Disclaimer: Early release articles are not considered as final versions. Any changes will be reflected in the online version in the month the article is officially released.
Volume 32, Number 5—May 2026
Dispatch
Exposure in Horses to Human Tick-Borne Relapsing Fever Agent Borrelia persica, Israel, 2025
Suggested citation for this article
Abstract
Human tick-borne relapsing fever caused by Borrelia persica is common in western Asia. A survey of 301 horses in Israel revealed 9.96% seropositivity toward B. persica antigens; 1 horse (0.33%) was also PCR positive for B. persica DNA. Phylogenetic analysis supported a transmission cycle involving ticks, humans, and horses.
Tick-borne relapsing fever (TBRF) is a human and veterinary illness caused by spirochetes of the genus Borrelia (1). Borrelia persica, transmitted by the argasid tick Ornithodoros tholozani, is the primary cause of TBRF in Israel and affects persons in other parts of the eastern Mediterranean basin and Asia (2–4). Relapsing fever in humans is characterized by episodes of recurrent fever, lethargy, and headache; an up to 10% mortality rate in untreated patients has been documented (5). TBRF caused by B. persica in dogs and cats is associated with fever, lethargy, anorexia, anemia, and thrombocytopenia and can be fatal (6). Several species of wild animals are known potential reservoirs for B. persica spirochetes, including the red fox (Vulpes vulpes), golden jackal (Canis aureus), European badger (Meles meles), and some rodent species (7–9). In contrast to Borrelia spp. that cause Lyme disease in horses, for which high seroprevalence has been reported in several countries (10), equine TBRF is poorly documented. One case of equine abortion has been reported in a mare from California infected with either B. parkeri or B. turicatae, which could not be distinguished (10,11). We tested samples from horses from throughout Israel for B. persica seroreactivity.
We collected samples from 301 clinically unremarkable horses from 27 stables throughout Israel, representing the geographic distribution of the equine population, and from 12 horses living in a nonendemic area for TBRF in southern Israel, where O. tholozani ticks have not been reported (7), which served as negative controls. Of the 301 horses, 1 (0.33%) was positive for B. persica DNA by using real-time PCR amplification, confirmed by sequencing of the flaB and glpQ genes, as described previously (9). Thirty (9.96%) horses were seroreactive against B. persica antigen by ELISA testing.
The ELISA assay was developed and performed as previously described for dogs and cats (8) with adaptation to horses. We extracted crude antigen from 1 L of B. persica culture containing 107 spirochetes/mL and lysed by sonication. We coated each well with 0.7 µg of antigen. We performed blocking by using 5% skimmed milk powder dissolved in phosphate-buffered saline (PBS) (Merck KGaA, https://www.emdgroup.com) and incubated the plates overnight at 4°C. We diluted the tested serum 1:500 in PBS with 0.1% Tween 20 (PBS-T) and 2% fetal bovine serum. We incubated serum bound antibodies for 1 hour at 37°C with horseradish peroxide-conjugated goat anti-horse IgG (ABCAM, https://www.abcam.com) diluted 1:50,000 in PBS-T and 2% fetal bovine serum. We read each plate when the absorbance (λ = 405 nm) of the positive reference serum reached an optical density (OD) value of 1.1. We determined a cutoff value of 0.35 OD by adding 3 SEs to the mean absorbance of serum from the negative control group. Serum from the horse found positive for B. persica by real-time PCR and sequencing was used as a positive control.
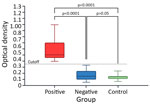
The median serologic OD was 0.472 for the seropositive, 0.145 for the seronegative, and 0.138 for the negative control horses (Figure 1). We found significant differences between the 3 groups (Kruskal-Wallis H = 64.72; p<0.0001). Posthoc Dunn tests demonstrated that seropositive horses had a higher OD than both seronegative (adjusted p<0.0001; mean rank difference = 156.98) and negative control (adjusted p<0.0001; mean rank difference = 182.00) groups. We did not observe a significant difference between seronegative and negative control groups (adjusted p = 1.000; mean rank difference = 25.02) (Figure 1). Seropositive horses were significantly older than seronegative ones (mean +SD 13.8 +6.3 years for seropositive vs. 10.8 +5.4 years for seronegative; Mann-Whitney U = 2,633, p = 0.009). Furthermore, we found age was a significant independent predictor of TBRF seropositivity with an odds ratio (OR) of 1.09 per year (95% CI 1.03–1.17; p = 0.007). Seropositivity did not significantly differ according to sex (χ2 = 2.32; p = 0.313) or housing type (χ2 = 2.01; p = 0.367).
