Volume 32, Number 2—February 2026
Dispatch
Autochthonous Rat Lungworm Angiostrongylus cantonensis Infections in Accidental and Definitive Hosts, San Diego, California, USA
Cite This Article
Citation for Media
Abstract
The rat lungworm, Angiostrongylus cantonensis, is an emerging veterinary and public health concern. We describe A. cantonensis infections in a zoo-housed parma wallaby and free-ranging Virginia opossums and roof rats in San Diego, California, USA. Angiostrongyliasis should be considered in central nervous system disease in humans and animals in this region.
Angiostrongylus cantonensis, the rat lungworm, is an invasive, zoonotic metastrongyle nematode that causes neurologic disease in humans and other vertebrate hosts (1). The usual life cycle of this parasite involves infection of the rodent definitive host by ingesting third-stage larvae (L3) found within gastropod intermediate hosts. Less commonly, paratenic hosts such as frogs, lizards, and crustaceans harbor the infective L3 and transfer A. cantonensis lungworm to rodents and accidental or aberrant hosts (1).
Since the original discovery of A. cantonensis lungworm in southern China in 1935 (2), the nematode has spread globally, including to Hawaii and more recently, the southeastern United States. In the continental United States, its range has gradually spread; infections in free-ranging rodents, gastropods, or both have been confirmed in Louisiana (3), Florida (4), Oklahoma (5), Georgia (6–7), and Texas (8). Hence, A. cantonensis lungworm is now considered endemic in those states. Cases in human and nonhuman accidental hosts have been reported in many more continental US states, with autochthonous infections suspected in Alabama (9), Mississippi (10), and Tennessee (11). In California, human cases have been sporadically reported in those who had traveled to endemic areas outside the continental United States and in those with unknown travel history (12). We describe A. cantonensis infections in a zoo-housed parma wallaby (Notamacropus parma) and free-ranging Virginia opossums (Didelphis virginiana) and roof rats (Rattus rattus) in San Diego, California, USA.
In mid-December 2024, a 7-year-old male parma wallaby that was born and raised at the San Diego Zoo (San Diego, CA, USA) showed progressive neurologic signs, including head shaking, nystagmus, central blindness, ataxia, hindlimb paresis, and extensor rigidity in all limbs. The wallaby was euthanized after 11 days of hospitalization. Necropsy revealed multifocal hemorrhage in the cerebellum and half a dozen nematodes on the leptomeningeal surfaces of the cerebellum, brainstem, and cervical spinal cord (Figure 1). Histopathology revealed necrotizing and lymphoplasmacytic meningoencephalomyelitis with fibrinoid vascular necrosis, infarction, and hemorrhage, most severely affecting the cerebellum and right cerebrum. The lesions were occasionally associated with intralesional and rarely intraparenchymal metastrongyle nematodes. A nematode collected in 70% ethanol was identified as A. cantonensis lungworm through PCR and sequencing (Appendix).
Because the diagnosis was unusual in this geographic area (3–11), we performed necropsies on free-ranging roof rats that were either euthanized as part of regular pest control or found dead on the San Diego Zoo grounds during January 14–February 14, 2025. We grossly examined 64 rats comprising both sexes and all life stages and evaluated lung and feces samples histologically and by the Baermann technique for all rats, regardless of gross findings. Two (3.1%) adult rats had lungworms and associated pneumonia. Histologically, the main findings were pulmonary arterial endothelial proliferation with thrombosis and adult metastrongyle nematodes (Figure 2) and granulomatous and fibrinosuppurative to fibrosing pneumonia with myriad intralesional adult and larval metastrongyle nematodes and eggs. Fecal examination of the 2 affected rats revealed numerous live, ≈300-μm–long larvae with coiled posterior ends. Molecular analyses of ethanol-fixed adult nematodes within the pulmonary artery and fresh feces revealed genetic sequences of A. cantonensis lungworm.
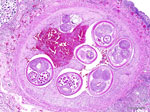
Concurrently, a local wildlife rehabilitation program had received multiple free-ranging juvenile Virginia opossums from throughout San Diego County (Figure 3) that exhibited >1 clinical signs, including dull mentation, circling, head tilt, ataxia, and respiratory distress. Most of the opossums were received during June–September in 2023–2025 and were euthanized after 0–3 days because of lack of clinical improvement. Ten opossums were submitted for postmortem examination. A consistent finding in 7 opossums was eosinophilic meningitis with intralesional metastrongyle nematodes (Appendix Figure), whereas 1 animal had eosinophilic meningitis without nematodes. The identity of the nematodes was confirmed as A. cantonensis lungworm in 6 of 7 animals through molecular analyses on formalin-fixed paraffin-embedded brain tissue (Appendix). We deposited generated nucleotide sequences from this study into GenBank (Table).
