Volume 32, Number 5—May 2026
Dispatch
One Health Investigation into Fatal Encephalitis Caused by Pigeon Paramyxovirus Type 1, France
Figure 4
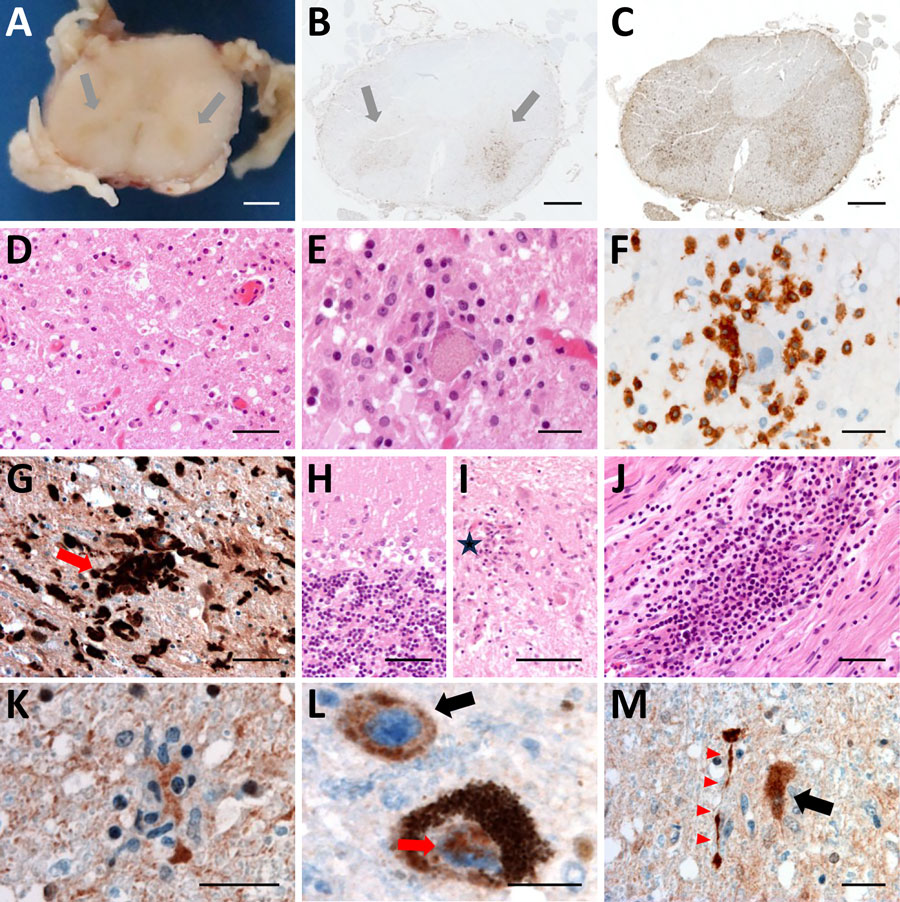
Figure 4. Histologic examination of the nervous tissue in fatal human encephalitis case caused by pigeon paramyxovirus type 1, France. A) Brown discoloration of the anterior horns (gray arrows) of the spinal cord at macroscopic examination. We evaluated the tissue microscopically by using hematoxylin and eosin staining, which highlighted a diffuse inflammatory infiltrate consisting of activated macrophages and microglia in the anterior horns of the spinal cord (throughout its entire length), the brainstem (including medulla oblongata, pons and midbrain), the thalamus, and the cerebellum. Scale bar = 2 mm. B) Anti-CD163 staining revealed marked infiltration of the anterior horns of the spinal cord by activated macrophages (gray arrows). Scale bar = 1.5 mm. C) Anti-Iba1 staining revealed activated microglia involving the anterior horns and the lateral cords of the spinal cord. This inflammatory infiltrate was associated with severe neuronal loss, particularly in the anterior horns of the spinal cord and the dentate nucleus and the Purkinje cell layer of the cerebellum. Scale bar = 1.5 mm. D, E) H&E staining revealed massive loss of motor neurons of the anterior horn (D; scale bar = 50 µm) associated with foci of neuronophagia (E; scale bar = 25 µm). F) Anti-CD3 staining revealed microglial activation was associated with T-cytotoxic lymphocyte (CD3+/CD8+) infiltration of the brain parenchyma in the most severely affected regions, the T lymphocytes surrounding a motor neuron in the gray matter of the spinal cord. Scale bar = 25 µm. G) Anti-Iba1 staining revealed multiple foci of neuronophagia and microglial nodules were observed, indicating high activity of the cellular immune response involving mainly neurons as seen in microglial nodule (red arrow) in the substantia nigra. Scale bar = 50 µm. H) H&E staining revealed that this inflammatory infiltrate was associated with severe neuronal loss, particularly in the Purkinje cell layer of the cerebellum. Scale bar = 50 µm. I) H&E staining revealed massive loss of Purkinje cells associated with Bergmann astrogliosis in the cerebellar cortex and in the anterior horns of the spinal cord and the dentate nucleus, loss of neurons in the dentate nucleus of the cerebellum associated with the presence of microglial nodules (star). Scale bar = 50 µm. J) H&E staining revealed a subtle lymphocytic infiltrate surrounding some of the vessels and a perivascular lymphocytic inflammatory infiltrate of the endoneurium of peripheral nerves. Scale bar = 50 µm. K–M) Anti-NVD staining revealed positivity in a microglial nodule in the anterior horn of the spinal cord (K), in the soma (black arrow) and in the nucleus (red arrow) of neurons in the substantia nigra (L), and (in the soma of a neuron (black arrow) and in neurites (red arrowheads) in the dentate nucleus of the cerebellum M). Scale bars = 25 µm). No cytopathic effects were noted. Immunohistochemistry testing with for herpes simplex virus 1, cytomegalovirus, polyomavirus, papovavirus, and rabies virus were negative. H&E, hematoxylin and eosin.
1These authors contributed equally to this article.
2These senior authors contributed equally to this article.