renal failure requiring hemodialysis, and multiple necrotic cutaneous lesions after acute cocaine intoxication. Febrile acute shoulder arthritis and sternal osteochondritis developed for which he underwent surgical debridement; S. apiospermum was identified in perioperative shoulder samples, and L. prolificans was identified in sternum samples. Thoracic-abdominal-pelvic CT and PET/CT scans revealed pseudonodular pulmonary lesions, abdominal aortitis and primitive iliac arteritis with perivascular contrasts, arterial wall thickenings, and intense vascular hypermetabolism (Figure 2, panels C, D). He was successfully treated with voriconazole, terbinafine, and miltefosine for 3 years, along with γ-interferon in the absence of initial improvement. Follow-up imaging performed 2.5 years after treatment initiation found a clear reduction of periaortic inflammatory thickening and vascular hypermetabolism. Antifungal treatment was stopped after 3 years without any clinical sign of relapse 1 year after interruption.
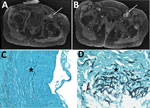
Case-patient 4 was a 51-year-old woman who had focal segment glomerulosclerosis requiring a kidney transplant, which was complicated by acute graft rejection. Closing surgery of her arteriovenous femoral fistula was complicated by a Pseudomonas aeruginosa infection at the surgical site, which was treated by a femoral bypass graft and antimicrobial drugs. Postoperatively, a femoral graft thrombosis and perivascular collection developed and was treated by graft removal, and a 3-month voriconazole course was begun because of positive results for S. apiospermum from a superficial swab culture. Eight months later, new onset of fever and left limb pain led to discovery of 2 mycotic aneurysms in the left common femoral artery (Figure 3, panels A, B), multiple left thigh abscesses, and left navicular osteitis. The mycotic aneurysms were surgically removed, and vascular reconstruction by arterial allograft bypass was performed. S. apiospermum was identified in cultured perioperative samples; histologic analysis revealed vascular invasion, particularly in the tunica media (Figure 3, panels C, D). The patient was successfully treated with voriconazole and caspofungin for 18 months; PET/CT showed no signs of relapse 1 year after stopping antifungal therapy.
Case-patient 5 was a 44-year-old man who had chronic myeloid leukemia and underwent allogeneic hematopoietic stem cell transplant. While he was hospitalized for chronic graft versus host disease, a febrile gingival abscess developed and was treated by dental avulsion. Abscess and blood culture samples tested positive for L. prolificans; voriconazole and terbinafine treatments were initiated. Although fungemia persisted during treatment, he complained about abdominal pain; CT revealed abdominal aortitis spreading to the superior mesenteric and left renal arteries manifested by vascular thickenings and perivascular contrast (Figure 4, panels A, B). Within the following month, he had intense abdominal pain and died, likely from mesenteric ischemia.
Case-patient 6 was a 56-year-old man who had diabetes and a lung transplant because of idiopathic chronic pulmonary fibrosis. A thoracotomy scar abscess developed rapidly after his transplantation surgery and was treated by surgical abscess drainage. S. apiospermum was documented in a perioperative specimen; therefore, he was treated with voriconazole. Four months later, while still undergoing antifungal treatment, he complained about a nonfebrile thoracic pain; CT revealed a rapidly growing ascending thoracic aortic aneurysm (Figure 5, panels A, B). Surgical aneurysm resection and prosthetic aortic replacement were performed, and he was treated with posaconazole and caspofungin. His perioperative tissue samples confirmed vascular fungal invasion (Figure 5, panels C, D), and cultures tested positive for S. apiospermum. After an initial clinical improvement, he died 3 months later; S. apiospermum fungemia had developed along with endocarditis complicated by massive ischemic stroke and mycotic aneurysm of the aortic prosthesis anastomosis (Appendix Figure 1). At the time of his death, he also had mycotic intrahepatic and splenic aneurysms.
