Volume 7, Number 5—October 2001
Research
Factors Contributing to the Emergence of Escherichia coli O157 in Africa
Cite This Article
Citation for Media
Abstract
In 1992, a large outbreak of bloody diarrhea caused by Escherichia coli O157 infections occurred in southern Africa. In Swaziland, 40,912 physician visits for diarrhea in persons ages >5 years were reported during October through November 1992. This was a sevenfold increase over the same period during 1990-91. The attack rate was 42% among 778 residents we surveyed. Female gender and consuming beef and untreated water were significant risks for illness. E. coli O157:NM was recovered from seven affected foci in Swaziland and South Africa; 27 of 31 patient and environmental isolates had indistinguishable pulsed-field gel electrophoresis patterns. Compared with previous years, a fivefold increase in cattle deaths occurred in October 1992. The first heavy rains fell that same month (36 mm), following 3 months of drought. Drought, carriage of E. coli O157 by cattle, and heavy rains with contamination of surface water appear to be important factors contributing to this outbreak.
Early in November 1992, physicians arriving for duty at a small hospital on a sugar plantation in Swaziland found >100 persons sprawled on the ground in front of the casualty department. Many had bloody diarrhea, and almost all were suffering severe abdominal pains. The next day the number of patients with the dysenteric illness nearly doubled, yet stool specimens sent to local laboratories did not yield common parasitic or bacterial pathogens, including Shigella spp. With the etiologic agent still unknown into the second week of the outbreak, specimens were forwarded to a reference laboratory in South Africa, where a surprising discovery was made: Escherichia coli O157 had emerged in Africa (1).
An outbreak of E. coli O157 infections was heretofore unheard of in Africa, or for that matter, anywhere in the developing world. E. coli O157 had been isolated only once before in southern Africa, from an elderly man undergoing surgery for lower gastrointestinal bleeding in Johannesburg in 1990 (2). We present a comprehensive account of a 1992 regional outbreak in Africa, perhaps the largest E. coli O157 outbreak ever reported.
The Kingdom of Swaziland occupies 17,360 km2 in southern Africa; 62% of the surface area is pastureland used primarily for cattle. The estimated human population in 1992 was 850,000. The sugar plantation referred to in this report is located in the Lubombo District, a lowland area that shares borders with Mozambique and South Africa (Figure 1). The plantation irrigates its extensive cane fields with water drawn from the nearby Usutu River. Twenty villages dispersed across the plantation housed approximately 5,000 workers and dependents. Standpipes with treated potable water and other pipes carrying untreated surface water were located in most villages. Plantation workers were provided with a weekly ration of beef obtained from local abattoirs.
When the outbreak occurred, two refugee settlements, Ndzevane (population 7,700) and Malindza (population 11,900), were located 19 km and 60 km, respectively, from the plantation. Refugees and Swazi nationals residing locally received health care at settlement clinics. Jeppe's Reef, Pongola, Empangeni, Piet Retief, and Ermelo are towns and villages in nearby South Africa.
Diarrheal Disease Surveillance
The number of patients with diarrhea at clinics in the affected area was obtained by reviewing outpatient logbooks at Malindza, Ndzevane, and the plantation hospital. National and district figures for reported diarrheal illness by month and year were provided by the Statistics Section of the Ministry of Health, Swaziland. Data on deaths were unavailable.
Rainfall Measurements
Data on rainfall at the Big Bend Experimental Farm, located approximately 1 km from the plantation, were obtained for 1991-92 from the Swaziland National Meteorological Service, Ministry of Transport and Communication. A meteorologic station on the plantation site also provided precipitation figures for October 1992.
Surveillance of Cattle Deaths
Periodic dipping of cattle with insecticides used for the control of ticks was mandatory, and losses were closely monitored in this manner. Figures for cattle deaths from 1988 through 1992 were obtained from the Ministry of Agriculture, Swaziland.
