Volume 31, Number 2—February 2025
Research
Prions in Muscles of Cervids with Chronic Wasting Disease, Norway
Figure 3
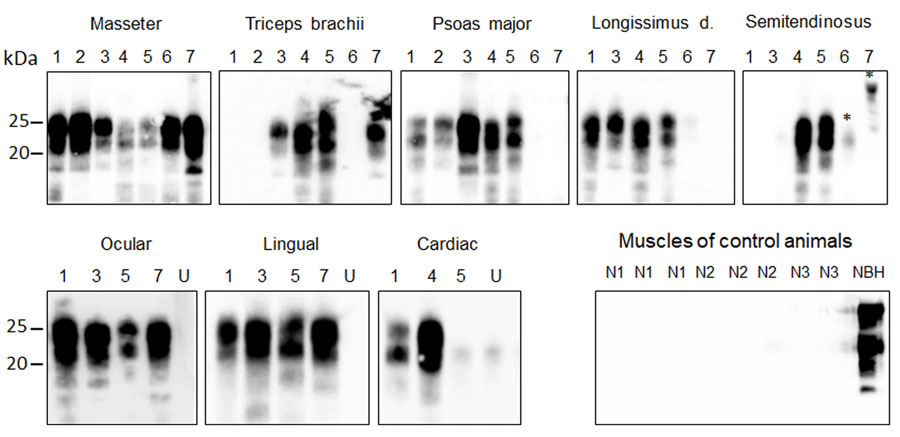
Figure 3. Detection of PrPSc (misfolded forms of the prion protein) amplification in muscle tissues of chronic wasting disease–affected cervids by protein misfolding cyclic amplification (PMCA) from a study of prions in muscles of cervids with chronic wasting disease, Norway. Muscle samples (10% homogenates) were subjected to 6 rounds of PMCA using bank vole brain homogenate substrate. PMCA products were treated with proteinase K before being analyzed by Western blot using Sha31 antibody to identify presence of PrPSc. Results demonstrate efficient amplification of PrPSc in skeletal and cardiac muscles of chronic wasting disease–affected reindeer, moose, and a red deer. No PrPSc was amplified in negative control samples. Lane designations: 1, reindeer A; 2, reindeer B; 3, moose A; 4, moose B; 5, moose C; 6, moose D; 7, red deer. Numbers at left indicate the molecular weight marker. Asterisks indicate unspecific signals. NBH, proteinase K–undigested bank vole brain homogenate used as electrophoretic migration marker of normal prion protein (PrPC); U, unseeded reaction included as a specificity control of PMCA reaction.
1These first authors contributed equally to this article.
2These authors are co–senior authors.