Case-patient 7 was a 43-year-old woman receiving first-line chemotherapy for acute myeloid leukemia. Four days after chemotherapy initiation, febrile nodular cutaneous lesions developed that persisted despite treatment with wide-spectrum antimicrobial drugs and intravenous liposomal amphotericin B. A blood culture tested positive for L. prolificans, and antifungal therapy was switched to voriconazole. She died a few hours later because of refractory sepsis and acute cardiac failure. Postmortem examination revealed massive multiorgan filamentous invasion within blood vessel walls and lumens, particularly in the abdominal aorta, and within the myocardium.
Cohort of SOS and Published Cases
We classified the 7 non-CNS vascular infection cases identified from the SOS as 1 aortitis, 2 peripheral arteritis, and 4 mixed aortitis and peripheral arteritis cases. We identified 26 articles from the published literature search as potential reports of non-CNS vascular scedosporiosis/lomentosporiosis (Appendix Figure 2). We excluded 13 reports, 1 about CNS mycotic aneurysms (n = 11), 1 about ventricular assist device thrombosis (n = 1), and 1 without available full text (n = 1). Thirteen case-reports from the literature search, including 9 aortitis and 4 peripheral arteritis cases, were eligible for analysis (20–32).
All 20 cases were proven IS (Appendix Table). Vascular infection was confirmed by either histology or vascular sample cultures in 15 cases. For the 5 remaining cases, vascular samples were not available. For those, diagnoses were made on the basis of radiologic imaging compatible with vascular infection and proven IS at another site. All mycologic documentations were obtained from cultures of normally sterile sites.
Host Factors/Underlying Diseases
All but 1 patient had >1 risk factor for IS (Table 1). The main underlying condition was solid organ transplant (10/20 [50%]), including lung (n = 5), kidney (n = 3), heart (n = 1), liver/kidney (n = 1) transplants, followed by hematologic malignancy (3/20 [15%]) and solid cancer (1/20 [5%]). IS occurred with a median delay of 165 (interquartile range 25.8–271.3) days after transplantation. Five (25%) cases were associated with trauma or near-drowning, 3 (15%) cases were breakthrough infections, and 2 (10%) patients had prior Scedosporium spp. or L. prolificans colonization.
Mycologic and Clinical Manifestations
S. apiospermum complex species were identified in 12 (60%) cases, and L. prolificans was identified in 7 (35%) cases. Co-infection with S. apiospermum and L. prolificans occurred in 1 patient. Fungemia was reported in 8 (44%) cases, more frequently among L. prolificans infections (4/5 [80%]) than S. apiospermum complex infections (4/12 [33%]). Blood samples were not cultured for 2 patients with L. prolificans infection. Disseminated infections occurred in 15 (75%) cases, among which 13 had >2 noncontiguous infectious sites.
IS manifested as fever in 9/15 (60%) cases (Table 2). Patients with aortitis frequently (7/14 [50%]) had thoracic or abdominal pain. Vascular impairment was not observed at the time of IS diagnosis in 10/20 (50%) cases. However, impairment was frequently diagnosed months or years after completion of antifungal treatment for nonvascular scedosporiosis/lomentosporiosis (9/20 [45%]) but also while patients were still undergoing antifungal treatment (1/20 [5%]). The median delay between completion of antifungal treatment and diagnosis was 3 (interquartile range 1–13) months (4 years maximum). Moreover, vascular impairment was associated with >1 other localization in 18/20 (90%) cases, mostly in osteoarticular (10/20 [50%]) and pulmonary (9/20 [45%]) locations; 5/10 (50%) osteoarticular localizations were spondylodiscitis. Most (7/10 [70%]) osteoarticular localizations were contiguous with the vascular infection site, which included 3 of 5 spondylodiscitis (2 abdominal and 1 thoracic aortitis) and 4 of 5 peripheral osteoarticular localizations (1 femoral arteritis and hip arthritis, 1 iliac arteritis and sacroiliac arthritis, 1 sternal osteomyelitis and thoracic aortitis, and 1 subclavian arteritis and sternoclavicular arthritis).
Vascular Impairment Type
Aortitis affected the abdominal aorta (8/13 [62%]) more frequently than the thoracic aorta (5/13 [39%]) (Table 2). The location of aortitis was not mentioned for 1 case. Aortitis was mostly identified by the presence of an aneurysmal lesion (8/11 [73%]). Indeed, aneurysmal lesions were noted for all thoracic aortitis and 3 of 6 abdominal aortitis cases. Abdominal aortitis with arterial wall thickening without aneurysms were described for SOS case-patients 1, 3, and 5. The type of imaging anomalies was unknown for 3 patients who had postmortem aortitis diagnoses.