Clinical and Environmental Laboratory Methods
Initial stool specimens were cultured at local laboratories in Swaziland, which did not test for E. coli O157. Subsequent specimens were submitted to the South African Institute of Medical Research (SAIMR), which routinely attempted to identify Vibrio cholerae, Salmonella spp., Shigella spp., and Yersinia enterocolitica from all specimens; methods for isolating E. coli O157 were introduced in response to the outbreak. Human stool specimens and rectal swabs, cattle dung, and food samples were plated on alkaline peptone water, thiosulfate citrate bile salts sucrose (TCBS) agar, blood agar, selenite broth, desoxycholate citrate agar (DCA), and sorbitol/MacConkey agar and were incubated overnight. Water samples were vacuum filtered, and the filters were cut in half and incubated overnight in alkaline peptone water and selenite broth. These waters and broths were then subcultured onto TCBS, blood, and sorbitol/MacConkey agars; the selenite broth was also subcultured onto DCA and Shigella-Salmonella agars. Sucrose-fermenting colonies were further investigated biochemically as potential V. cholerae, and sorbitol-nonfermenting colonies as potential E. coli O157. Final identification was done by serotyping of V. cholerae with Wellcome polyvalent (Burroughs Wellcome Co., Research Triangle Park, NC), Inaba and Ogawa agglutinating sera, and of E. coli with the Prolex latex agglutination test kit. All other lactose-nonfermenting colonies were further investigated.
A sample of isolates identified as E. coli O157 by the Prolex kit underwent confirmatory testing at the Centers for Disease Control and Prevention (CDC), using standard methods (3). Isolates were also examined for the presence of genes encoding Shiga toxins 1 and 2, and the uidA allele specific for E. coli O157:H7 by polymerase chain reaction (PCR) (4,5). All isolates were characterized by macrorestriction analysis pulsed-field gel electrophoresis (PFGE) (6). The fliC gene from selected isolates was amplified and restriction digested as previously described to determine whether the isolates had the allele characteristic of E. coli O157:H7 (7).
Survey Methods
To characterize the diarrhea and identify risk factors for illness during the outbreak, a survey was conducted at the sugar plantation. The survey questionnaire solicited information on demographic characteristics, travel, and food and water consumption in the 2 weeks before the outbreak (October 15 to 31) and diarrheal illness from November 1 to December 31, 1992. All villages that had a village health worker available to assist on the day of the survey were included (16 of the 20 villages). Maps of each village were reviewed, and houses were randomly selected. All persons at home at the time of survey who had lived on the premises since October 1, 1992, were included.
Statistical analyses were conducted by using EpiInfo Version 6.4c. Confidence intervals (CI) for means and proportions were calculated according to the Fleiss-Quadratic method, taking stratified-cluster sampling framework into consideration. An exposure effect was considered to be statistically significant if the 95% CI for the summary risk ratio did not include 1.0.
Surveillance of Cattle Deaths
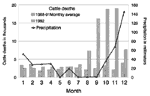
Reports from Swaziland for the 5 years from 1988 to 1992 indicate that an increase in cattle deaths began in August 1992 (Figure 2). In Lubombo District, 12,685 cattle died in September and October 1992; this represented a sevenfold increase over the monthly average of cattle deaths in Lubombo for the previous 8 months (data not shown).
Rainfall Measurements
The Big Bend Agricultural Experimental Farm reported 1.5 mm of precipitation during the 3 months preceding October 1992 (Figure 2), compared with 40 mm of precipitation for the same period in 1991 (data not shown). Of the 36 mm of rain that fell on Big Bend during October 1992, 27 mm (75%) fell on the 29th and 6 mm (17%) the next day. At the plantation, 34 mm of rain fell on October 29, accounting for 72% of the monthly total.
Diarrheal Disease Surveillance
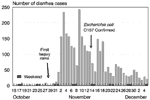
The number of visits for diarrhea at the plantation hospital from October 15 through December 5, 1992, is shown in Figure 3. Three days after the heavy rainfall, the number of patients with diarrhea seen at the plantation hospital began to increase rapidly. A total of 2,868 persons were treated for diarrhea in November; 1,884 (66%) of these presented in the first 2 weeks of the outbreak.
When the plantation outbreak occurred, nearby refugee settlements in Swaziland and townships in proximal areas of South Africa were also reporting increases in diarrheal illness. Clinician accounts indicated that patients seen at the refugee settlements in early November had abdominal pain and bloody diarrhea and were frequently locally residing Swazi nationals, not refugees. By the end of the month, however, the clinical picture had shifted to profuse watery diarrhea, characteristic of cholera, initially observed among the refugees. These impressions are supported by data from the refugee settlement clinics (Figure 4). Ndzevane clinic staff routinely recorded whether the patient was a local Swazi resident or refugee. Examining the visits of local residents and refugees separately indicates that there were two waves of diarrheal illness at Ndzevane: the first, predominantly among local residents, peaked in the second week of November; the second, primarily among refugees, peaked in early December 1992. Figures for the Malindza clinic confirm that a large outbreak occurred there as well, also peaking in the second week of November, followed by a second, subtler wave of diarrheal illness that peaked in the latter half of December. The Malindza clinic staff classified cases of diarrhea as dysentery if blood was visible in the stool. Although this diagnosis was usually made infrequently, for the week ending November 4, 1992, 74 (29%) of the 255 diarrheal illnesses treated were recorded as dysentery.