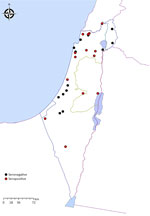
The PCR-positive horse was also found to be seropositive (OD 1.04). It was a 10-year-old gelding from a stall in the city of Ramat Gan, Israel. Sequencing analysis of a 272-bp segment of the flaB and a 224-bp segment of the glpQ gene fragments from the positive horse revealed 100% identity and coverage to a B. persica amplified from a human (GenBank accession no. DQ679907.1 [flab]) and from an O. tholozani tick (GenBank accession no. HM161658.1 [glp]). Seropositive horses resided in 15 different locations (Figure 2). All positive horses were adults with a median age of 13 years (range 3–38 years); there were 18 geldings and 12 mares.
Phylogenetic analysis on the basis of a 272-bp segment of the flaB gene sequence (Figure 3) revealed that the B. persica sequence from the positive horse clustered together with a B. persica genotype II sequence previously amplified from a human in Israel (GenBank DQ679907.1) (12). All B. persica sequences clustered separately from other Old World relapsing fever Borrelia spp. that are transmitted by ixodid ticks, including B. lonestari and B. theileri, which clustered together, but separately from B. miyamotoi, which clustered with B. hermsii. Phylogenetic analysis on the basis of a 224-bp segment of the glpQ gene (Figure 4) also revealed that the sequence from the B. persica–positive horse clustered with other B. persica sequences from a human, a cat, a European badger, and an O. tholozani tick and with the corresponding sequence of the only published whole-genome reference of B. persica from an Ornithodoros papillipes tick in Uzbekistan (GenBank accession no. AYOT00000000).
This study describes a survey of TBRF Borrelia spp. in horses. Furthermore, it introduces a new ELISA optimized for horse serum to detect antibodies against B. persica, enabling seroepidemiologic investigations. Our findings indicate an unexpectedly high seroprevalence (9.96%) among clinically healthy horses, suggesting high exposure to B. persica and to infected O. tholozani ticks. B. persica DNA was detectable in 1 of the seropositive horses, which indicates that horses are not only exposed to this spirochete but also might develop a circulating infection. The horses included in the study were all apparently healthy, but, as is the case for humans, dogs, and cats, illness might develop after infection. Furthermore, horses might serve as potential reservoir hosts for TBRF, which could then be transmitted to humans and other animals.
Israel and the surrounding region are not endemic for Lyme borreliosis, and no autochthonous infections have been reported in humans or animals in Israel to our knowledge. Therefore, serologic cross-reactivity with a Lyme borreliosis agent is unlikely in the studied horses. Nevertheless, the potential occurrence of other relapsing fever species besides B. persica in Israel is possible (13). The glpQ phylogram displays variation in 2 B. persica sequences (GenBank accession nos. EU914143.1 [tick] and AY530742.1 [human]) that were amplified in Israel. Those sequences share <95% identity with other B. persica glpQ sequences deposited into GenBank and could possibly belong to different species.
Increasing age was associated with seropositivity, which aligns with prolonged or cumulative exposure to tick vectors during the horse lifespan. Neither horse sex nor housing conditions was associated with seropositivity, indicating exposure regardless of management practices.
The phylogenetic analyses we report support a potential shared transmission cycle of B. persica involving horses, humans, and O. tholozani ticks, and suggesting a potential zoonotic risk associated with infected equine populations. Horses might serve as reservoirs and sentinels for human infection with B. persica, and One Health approach strategies should be implemented to monitor and manage risks posed by this pathogen.
Dr. Shwartz is a veterinary doctor and a PhD candidate in the Koret School of Veterinary Medicine, Hebrew University of Jerusalem, Israel. His research interests focus on vector-borne zoonotic diseases.
Acknowledgments
This study was approved by the Internal Research Committee of the Koret School of Veterinary Medicine Veterinary Teaching Hospital at the Hebrew University (approval no. KSVM-VTH/23_2014).
All data generated or analyzed during this study are included in this manuscript. Sequences obtained in this study were deposited in GenBank (accession nos. PV925730.1 [flab] and PV925731.1 [glpQ]).