Peripheral arteritis localizations were diverse, affecting the iliac or femoral arteries (4/10 [40%]) and other arteries, such as hepatic (n = 2), pulmonary (n = 1), subclavian (n = 1), renal and mesenteric (n = 1), or arteriovenous fistula (n = 1) arteries. Peripheral arteritis was mainly revealed by the presence of aneurysmal lesions (6/10 [60%]); septic thrombosis was described in 3 cases.
PET/CT consistently showed an elective hypermetabolism of the vascular wall in 3 aneurysmal aortitis, 2 nonaneurysmal aortitis, and 1 peripheral mycotic aneurysm cases. All positive PET/CT scans were associated with concordant CT or magnetic resonance imaging angiography. PET/CT follow-up was available for 3 of 6 patients, and a clear regression of hypermetabolism during IS treatment was reported in all 3 cases, including 1 normalization at the end of treatment.
Histopathologic Lesions
Histopathologic analyses were available for 11/20 patients (8 perioperative vascular samples and 3 autopsies) and showed an invasion of the arterial wall by fungal septate hyphae in 10 of 11 samples. Arterial wall necrosis was reported in 6 cases and endoluminal thrombi caused by fungi were reported in 4 cases. Only 1 case showed an arterial granuloma lesion, and 2 cases had arterial wall abscesses. Although rarely specified, fungal invasion affected the tunica media (n = 3) or tunica adventitia (n = 3).
Treatment and Outcomes
After microbiologic identification, voriconazole was the first-line antifungal drug prescribed to all patients (Table 3). A combination of 2 antifungal drugs was frequently (10/16 [63%]) used, mostly voriconazole combined with terbinafine or caspofungin. Four patients did not receive antifungal treatment because the IS diagnosis was made postmortem. Radical surgery was performed in 8 (50%) of 16 patients with an antemortem IS diagnosis, which consisted of 3 biologic vascular reconstructions (2 arterial allografts and 1 arterial homograft), 3 prosthetic vascular reconstructions (Dacron grafts in all cases), and 2 cases without vascular reconstruction. Signs of infection relapse at the vascular sites were reported in the 3 prosthetic vascular reconstruction cases (pseudoaneurysm appearance or prosthetic graft thrombosis).
The IFD-related death rate was 67% (12/18) and 100% (7/7) when fungemia was present. Three IFD-related deaths were attributed to a direct complication of the vascular infection (mesenteric ischemia complicating abdominal aortitis in all cases), but the cause of death was missing for 4 patients. The death rate was higher (11/14 [79%]) for patients with aortitis (whether or not associated with peripheral arteritis) than isolated peripheral arteritis (1/4 [25%]). Patients treated with only antifungal drugs had a higher (7/9 [78%]) death rate than those who underwent radical surgery combined with antifungal drug therapy ( 2/6 [33%]).
We describe non-CNS vascular infections caused by Scedosporium spp. and L. prolificans, highlighting an underreported localization of clinical significance that affects the management of such infections. Solid organ transplant was the main host risk factor; however, several cases occurred in immunocompetent patients, particularly those who had cutaneous trauma. Vascular invasion was almost always associated with other infectious localizations, such as contiguous osteoarticular localizations. The main radiologic manifestation of infection was a vascular aneurysm, which was observed in all thoracic aortitis and most peripheral arteritis cases. Isolated vascular wall thickening was frequently described for abdominal aortitis and contiguous aortic branches, constituting overlap between abdominal aortitis and iliofemoral arteritis.