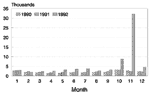
Swaziland's national diarrheal disease surveillance figures for 1990 through 1992 for persons >5 years of age are shown in Figure 5. A substantial increase in diarrheal illness in Swaziland began in October 1992, and 40,912 consultations for diarrheal illness in persons aged >5 years were reported during October and November, representing a sevenfold increase over the mean number of visits for diarrhea reported for the comparable period during 1990 and 1991. This increase was also observed among children <5 years of age, who had 15,312 consultations for acute diarrheal illness reported in November 1992, a figure more than double the monthly average for the preceding 34 months. All areas of Swaziland had increases in reported diarrheal illness in October and November 1992; this increase was most pronounced in Lubombo District (data not shown).
Laboratory Results
Clinical Specimens
Specimens submitted to local laboratories in Swaziland during the first 10 days of the outbreak did not yield a pathogen. At SAIMR, four of five rectal swabs collected on November 13, 1992, from patients treated at the plantation hospital yielded E. coli O157; no other pathogens were identified. From November 21 to December 3, 1992, 51 stool specimens were collected at the Ndzevane clinic; 7 yielded only E. coli O157, 18 yielded only V. cholerae El Tor, serotype Ogawa, and 8 yielded both pathogens.
In neighboring areas of South Africa, stool specimens obtained during nearly simultaneous outbreaks of diarrhea also yielded E. coli O157, including 3 of 20 specimens received on December 2 and 3, 1992, from Ermelo; 1 of 8 specimens received on December 2, 1992, from Empangeni; and 4 of 40 specimens collected on December 17, 1992, in Peit Retief. None of these stool specimens yielded V. cholerae, although nontyphoid Salmonella spp. were isolated from two stool specimens from Piet Retief.
Environmental Specimens
Between November 21 and December 4, 1992, E. coli O157 was isolated from 16 of 81 water samples from Swaziland and South Africa. Eight of the positive samples came from surface water on the sugar plantation (including the Usutu River), four were obtained from various sites in Ndzevane (including a borehole, a water distribution standpipe, and water stored in the home of a patient), and four were collected near Jeppe's Reef during a diarrhea outbreak (including samples from the Magadu and Sithlangu rivers). V. cholerae was not isolated from any water sample.
E. coli O157 was isolated from 3 (20%) of 15 cattle dung specimens collected at the sugar plantation on November 21, and from 3 (11%) of 27 cattle dung specimens collected near Jeppe's Reef on December 4, 1992. A single sample of day-old cooked maize collected December 3, 1992, in the Ndzevane refugee camp also yielded E. coli O157, as did two of two sewage specimens received December 2, 1992, from the Pongola area.
Plantation Survey
A total of 778 (92%) of 850 survey questionnaires distributed to residents of the 16 participating villages on the plantation were completed. Four hundred (53%) of the 752 respondents were male. The median age was 26 years.
A total of 327 (42%; 95% CI 38% to 46%) of the 778 plantation residents surveyed reported having diarrheal illness during November or December 1992; 128 (60%) of 212 persons who recalled the time of onset indicated that the illness began in the first 2 weeks of November. Of those ill, 90 (28%) reported bloody diarrhea, 285 (87%) abdominal pain, and 145 (44%) vomiting. The mean duration of illness was 4.2 days (95% CI: 3.6 to 4.8). A total of 257 (79%) of ill persons sought care at the plantation hospital, and 52% indicated they were the first person in their household to have diarrhea. All ages were affected; diarrheal attack rates were 25% to 56% for all age groups when aggregated by 5-year intervals; the attack rate varied widely among villages, from 13% to 75%.
