Volume 32, Number 3—March 2026
Research
Lymphocytic Choriomeningitis Virus Seroprevalence among Urban Pregnant Women and Newborns, Philadelphia, Pennsylvania, USA, 2021
Figure
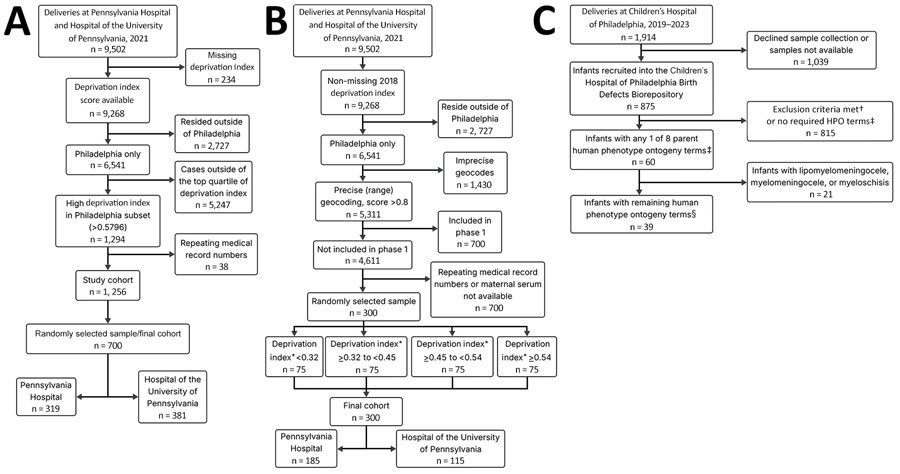
Figure. Flowchart of 3 phases of a study of lymphocytic choriomeningitis virus seroprevalence among urban pregnant women and newborns, Philadelphia, Pennsylvania, USA, 2021. A) Phase 1; B) phase 2; C) phase 3. Phases 1 and 2 used deliveries at Pennsylvania Hospital and the Hospital of the University of Pennsylvania in 2021 as the starting population. Repeated medical record numbers indicate parturient patients with multiple deliveries or >1 delivery during the study period, which would result in replicate maternal serum samples if retained. Phase 3’s starting population was the Children’s Hospital of Philadelphia Special Delivery Unit newborns delivered from the inception of the Birth Defects Biorepository during June 1, 2019–June 1, 2023. *Deprivation index ranges were determined using the Community Deprivation Index (https://github.com/geomarker-io/dep_index) on the basis of the geocoded address and quartile values were calculated on the full eligible cohort before phase-specific exclusions (n = 4,611). †Exclusion criteria included no cytomegalovirus testing on file, no placenta tissue available, and plasma not available for both maternal/infant matched samples even within the first few weeks after delivery. ‡The 8 overall HPO terms (https://hpo.jax.org) used encompassed 615 conditions. Those 8 HPO terms were abnormal choroid morphology, abnormal retinal morphology, cerebral calcification, intracranial cystic lesion, abnormality of neuronal migration, abnormal cerebral morphology, open neural tube defect, and abnormal cerebral ventricle morphology. §After excluding lipomyelomeningocele, myelomeningocele, and myeloschisis, 612 children’s HPO conditions remained. HPO, Human Phenotype Ontology.
1These first authors contributed equally to this article.