The SOS found that 24% of disseminated scedosporiosis/lomentosporiosis cases involved vascular infections (5–11,13–17). No non-CNS vascular infections were reported among 273 IS cases according to the Fungiscope registry (5), which gathers rare IFD data from countries around the world. Only 2 cases of mycotic aneurysms were reported among 80 scedosporiosis/lomentosporiosis cases in transplant patients, of which 40 were disseminated (33). One possible explanation for this underreporting might be that vascular impairment is rarely at the forefront of IS diagnostic considerations and might not be systematically tracked by imaging checkups or might be insufficiently reported in disseminated cases. All cases from published literature were reported since 2000, which could be partly explained by recent improvements in imaging access. In addition, vascular impairment was diagnosed in many (45%) cases after patients completed antifungal treatment for another IS localization, suggesting that undiagnosed vascular infections might lead to relapse because of insufficient treatment. We found that 50% of IS cases were associated with osteoarticular localization, whereas that localization was usually described in only 18%–26% IS and 14%–17% of disseminated scedosporiosis/lomentosporiosis cases (5,12,17). This finding suggests a potential association between vascular and osteoarticular localizations, especially because most of those were contiguous. In particular, we highlighted a frequent association of peripheral arteritis with aortitis resulting from spondylodiscitis and peripheral osteoarthritis. Finally, we reported high death rates, consistent with previous estimations for overall IS (5,7).
We focused on non-CNS vascular infections because cerebral mycotic aneurysms caused by Scedosporium spp. have already been specifically addressed for near-drowning situations (34). The ability of molds to invade vessels is well known; angioinvasion is a key pathogenic characteristic of invasive aspergillosis and mucormycosis. Several reports have described cerebral and pulmonary mycotic aneurysms located contiguously or distant to invasive aspergillosis or mucormycosis (35–39). However, few studies have reported thoracic aortitis caused by Aspergillus spp., which are frequently associated with endocarditis (40–42).
The strength of this work is that it confirms arterial wall invasion by Scedosporium or Lomentospora hyphae; histopathologic analyses were available for most cases. Nevertheless, IFD reporting is voluntary in France, and the National Reference Center might not have gathered data for all IFD cases in France during the study period. Moreover, we could not provide microbiologic identification at the species level for S. apiospermum complex species because genotypic analysis was conducted in only 4 of 13 cases reported in published literature, and several diagnoses were made before the last taxonomy classification update. We could not analyze the median duration of antifungal therapy because all but 3 patients were still undergoing treatment at last contact. Also, anticoagulant and antiplatelet drug management could not be assessed because of missing data. We were unable to evaluate the effects of vascular impairment on scedosporiosis/lomentosporiosis prognosis because assessing the exact circumstances of death was difficult in many cases; however, >3 of the 12 IFD-related deaths seemed to be related to a vascular complication. Finally, we observed that patients who underwent radical surgery seemed to have better prognoses. However, the retrospective study design, small number of cases, and patient population heterogeneity do not permit assessment of the best curative treatment, leading us to interpret the results for radical surgery with caution.
In conclusion, arterial wall infection is observed in 24% of patients with disseminated Scedosporium/Lomentospora infections and is mainly responsible for arterial aneurysms either by contiguous contamination or systemic dissemination. Our case series and literature review suggest that Scedosporium spp. and L. prolificans arterial infections might be latent for months after IS diagnosis and treatment, and ad hoc vascular imaging is justified in cases of osteoarticular localization or dissemination. Systematic screening for vascular involvement of Scedosporium/Lomentospora infections, including aortitis and peripheral arteritis, is needed to improve therapeutic strategies and prognosis.
Dr. Vignals is an infectious diseases doctor in the Department of Infectious and Tropical Diseases, Bordeaux University Hospital in France. Her research interests focus on infections occurring in immunocompromised hosts, especially fungal infections, and epidemiology.
References
- Ramirez-Garcia A, Pellon A, Rementeria A, Buldain I, Barreto-Bergter E, Rollin-Pinheiro R, et al. Scedosporium and Lomentospora: an updated overview of underrated opportunists. Med Mycol. 2018;56(suppl_1):102–25. DOIPubMedGoogle Scholar
- Miceli MH, Lee SA. Emerging moulds: epidemiological trends and antifungal resistance. Mycoses. 2011;54:e666–78. DOIPubMedGoogle Scholar
- Lackner M, de Hoog GS, Yang L, Ferreira Moreno L, Ahmed SA, Andreas F, et al. Proposed nomenclature for Pseudallescheria, Scedosporium and related genera. Fungal Divers. 2014;67:1–10. DOIGoogle Scholar
- Blez D, Bronnimann D, Rammaert B, Zeller V, Delhaes L, Hustache L, et al. Invasive bone and joint infections from the French Scedosporiosis/lomentosporiosis Observational Study (SOS) cohort: no mortality with long-term antifungal treatment and surgery. Med Mycol. 2023;61:myad023.