Univariate analyses identified consumption of beef or untreated water in various forms, as well as female gender, as factors significantly associated with diarrheal illness (Table). These associations remained significant even when the case definition was restricted to those who became ill during the first 2 weeks of the outbreak or to those who stated they had been the first ill person in their household. Defining cases as only bloody diarrhea generally increased the relative risk for each of the dietary exposures. The respondents' job duties on the plantation, a history of travel away from the plantation during the last 2 weeks of October, location of their homestead in Swaziland, and household size were not associated with diarrheal illness.
Stratified analyses were conducted to assess possible confounding or interaction between the exposures identified as significant on univariate analyses. All exposure variables except the consumption of beef not purchased from the butcher remained statistically associated with self-reported diarrheal illness when the variables were stratified by each of the other exposures individually.
Molecular Analysis and PFGE Typing
Thirty-one suspected E. coli O157 isolates obtained from clinical and environmental specimens were referred to CDC, and all were confirmed as E. coli O157:NM. All isolates had the uidA allele specific for E. coli O157:H7 and the genes encoding Shiga toxins 1 and 2. All eight isolates characterized by restriction digestion of the PCR-amplified fliC gene showed a pattern characteristic of the H7 allele found in E. coli O157:H7 isolates. Twenty-seven (87%) of the 31 isolates had indistinguishable PFGE patterns (Pattern A in Figure 1), indicating that the outbreak was largely caused by a single predominant clone. This clone was isolated from various specimens, including human stools, cattle dung, surface water, sewage water, and maize, and was recovered from all six sites where specimens were obtained and tested by PFGE (isolates from a seventh site, Empangeni, were not evaluated by PFGE). Two other PFGE patterns were identified among the four remaining isolates.
This report provides comprehensive data on the first outbreak of E. coli O157 infections from the developing world. Drought, carriage of E. coli O157 by cattle, and contamination of surface water appear to have been important contributing factors. Given the extent of the outbreak, the multiple modes of transmission, and the likely recurrence of predisposing factors, the emergence of E. coli O157 has major implications for diarrheal disease control strategies in Africa.
Although the simultaneous occurrence of cholera in some areas where E. coli O157 infections were detected makes it difficult to definitively determine the magnitude of the E. coli O157 outbreak, national disease surveillance figures for Swaziland leave little doubt that a massive outbreak of diarrheal illness occurred there in 1992, and several lines of evidence suggest E. coli O157 played a major role.
First, the clinical presentation of illnesses at the plantation and of the initial patients at the refugee settlements is consistent with E. coli O157 infection. Symptomatic cholera infection almost always manifests as profuse watery diarrhea. Severe abdominal pain and bloody stools are very uncharacteristic for cholera but are the hallmark of hemorrhagic colitis caused by E. coli O157 (8-10). E. coli O157 can also present as nonbloody diarrhea (11,12). In a household survey performed during a waterborne outbreak of E. coli O157 infections in the United States, bloody stools were reported by only 35% of those with diarrhea (13). This figure is similar to the proportion of ill persons reporting bloody stools in the plantation survey (28%) and to that observed among the patients seen initially at the Malindza clinic (29%). It seems reasonable, therefore, to infer that some of the cases of nonbloody diarrhea, which were temporally and geographically associated with cases of bloody diarrhea caused by E. coli O157, represent milder manifestations of illness caused by this pathogen.
Second, medical personnel clearly recalled that there were two waves of diarrhea, dysentery followed by watery diarrhea, with each surge initially affecting different populations. (Although the term dysentery technically refers to the presence of blood and pus in stools, it is used here to mean nonwatery diarrhea, which may be bloody, accompanied by severe abdominal pain.) In a report filed November 27, 1992, following an assessment of the situation at Ndzevane, the World Health Organization (WHO) representative in Swaziland documented this impression, stating "the type of diarrhea [seen at Ndzevane among the refugees] appeared to be different from that noticed in the neighboring areas," i.e., among the Swazi nationals residing outside the camp (14). Although we suspect that cholera was the likely cause of much of the diarrhea during the second wave of illnesses at the refugee camps, we do not believe the initial dysentery outbreak among the local Swazi population can be attributed to this pathogen. (Although the term dysentery technically refers to the presence of blood and pus in stools, it is used here to mean nonwatery diarrhea, which may be bloody, accompanied by severe abdominal pain.)
Third, E. coli O157 was isolated from specimens collected from seven different diarrhea-affected foci that spanned a distance of several hundred kilometers. The actual area involved may have been even greater because in the months immediately following this outbreak, investigators in Malawi and Angola reported outbreaks of diarrheal illness in which nucleic acid amplification tests of stool specimens suggested E. coli O157 as the cause (15,16).