References
- Dworkin MS, Schwan TG, Anderson DE Jr, Borchardt SM. Tick-borne relapsing fever. [viii. ]. Infect Dis Clin North Am. 2008;22:449–68.DOIPubMedGoogle Scholar
- Safdie G, Farrah IY, Yahia R, Marva E, Wilamowski A, Sawalha SS, et al. Molecular characterization of Borrelia persica, the agent of tickborne relapsing fever in Israel and the Palestinian Authority. PLoS One. 2010;5:
e14105 . DOIPubMedGoogle Scholar - Muigg V, Seth-Smith HMB, Goldenberger D, Egli A, Nickel B, Dürig R, et al. Tick-borne relapsing fever caused by Borrelia persica in traveler to Central Asia, 2019. Emerg Infect Dis. 2020;26:824–6. DOIPubMedGoogle Scholar
- Davis GE, Hoogstraal H. Biology of the spirochete Borrelia persica, found in the tick Ornithodorus tholazani (Argasinae) collected in the governorat of the Western Egyptian Desert; comments on the distribution and ecology of the vector tick [in French]. Ann Parasitol Hum Comp. 1956;31:147–54. DOIPubMedGoogle Scholar
- Barbour AG. Relapsing fever and Borrelia miyamotoi disease. In: Loscalzo J, Fauci A, Kasper D, Hauser S, Longo D, Jameson JL, editors. Harrison’s principles of internal medicine. 21st ed. New York: McGraw-Hill Education; 2022.
- Baneth G, Nachum-Biala Y, Halperin T, Hershko Y, Kleinerman G, Anug Y, et al. Borrelia persica infection in dogs and cats: clinical manifestations, clinicopathological findings and genetic characterization. Parasit Vectors. 2016;9:244. DOIPubMedGoogle Scholar
- Kleinerman G, Eshed T, Nachum-Biala Y, King R, Baneth G. Transmission of the human relapsing fever spirochete Borrelia persica by the argasid tick Ornithodoros tholozani involves blood meals from wildlife animal reservoirs and mainly transstadial transfer. Appl Environ Microbiol. 2021;87:e03117–20. DOIPubMedGoogle Scholar
- Baneth G, Dvorkin A, Ben-Shitrit B, Kleinerman G, Salant H, Straubinger RK, et al. Infection and seroprevalence of Borrelia persica in domestic cats and dogs in Israel. Parasit Vectors. 2022;15:102. DOIPubMedGoogle Scholar
- Shwartz D, Nachum-Biala Y, Oren S, Aharoni K, Edery N, Moss L, et al. Borrelia persica infection in wild carnivores in Israel: molecular characterization and new potential reservoirs. Parasit Vectors. 2023;16:337. DOIPubMedGoogle Scholar
- Divers TJ, Gardner RB, Madigan JE, Witonsky SG, Bertone JJ, Swinebroad EL, et al. Borrelia burgdorferi infection and Lyme disease in North American horses: a consensus statement. J Vet Intern Med. 2018;32:617–32. DOIPubMedGoogle Scholar
- Walker RL, Read DH, Hayes DC, Nordhausen RW. Equine abortion associated with the Borrelia parkeri-B. turicatae tick-borne relapsing fever spirochete group. J Clin Microbiol. 2002;40:1558–62. DOIPubMedGoogle Scholar
- Assous MV, Wilamowski A, Bercovier H, Marva E. Characterization of relapsing fever. Emerg Infect Dis. 2006;12:1740–3. DOIPubMedGoogle Scholar
- Halperin T, Orr N, Cohen R, Hasin T, Davidovitch N, Klement E, et al. Detection of relapsing fever in human blood samples from Israel using PCR targeting the glycerophosphodiester phosphodiesterase (GlpQ) gene. Acta Trop. 2006;98:189–95. DOIPubMedGoogle Scholar
Figures
Suggested citation for this article: Shwartz D, Haras L, Nachum-Biala Y, Tirosh-Levy S, Steinman A, Baneth G. Exposure in horses to human tick-borne relapsing fever agent Borrelia persica, Israel, 2025. Emerg Infect Dis. 2026 May [date cited]. https://doi.org/10.3201/eid3205.251283
Original Publication Date: April 28, 2026
Table of Contents – Volume 32, Number 5—May 2026
| EID Search Options |
|---|
|
|
|
|
|
|


Please use the form below to submit correspondence to the authors or contact them at the following address:
Dor Shwartz, The Koret School of Veterinary Medicine, P.O. Box 12, Rheovot 7610001, Israel
Top