- Seidel D, Meißner A, Lackner M, Piepenbrock E, Salmanton-García J, Stecher M, et al. Prognostic factors in 264 adults with invasive Scedosporium spp. and Lomentospora prolificans infection reported in the literature and FungiScope®. Crit Rev Microbiol. 2019;45:1–21. DOIPubMedGoogle Scholar
- Jenks JD, Seidel D, Cornely OA, Chen S, van Hal S, Kauffman C, et al. Clinical characteristics and outcomes of invasive Lomentospora prolificans infections: Analysis of patients in the FungiScope® registry. Mycoses. 2020;63:437–42. DOIPubMedGoogle Scholar
- Álvarez-Uría A, Guinea JV, Escribano P, Gómez-Castellá J, Valerio M, Galar A, et al. Invasive Scedosporium and Lomentosora infections in the era of antifungal prophylaxis: A 20-year experience from a single centre in Spain. Mycoses. 2020;63:1195–202. DOIPubMedGoogle Scholar
- Lamaris GA, Chamilos G, Lewis RE, Safdar A, Raad II, Kontoyiannis DP. Scedosporium infection in a tertiary care cancer center: a review of 25 cases from 1989-2006. Clin Infect Dis. 2006;43:1580–4. DOIPubMedGoogle Scholar
- Troke P, Aguirrebengoa K, Arteaga C, Ellis D, Heath CH, Lutsar I, et al.; Global Scedosporium Study Group. Treatment of scedosporiosis with voriconazole: clinical experience with 107 patients. Antimicrob Agents Chemother. 2008;52:1743–50. DOIPubMedGoogle Scholar
- Idigoras P, Pérez-Trallero E, Piñeiro L, Larruskain J, López-Lopategui MC, Rodríguez N, et al. Disseminated infection and colonization by Scedosporium prolificans: a review of 18 cases, 1990-1999. Clin Infect Dis. 2001;32:E158–65. DOIPubMedGoogle Scholar
- Heath CH, Slavin MA, Sorrell TC, Handke R, Harun A, Phillips M, et al.; Australian Scedosporium Study Group. Population-based surveillance for scedosporiosis in Australia: epidemiology, disease manifestations and emergence of Scedosporium aurantiacum infection. Clin Microbiol Infect. 2009;15:689–93. DOIPubMedGoogle Scholar
- Bronnimann D, Garcia-Hermoso D, Dromer F, Lanternier F, Maulin L, Leprince Y, et al.; French Mycoses Study Group; Characterization of the isolates at the NRCMA. Scedosporiosis/lomentosporiosis observational study (SOS): Clinical significance of Scedosporium species identification. Med Mycol. 2021;59:486–97. DOIPubMedGoogle Scholar
- Berenguer J, Rodríguez-Tudela JL, Richard C, Alvarez M, Sanz MA, Gaztelurrutia L, et al.; Scedosporium Prolificans Spanish Study Group. Deep infections caused by Scedosporium prolificans. A report on 16 cases in Spain and a review of the literature. Medicine (Baltimore). 1997;76:256–65. DOIPubMedGoogle Scholar
- Tintelnot K, Just-Nübling G, Horré R, Graf B, Sobottka I, Seibold M, et al. A review of German Scedosporium prolificans cases from 1993 to 2007. Med Mycol. 2009;47:351–8. DOIPubMedGoogle Scholar
- Seidel D, Hassler A, Salmanton-García J, Koehler P, Mellinghoff SC, Carlesse F, et al. Invasive Scedosporium spp. and Lomentospora prolificans infections in pediatric patients: Analysis of 55 cases from FungiScope® and the literature. Int J Infect Dis. 2020;92:114–22. DOIPubMedGoogle Scholar
- Johnson LS, Shields RK, Clancy CJ. Epidemiology, clinical manifestations, and outcomes of Scedosporium infections among solid organ transplant recipients. Transpl Infect Dis. 2014;16:578–87. DOIPubMedGoogle Scholar