Estimates of the magnitude of the E. coli O157 outbreak ultimately depend on how one apportions the 64,699 consultations for diarrhea (all ages) in Swaziland during October and November 1992, after counterbalancing the identification of cholera in late November with widespread reports of dysentery in the local population and confirmation of E. coli O157 over an extended area. Even if E. coli O157 accounted for only a small proportion of visits, this would represent thousands, perhaps tens of thousands, of infections and one of the largest outbreaks of this agent in history.
Understanding the complex interaction of environmental and behavioral factors that enabled E. coli O157 to emerge so intensely in Africa is important for future diarrheal disease control efforts. Cattle are the major reservoir of E. coli O157 and shed this organism more frequently when stressed (17). This region had been experiencing several years of drought, a situation that worsened from July to September 1992. As the drought continued, cattle began to graze close to the remaining sources of surface water and vegetation. The marked increase in cattle deaths beginning in August 1992 indicates that available pastures and water sources were often insufficient.
Although the number of cattle decreased, demand for beef apparently did not. Nearly one quarter of persons surveyed on the plantation indicated that they had eaten beef from cattle known to have been found dead. Commercial beef slaughter and distribution practices were also affected by the shortage of cattle. Earlier in the year, cattle imports from South Africa to Swaziland were banned, a decision said to be supported by some ranchers because it enabled them to sell off dying animals (18). A senior health inspector noted that a single large abattoir was supplying meat to many butchers in the country since the drought and that "preventive health is not taken as a matter of priority by the look of things" at the abbatoir (19). Plantation staff also indicated, for the first time in their memory, that just before the outbreak some of the weekly beef rations were spoiled. Although adequate cooking temperatures will kill E. coli O157, much of this beef would have been grilled, leaving the possibility that portions remained undercooked.
How E. coli O157 originally found its way to cattle in southern Africa is not known, but once there it is not difficult to speculate on how it spread. Recovery of E. coli O157 from surface water in Swaziland and South Africa plausibly explains how this pathogen might be rapidly dispersed over vast stretches of land. According to local residents and our own observations, during the drought, cattle often frequented the trickling streambeds, defecating and dying there (Figure 6). With the onset of heavy rains, water contaminated with cattle feces (and presumably E. coli O157) would have become readily available to both livestock and residents in villages downstream. Such a scenario is consistent with the epidemic curve from the sugar plantation outbreak; E. coli O157 has a median incubation period of 3 to 4 days, and the outbreak exploded 3 days after the first heavy rains in 4 months (10). PFGE results indicate that a single clone was responsible for much of the outbreak, suggesting that either there was an exceptionally narrow range of diversity among E. coli O157 isolates in south African cattle or that a single clone was able to expeditiously spread over large distances, perhaps via surface waterways revitalized by heavy rains.
By early 1993 we no longer detected E. coli O157 in patient stools or environmental specimens obtained from the affected area, intimating that the outbreak had ended almost as abruptly as it started. Since that time, however, two other outbreaks of dysentery have been attributed to E. coli O157 elsewhere in Africa, the first in Central African Republic in 1996, and the second in Cameroon beginning in 1997 (20,21). Both these reports have some constraints, however; in the Central African Republic, E. coli O157 was not isolated at the outbreak site but only implicated from molecular tests. In the Cameroon outbreak, E. coli O157 and Shigella were each isolated in about half of the specimens tested.
Although reported outbreaks of E. coli O157 in Africa have been few to date, available information indicates that the pathogen has wide geographic distribution. Since the 1992 outbreak, culture-proven E. coli O157 diarrheal illness has been reported from multiple locations, including Kenya, Nigeria, Côte d'Ivoire, and Central Africa Republic (22-25). In Egypt, 6 (5%) of 125 meat specimens obtained from slaughterhouses yielded E. coli O157 (26). Because E. coli O157 is not detected by the usual methods used to isolate and identify traditional enteric bacterial pathogens and microbiology laboratories in many countries in Africa do not routinely test for this pathogen, E. coli O157 infections may go unrecognized (27,28). Reports on African dysentery outbreaks attributed to Shigella sometimes indicate that specimens were not tested until several months into the outbreak or do not describe laboratory methods that are suitable for detecting E. coli O157 (29,30). This is unfortunate because the spectrum of clinical illness resulting from Shigella infection overlaps considerably with that of E. coli O157 and mixed outbreaks have been reported (20).