In addition, in an effort to investigate A. cantonensis lungworm in intermediate hosts, we opportunistically collected free-ranging slugs from the wallaby enclosure and adjacent areas at the San Diego Zoo (Appendix). One slug had nematode larvae encysted in various tissues, but we were unable to confirm the species identity.
We documented A. cantonensis infections in a zoo-housed parma wallaby (accidental host), free-ranging Virginia opossums (accidental host), and free-ranging roof rats (definitive host) in San Diego, California. Most cases were identified in 2025, but infection occurred as early as August 2023 in 1 opossum. Our findings indicate autochthonous infections, which pose a substantial risk to humans and accidental vertebrate hosts. Whereas autochthonous infections had not previously been documented in the United States west of Texas, identifying angiostrongyliasis cases in wildlife in San Diego County provides support that A. cantonensis lungworm could now be considered endemic in this portion of southern California, with the potential to spread to other parts of the western continental United States.
Identifying the source of introduction of A. cantonensis lungworm into San Diego was beyond the scope of this study. In Louisiana, where the nematode was first identified in the continental United States in 1987, introduction was suspected to have been from infected rats on ships that docked in New Orleans (3). A similar situation is possible in this instance, given that San Diego is a major port city. However, the volume of traffic between regions on both national and international scales has dramatically expanded since the 1980s; thus, introduction of invasive species to new areas is a constant threat (13). A likely additional factor is the distributional spread of invasive gastropods (4,8) (Appendix).
Infections in the zoo-housed wallaby and free-ranging opossums were likely the result of ingestion of an infected intermediate host. The wallaby was fed a commercially manufactured extruded diet with a mixture of grass hay and occasional browse material and fresh produce safe for human consumption; ingestion of an intermediate host was considered accidental environmental exposure. In contrast, opossums are naturally more vulnerable to infection, given that gastropods constitute a prominent part of their native diet, especially during the summer months (14). All the opossums identified with angiostrongyliasis in our study were juveniles found in the summer, suggesting the juvenile age group and summer season are risk factors for infection in this species.
A. cantonensis infection has previous been identified at the San Diego Zoo. In 2011, an African pygmy falcon (Polihierax semitorquatus) hatched and raised at the San Diego Zoo was diagnosed with meningoencephalitis caused by A. cantonensis infection (15). At that time, 20 free-ranging rats on the zoo grounds were opportunistically screened for nematode larvae in feces, but infection was not detected. With no detection in definitive hosts, the source of infection in that case and in 2 subsequent African pygmy falcons in 2014 and 2018 (San Diego Zoo, unpub. data) was suspected to be live feeder lizards (paratenic hosts) imported from Southeast Asia, where the parasite is endemic. However, given the prevalence (albeit low at 3.1%) of infected rats in our study, the parasite possibly has been present but undetected in San Diego County for some time.
In conclusion, we documented autochthonous A. cantonensis infections in southern California, highlighting a notable expansion of the range of this parasite in North America. Further studies are needed to analyze the effect of this geographic expansion and associated risks in California. Nevertheless, angiostrongyliasis should be part of the differential diagnosis for central nervous system disease in humans and animals in the wider southern United States.
Dr. Nakagun is a veterinary pathologist at the San Diego Zoo Wildlife Alliance, San Diego, California, USA. His research interest focuses on infectious diseases in zoo and wildlife species.
Acknowledgments
We thank the Disease Investigations, Veterinary Services, and Clinical Laboratory teams as well as Trent Stanley and the animal care staff at the San Diego Zoo Wildlife Alliance for their assistance in this study. We additionally thank the San Diego Humane Society’s Project Wildlife team for their support in sample collection and record maintenance, as well as John Randolph for producing the map used in this study and Molly Jarrells for assistance with case tracking. We also acknowledge Hernando Acevedo for assisting with postmortem examination of a Virginia opossum.
Testing of Virginia opossums was funded by the California Department of Fish and Wildlife through agreement no. P2480005 with the San Diego Zoo Wildlife Alliance.