- Rodriguez-Tudela JL, Berenguer J, Guarro J, Kantarcioglu AS, Horre R, de Hoog GS, et al. Epidemiology and outcome of Scedosporium prolificans infection, a review of 162 cases. Med Mycol. 2009;47:359–70. DOIPubMedGoogle Scholar
- Carrer M, Vignals C, Berard X, Caradu C, Battut AS, Stenson K, et al. Retrospective multicentric study comparing infectious and noninfectious aortitis. Clin Infect Dis. 2023;76:e1369–78. DOIPubMedGoogle Scholar
- Donnelly JP, Chen SC, Kauffman CA, Steinbach WJ, Baddley JW, Verweij PE, et al. Revision and Update of the Consensus Definitions of Invasive Fungal Disease From the European Organization for Research and Treatment of Cancer and the Mycoses Study Group Education and Research Consortium. Clin Infect Dis. 2020;71:1367–76. DOIPubMedGoogle Scholar
- Ong A, Blyth CC, Bency R, Vicaretti M, Harun A, Meyer W, et al. Fatal mycotic aneurysms due to Scedosporium and Pseudallescheria infection. J Clin Microbiol. 2011;49:2067–71. DOIPubMedGoogle Scholar
- Howden BP, Slavin MA, Schwarer AP, Mijch AM. Successful control of disseminated Scedosporium prolificans infection with a combination of voriconazole and terbinafine. Eur J Clin Microbiol Infect Dis. 2003;22:111–3. DOIPubMedGoogle Scholar
- Ortmann C, Wüllenweber J, Brinkmann B, Fracasso T. Fatal mycotic aneurysm caused by Pseudallescheria boydii after near drowning. Int J Legal Med. 2010;124:243–7. DOIPubMedGoogle Scholar
- Malinowski MJ, Halandras P. Arterial reconstruction for atypical mycotic aneurysms. Vasc Endovascular Surg. 2013;47:45–7. DOIPubMedGoogle Scholar
- Centellas Pérez FJ, Martínez Antolinos C, Piqueras Sánchez S, Lorenzo González I, Llamas Fuentes F, Gómez Roldán C. [Scedosporium apiospermum infection in a kidney transplant recipient] [in Spanish]. Rev Iberoam Micol. 2019;36:48–50.PubMedGoogle Scholar
- Klinken EM, Stevenson BR, Kwok CHR, Hockley JA, Lucas M. Diffuse inflammatory aneurysmal aortitis secondary to Scedosporium apiospermum complex in an immunocompetent individual. Pathology. 2019;51:316–8. DOIPubMedGoogle Scholar
- Blasco-Lucas A, Reyes-Juárez JL, Nazarena Pizzi M, Permanyer E, Evangelista A, Galiñanes M. Aortic arch mycotic aneurysm due to Scedosporium apiospermum reconstructed with homografts. Ann Thorac Surg. 2015;99:2218–20. DOIPubMedGoogle Scholar
- Thomson S, Alibhai K, Winkelaar G, Lien D, Halloran K, Kapasi A, et al. Case report of vertebral osteomyelitis and mycotic abdominal aortic aneurysm caused by Scedosporium apiospermum in a lung transplant patient with cystic fibrosis. Transplant Proc. 2015;47:204–9. DOIPubMedGoogle Scholar
- Sayah DM, Schwartz BS, Kukreja J, Singer JP, Golden JA, Leard LE. Scedosporium prolificans pericarditis and mycotic aortic aneurysm in a lung transplant recipient receiving voriconazole prophylaxis. Transpl Infect Dis. 2013;15:E70–4. DOIPubMedGoogle Scholar
- Wakabayashi Y, Okugawa S, Tatsuno K, Ikeda M, Misawa Y, Koyano S, et al. Scedosporium prolificans endocarditis: case report and literature review. Intern Med. 2016;55:79–82. DOIPubMedGoogle Scholar