WHO has stated that Shigella dysenteriae type 1 "is the only cause of large-scale, regional outbreaks of dysentery" (emphasis WHO), but data presented here suggest that this assertion may be too restrictive (27). We are concerned that if another large outbreak of severe bloody diarrhea caused by E. coli O157 occurred today in Africa, it might be assumed to be due to Shigella spp. and E. coli O157 would not be sought.
Determining the extent to which E. coli O157 is responsible for dysentery in Africa is of more than academic concern because syndromic management protocols for dysentery in areas where the etiologic agent cannot be readily confirmed often include empiric treatment with antibiotics. Antibiotics have not been clearly shown to ameliorate E. coli O157 infection, and recent data indicate that treating these infections with antibiotics may predispose the patient to developing serious complications, including hemolytic uremic syndrome (HUS) (10,31). HUS is estimated to occur in 8% of children and an unknown proportion of adults with E. coli O157 and, without dialysis, can be fatal (10).
More work is needed to better define the incidence of E. coli O157-associated dysentery in Africa so that optimal recommendations for empiric antibiotic therapy can be provided. At present, we recommend that in outbreaks of dysentery in Africa, stools from ill patients be tested for E. coli O157 in addition to Shigella spp. If the etiologic agent is not identified, consideration should be given to examining stools for other Shiga toxin-producing E. coli. Regional studies of E. coli O157 carriage rates among livestock in various parts of Africa might also be useful in assessing the potential for future outbreaks.
In this investigation, E. coli O157 was recovered from multiple water sources, including a borehole, a standpipe, and water stored in the home. Thus, ensuring the purity of potable water through practical, low-cost strategies might have reduced the risk for infection (32-34).
Several limitations of this investigation should be noted. First, the survey at the plantation did not adjust the period of exposure inquired about to account for differences in date of illness onset for each patient. Instead we asked all persons, both ill and well, about exposures during the 2 weeks preceding the outbreak.
Second, with conditional logistic regression analyses we were unable to identify significant independent predictors of illness among the risk exposures reported by the plantation residents. Many household clusters had at least one indeterminate response to an exposure question (e.g., replied, "Don't remember" or no response) and therefore the entire cluster had to be excluded from such analyses, resulting in conditional regression models that were unstable.
A third limitation is our inability to assess the impact of the E. coli O157 outbreak on human deaths. While visiting area hospitals, we heard anecdotal accounts of patients who became edematous and died without known cause. We suspect that these cases represented end-stage manifestations of HUS, but quantifiable data were not available.
Despite widespread recognition of the importance of environmental factors in facilitating the emergence of infectious agents, the information available in the scientific literature remains relatively limited (35-38). This manuscript combines environmental and veterinary data together with disease incidence, environmental testing, and microbial "fingerprinting" to develop one of the more complete examples currently available. The data presented suggest that carriage of E. coli O157 by cattle, cattle deaths secondary to drought, and heavy rains that resulted in contamination of surface water were important factors contributing to the emergence of E. coli O157 in Africa. Given that drought and heavy rains will likely recur in Africa, the possibility that E. coli O157 will once again emerge to cause a major regional outbreak cannot be excluded. Clinicians need to be aware of this so that delayed diagnosis and inappropriate treatment resulting in loss of lives can be avoided.
Dr. Effler is State Epidemiologist for the Hawaii Department of Health. His current responsibilities encompass disease surveillance and outbreak control, oversight of the state's immunization program, and bioterrorism preparedness.
Acknowledgment
The authors thank Serge Male and the United Nations High Commission for Refugees for their support in conducting this investigation. The authors also thank Lynne McIntyre and April Bogard for their valuable editorial assistance.
References
- Isaäcson M, Canter PH, Effler P, Arntzen L, Bomans P, Heenan R. Haemorrhagic colitis epidemic in Africa. Lancet. 1993;341:961. DOIPubMedGoogle Scholar
- Browning NG, Botha JR, Sacho H, Moore PJ. Escherichia coli O157:H7 haemorrhagic colitis: report of the first South African case. S Afr Med J. 1990;28:28–9.PubMedGoogle Scholar
- Ewing WH. Edwards and Ewing's identification of enterobacteriaceae. 4th ed. New York: Elsevier Science Publishing Co. Inc.; 1986.