References
- Wang QP, Lai DH, Zhu XQ, Chen XG, Lun ZR. Human angiostrongyliasis. Lancet Infect Dis. 2008;8:621–30. DOIPubMedGoogle Scholar
- Chen HT. A new pulmonary nematode, Pulmonema cantonensis n. g., n. sp., from rats in Canton [in French]. Ann Parasitol Hum Comp. 1935;13:312–7. DOIGoogle Scholar
- Campbell BG, Little MD. The finding of Angiostrongylus cantonensis in rats in New Orleans. Am J Trop Med Hyg. 1988;38:568–73. DOIPubMedGoogle Scholar
- Stockdale Walden HD, Slapcinsky JD, Roff S, Mendieta Calle J, Diaz Goodwin Z, Stern J, et al. Geographic distribution of Angiostrongylus cantonensis in wild rats (Rattus rattus) and terrestrial snails in Florida, USA. PLoS One. 2017;12:
e0177910 . DOIPubMedGoogle Scholar - York EM, Creecy JP, Lord WD, Caire W. Geographic range expansion for rat lungworm in North America. Emerg Infect Dis. 2015;21:1234–6. DOIPubMedGoogle Scholar
- Gottdenker NL, Nascimento Ramos RA, Hakimi H, McHale B, Rivera S, Miller BM, et al. Angiostrongylus cantonensis infection in brown rats (Rattus norvegicus), Atlanta, Georgia, USA, 2019–2022. Emerg Infect Dis. 2023;29:2167–70. DOIPubMedGoogle Scholar
- Achatz TJ, Chun CH, Young MA, Page J, Rowe M, Cooper C, et al. Detection of rat lungworms in invasive mollusks, Georgia, USA, 2024. Emerg Infect Dis. 2025;31:1852–4. DOIPubMedGoogle Scholar
- Sierra DA, Sanders TL, Edwards EE, Molter CM, Verocai GG. Zoonotic rat lungworm Angiostrongylus cantonensis in black rats, Houston, Texas, 2024. Emerg Infect Dis. 2025;31:1857–60. DOIPubMedGoogle Scholar
- Kottwitz JJ, Perry KK, Rose HH, Hendrix CM. Angiostrongylus cantonensis infection in captive Geoffroy’s tamarins (Saguinus geoffroyi). J Am Vet Med Assoc. 2014;245:821–7. DOIPubMedGoogle Scholar
- Costa LRR, McClure JJ, Snider TG III, Stewart TB. Verminous meningoencephalomyelitis by Angiostrongylus (=Parastrongylus) cantonensis in an American miniature horse. Equine Vet Educ. 2000;12:2–6. DOIGoogle Scholar
- Flerlage T, Qvarnstrom Y, Noh J, Devincenzo JP, Madni A, Bagga B, et al. Angiostrongylus cantonensis eosinophilic meningitis in an infant, Tennessee, USA. Emerg Infect Dis. 2017;23:1756–8. DOIPubMedGoogle Scholar
- Liu EW, Schwartz BS, Hysmith ND, DeVincenzo JP, Larson DT, Maves RC, et al. Rat lungworm infection associated with central nervous system disease—eight U.S. states, January 2011–January 2017. MMWR Morb Mortal Wkly Rep. 2018;67:825–8. DOIPubMedGoogle Scholar
- Hulme PE. Trade, transport and trouble: managing invasive species pathways in an era of globalization. J Appl Ecol. 2009;46:10–8. DOIGoogle Scholar
- Hopkins DD, Forbes RB. Dietary patterns of the Virginia opossum in an urban environment. Murrelet. 1980;61:20–30. DOIGoogle Scholar
- Burns RE, Bicknese EJ, Qvarnstrom Y, DeLeon-Carnes M, Drew CP, Gardiner CH, et al. Cerebral Angiostrongylus cantonensis infection in a captive African pygmy falcon (Polihierax semitorquatus) in southern California. J Vet Diagn Invest. 2014;26:695–8. DOIPubMedGoogle Scholar
Figures
Table
Cite This ArticleOriginal Publication Date: February 12, 2026
Table of Contents – Volume 32, Number 2—February 2026
| EID Search Options |
|---|
|
|
|
|
|
|

Please use the form below to submit correspondence to the authors or contact them at the following address:
Shotaro Nakagun, San Diego Zoo Wildlife Alliance, PO Box 120551, San Diego, CA 92112-0551, USA
Top