- Valerio M, Vásquez V, Álvarez-Uria A, Zatarain-Nicolás E, Pavone P, Martínez-Jiménez MDC, et al. Disseminated lomentosporiosis in a heart transplant recipient: Case report and review of the literature. Transpl Infect Dis. 2021;23:
e13574 . DOIPubMedGoogle Scholar - Holmes NE, Trevillyan JM, Kidd SE, Leong TYM. Locally extensive angio-invasive Scedosporium prolificans infection following resection for squamous cell lung carcinoma. Med Mycol Case Rep. 2013;2:98–102. DOIPubMedGoogle Scholar
- Campa-Thompson MM, West JA, Guileyardo JM, Spak CW, Sloan LM, Beal SG. Clinical and morphologic findings in disseminated Scedosporium apiospermum infections in immunocompromised patients. Proc Bayl Univ Med Cent. 2014;27:253–6. DOIPubMedGoogle Scholar
- Husain S, Muñoz P, Forrest G, Alexander BD, Somani J, Brennan K, et al. Infections due to Scedosporium apiospermum and Scedosporium prolificans in transplant recipients: clinical characteristics and impact of antifungal agent therapy on outcome. Clin Infect Dis. 2005;40:89–99. DOIPubMedGoogle Scholar
- Katragkou A, Dotis J, Kotsiou M, Tamiolaki M, Roilides E. Scedosporium apiospermum infection after near-drowning. Mycoses. 2007;50:412–21. DOIPubMedGoogle Scholar
- Hurst RW, Judkins A, Bolger W, Chu A, Loevner LA. Mycotic aneurysm and cerebral infarction resulting from fungal sinusitis: imaging and pathologic correlation. AJNR Am J Neuroradiol. 2001;22:858–63.PubMedGoogle Scholar
- Kasliwal MK, Reddy VSK, Sinha S, Sharma BS, Das P, Suri V. Bilateral anterior cerebral artery aneurysm due to mucormycosis. J Clin Neurosci. 2009;16:156–9. DOIPubMedGoogle Scholar
- Sundaram C, Goel D, Uppin SG, Seethajayalakshmi S, Borgohain R. Intracranial mycotic aneurysm due to Aspergillus species. J Clin Neurosci. 2007;14:882–6. DOIPubMedGoogle Scholar
- Coffey MJ, Fantone J III, Stirling MC, Lynch JP III. Pseudoaneurysm of pulmonary artery in mucormycosis. Radiographic characteristics and management. Am Rev Respir Dis. 1992;145:1487–90. DOIPubMedGoogle Scholar
- Lopez-Pastorini A, Koryllos A, Brockmann M, Windisch W, Stoelben E. Pseudoaneurysm of the pulmonary artery with massive haemoptysis due to an invasive pulmonary mucormycosis. Thorax. 2016;71:199–200. DOIPubMedGoogle Scholar
- Silva ME, Malogolowkin MH, Hall TR, Sadeghi AM, Krogstad P. Mycotic aneurysm of the thoracic aorta due to Aspergillus terreus: case report and review. Clin Infect Dis. 2000;31:1144–8. DOIPubMedGoogle Scholar
- Rose HD, Stuart JL. Mycotic aneurysm of the thoracic aorta caused by Aspergillus fumigatus. Chest. 1976;70:81–4. DOIPubMedGoogle Scholar
- Sanchez-Recalde A, Maté I, Merino JL, Simon RS, Sobrino JA. Aspergillus aortitis after cardiac surgery. J Am Coll Cardiol. 2003;41:152–6. DOIPubMedGoogle Scholar
Figures
Tables
Cite This ArticleOriginal Publication Date: May 03, 2024
Table of Contents – Volume 30, Number 6—June 2024
| EID Search Options |
|---|
|
|
|
|
|
|
Please use the form below to submit correspondence to the authors or contact them at the following address:
Carole Vignals, service de maladies infectieuses et tropicales, Hôpital Pellegrin, Place Amélie Raba Léon, 33000 Bordeaux, France
Top