- Olsvik O, Rimstead E, Hornes E, Strockbine N, Wasteson Y, Lund A, A nested PCR followed by magnetic separation of amplified fragments for detection of Escherichia coli Shiga-like toxin genes. Mol Cell Probes. 1991;5:429–35. DOIPubMedGoogle Scholar
- Cebula TA, Payne WL, Feng P. Simultaneous identification of strains of Escherichia coli O157:H7 and their Shiga-like toxin type by mismatch amplification mutation assay-multiplex PCR. J Clin Microbiol. 1995;33:248–50.PubMedGoogle Scholar
- Barrett TJ, Lior H, Green JH, Khakhria R, Wells JG, Bell BP, Laboratory investigation of a multistate food-borne outbreak of Escherichia coli O157:H7 by using pulsed-field gel electrophoresis and phage typing. J Clin Microbiol. 1994;32:3013–7.PubMedGoogle Scholar
- Fields PI, Blom K, Hughes HJ, Helsel LO, Feng P, Swaminathan B. Molecular characterization of the gene encoding H antigen in Escherichia coli and development of a PCR-restriction fragment length polymorphism test for identification of E. coli O157:H7 and O157:NM. J Clin Microbiol. 1997;35:1066–70.PubMedGoogle Scholar
- Boyce TG, Swerdlow DL, Griffin PM. Escherichia coli O157:H7 and the hemolytic-uremic syndrome. N Engl J Med. 1995;333:364–6. DOIPubMedGoogle Scholar
- Griffin PM, Ostroff SM, Tauxe RV, Greene KD, Wells JG, Lewis JH, Illnesses associated with Escherichia coli O157:H7 infections: a broad clinical spectrum. Ann Intern Med. 1988;109:705–12.PubMedGoogle Scholar
- Griffin PM, Tauxe RV. The epidemiology of infections caused by Escherichia coli O157:H7, other enterohemorrhagic E. coli, and the associated hemolytic uremic syndrome. Epidemiol Rev. 1991;13:60–91.PubMedGoogle Scholar
- Rodrigue DC, Mast EE, Greene KD, Davis JP, Hutchinson MA, Wells JG, A university outbreak of Eschericheria coli O157:H7 infections associated with roast beef and an unusually benign clinical course. J Infect Dis. 1995;172:1122–5. DOIPubMedGoogle Scholar
- Swerdlow DL, Woodruff BA, Brady RC, Griffin PM, Tippen S, Donnell JD, A waterborne outbreak in Missouri of Escherichia coli O157:H7 associated with bloody diarrhea and death. Ann Intern Med. 1992;117:812–9.PubMedGoogle Scholar
- Mwambazi WC. Internal report. Swaziland: World Health Organization; 27 Nov 1992.
- Paquet C, Perea W, Grimont F, Collin M, Guillod M. Aetiology of haemorrhagic colitis epidemic in Africa. Lancet. 1993;342:175. DOIPubMedGoogle Scholar
- Calundungo R, Maddau G, Rappelli P, Cappuccinelli P, Leite F, Escherichia coli and enteritis in Angola. Lancet. 1994;344:538–9. DOIPubMedGoogle Scholar
- Armstrong GL, Hollingsworth J, Morris JJG. Emerging foodborne pathogens: Escherichia coli O157:H7 as a model of entry of a new pathogen into the food supply of the developed world. Epidemiol Rev. 1996;18:29–51.PubMedGoogle Scholar
- Mkwanazi B. Butcheries threaten closure. The Times of Swaziland. Aug 13, 1992.
- Memorandum from the Senior Health Inspector, Ministry of Health, Government of Swaziland to the Principal Secretary, Ministry of Interior, RE: Control of diarrhoeal outbreak; 28 Nov 1992.
- Cunin P, Tedjouka E, Germani Y, Ncharre C, Bercion R, Morvan J, An epidemic of bloody diarrhea: Escherichia coli O157 emerging in Cameroon? Emerg Infect Dis. 1999;5:285–90. DOIPubMedGoogle Scholar
- Germani Y, Soro B, Vohito M, Morel O, Morvan J. Enterohaemorrhagic Escherichia coli in Central African Republic. Lancet. 1997;349:1670. DOIPubMedGoogle Scholar
- Sang WK, Saidi SM, Yamamoto H, Ezaki T, Iida T, Yoh M, Haemorrhagic colitis due to Escherichia coli O157:H7 in Kenya. J Trop Pediatr. 1996;42:118–9. DOIPubMedGoogle Scholar
- Akinyemi KO, Oyefolu AO, Opere B, Otunba-Payne VA, Oworu AO. Escherichia coli in patients with acute gastroenteritis in Lagos, Nigeria. East Afr Med J. 1998;75:512–5.PubMedGoogle Scholar
- Dadie A, Karou T, Adom N, Kette A, Dosso M. Isolation of enteric pathogeic agents in Côte d'Ivoire: Escherichia coli O157:H7 and enteroaggregative E. coli. Bull Soc Pathol Exot. 2000;93:95–6.PubMedGoogle Scholar
- Germani Y, Minssart P, Vohito M, Yassibanda S, Glaziou P, Hocquet D, Etiologies of acute, persistent, and dysenteric diarrheas in adults in Bangui, Central African Republic, in relation to human immunodeficiency virus serostatus. Am J Trop Med Hyg. 1998;59:1008–14.PubMedGoogle Scholar
- Abdul-Raouf UM, Ammar MS, Beuchat LR. Isolation of Escherichia coli O157:H7 from some Egyptian foods. Int J Food Microbiol. 1996;29:423–6. DOIPubMedGoogle Scholar
- World Health Organization. Guidelines for the control of epidemics due to Shigella dysenteriae type 1 (WHO/CDR/95.4). Geneva: The Organization; 1995. p. 1,47.
- Wittenberg DF. Emerging and re-emerging diseases--epidemic enterohaemorrhagic infections 100 years after Shiga. S Afr Med J. 1999;89:750–2.PubMedGoogle Scholar
- Aragon M, Barreto A, Chambule J, Noya A, Tallarico M. Shigellosis in Mozambique: the 1993 outbreak rehabilitation-a follow-up study. Trop Doct. 1995;25:159–62.PubMedGoogle Scholar
- Malakooti MA, Alaii J, Shanks GD, Phillips-Howard PA. Epidemic dysentery in western Kenya. Trans R Soc Trop Med Hyg. 1997;91:541–3. DOIPubMedGoogle Scholar
- Wong CS, Jelacic S, Habeeb RL, Watkins SL, Tarr PI. The risk of the hemolytic-uremic syndrome after antibiotic treatment of Escherichia coli O157:H7 infections. N Engl J Med. 2000;342:1930–6. DOIPubMedGoogle Scholar
- Mintz ED, Reiff FM, Tauxe RV. Safe water treatment and storage in the home: a practical new strategy to prevent waterborne disease. JAMA. 1995;273:948–53. DOIPubMedGoogle Scholar
- Reiff F, Roses M, Venczel L, Quick R, Witt V. Low cost safe water for the world: a practical interim solution. Health Policy. 1996;17:389–408.PubMedGoogle Scholar
- Quick RE, Venczel LV, Mintz ED, Soleto L, Aparicio J, Gironaz M, Diarrhoea prevention in Bolivia through point-of-use water treatment and safe storage: a promising new strategy. Epidemiol Infect. 1999;122:83–90. DOIPubMedGoogle Scholar
- Morse SS. Factors in the emergence of infectious diseases. Emerg Infect Dis. 1995;1:7–15. DOIPubMedGoogle Scholar
- Colwell R, Epstein P, Gubler D, Hall M, Reiter P, Shukla J, Global climate change and infectious diseases. Emerg Infect Dis. 1998;4:451–2. DOIPubMedGoogle Scholar
- Engelthaler DM, Mosley DG, Cheek JE, Levy CE, Komatsu KK, Ettestad P, Climatic and environmental patterns associated with hantavirus pulmonary syndrome, Four Corners region, United States. Emerg Infect Dis. 1999;5:87–94. DOIPubMedGoogle Scholar
- Feldbaum H. Forging new perspectives on disease surveillance. Emerg Infect Dis. 1998;4:337–8. DOIPubMedGoogle Scholar
Figures
Table
Cite This ArticleTable of Contents – Volume 7, Number 5—October 2001
| EID Search Options |
|---|
|
|
|
|
|
|



Please use the form below to submit correspondence to the authors or contact them at the following address:
Paul V. Effler, State of Hawaii Department of Health, 1250 Punchbowl Street, Room 444, Honolulu, Hawaii 96813, USA; fax: 808-586-8347
Top