Volume 17, Number 10—October 2011
Perspective
Global Spread of Carbapenemase-producing Enterobacteriaceae
Cite This Article
Citation for Media
Abstract
Carbapenemases increasingly have been reported in Enterobacteriaceae in the past 10 years. Klebsiella pneumoniae carbapenemases have been reported in the United States and then worldwide, with a marked endemicity at least in the United States and Greece. Metallo-enzymes (Verona integron–encoded metallo-β-lactamase, IMP) also have been reported worldwide, with a higher prevalence in southern Europe and Asia. Carbapenemases of the oxacillinase-48 type have been identified mostly in Mediterranean and European countries and in India. Recent identification of New Delhi metallo-β-lactamase-1 producers, originally in the United Kingdom, India, and Pakistan and now worldwide, is worrisome. Detection of infected patients and carriers with carbapenemase producers is necessary for prevention of their spread. Identification of the carbapenemase genes relies mostly on molecular techniques, whereas detection of carriers is possible by using screening culture media. This strategy may help prevent development of nosocomial outbreaks caused by carbapenemase producers, particularly K. pneumoniae.
Enterobacteriaceae are inhabitants of the intestinal flora and are among the most common human pathogens, causing infections such as cystitis and pyelonephritis with fever, septicemia, pneumonia, peritonitis, meningitis, and device-associated infections. Enterobacteriaceae are the source of community- and hospital-acquired infections. They have the propensity to spread easily between humans (hand carriage, contaminated food and water) and to acquire genetic material through horizontal gene transfer, mediated mostly by plasmids and transposons.
Since 2000, spread of community-acquired enterobacterial isolates (Escherichia coli) that produce extended-spectrum β-lactamases (ESBLs) capable of hydrolyzing almost all cephalosporins except carbapenems has been reported worldwide (1). It is therefore mandatory to maintain the clinical efficacy of carbapenems (imipenem, ertapenem, meropenem, doripenem), which have become antimicrobial drugs of last resort. These agents are crucial for preventing and treating life-threatening nosocomial infections, which are often associated with techniques developed in modern medicine (transplantation, hospitalization in an intensive care unit, highly technical surgery).
Carbapenem-resistant Enterobacteriaceae have been reported worldwide as a consequence largely of acquisition of carbapenemase genes (2). The first carbapenemase producer in Enterobacteriaceae (NmcA) was identified in 1993 (3). Since then, a large variety of carbapenemases has been identified in Enterobacteriaceae belonging to 3 classes of β-lactamases: the Ambler class A, B, and D β-lactamases (2). In addition, rare chromosome-encoded cephalosporinases (Ambler class C) produced by Enterobacteriaceae may possess slight extended activity toward carbapenems, but their clinical role remains unknown (2,4).
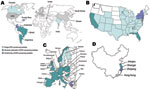
A variety of class A carbapenemases have been described; some are chromosome encoded (NmcA, Sme, IMI-1, SFC-1), and others are plasmid encoded (Klebsiella pneumoniae carbapenemases [KPC], IMI-2, GES, derivatives), but all effectively hydrolyze carbapenems and are partially inhibited by clavulanic acid (2). KPCs are the most clinically common enzymes in this group. The first KPC producer (KPC-2 in K. pneumoniae) was identified in 1996 in the eastern United States (5).Within a few years, KPC producers had spread globally and have been described across the contiguous United States (still mostly in eastern coast states) and, in particular, in Puerto Rico, Colombia, Greece, Israel, and the People’s Republic of China (6,7) (Figure 1). Outbreaks of KPC producers also have been reported in many European countries and in South America (6,7) (Figure 1).
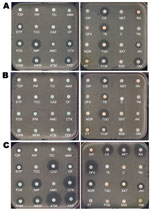
KPC producers have been reported, mostly from nosocomial K. pneumoniae isolates and to a much lesser extent from E. coli (especially in Israel) and from other enterobacterial species (6). A single K. pneumoniae clone (sequence type [ST]-258) was identified extensively worldwide, indicating that it may have contributed to the spread of the blaKPC genes (8).Within a given geographic location, several KPC clones are disseminating that differ by multilocus sequence type; additional β-lactamase content; and by size, number, and structure of plasmids, but the blaKPC genes are associated with a single genetic element (transposon Tn4401) (8). Although community-acquired KPC producers have been reported, they are rare, with the exception of isolates from Israel a few years ago (6).The level of resistance to carbapenems of KPC producers may vary markedly; ertapenem is the carbapenem that has the lowest activity (5–7), (Table 1). KPC producers are usually multidrug resistant (especially to all β-lactams), and therapeutic options for treating KPC-related infections remain limited (6) (Figure 2, panel A). Death rates attributed to infections with KPC producers are high (>50%) (9–11).
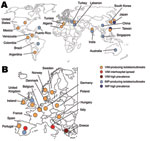
Class B metallo-β-lactamases (MBLs) are mostly of the Verona integron–encoded metallo-β-lactamase (VIM) and IMP types and, more recently, of the New Delhi metallo-β-lactamase-1 (NDM-1) type (2,12).The first acquired MBL, IMP-1, was reported in Serratia marcescens in Japan in 1991 (13). Since then, MBLs have been described worldwide (2,12) (Figure 3). Endemicity of VIM- and IMP-type enzymes has been reported in Greece, Taiwan, and Japan (2,12), although outbreaks and single reports of VIM and IMP producers have been reported in many other countries (Figure 3). These enzymes hydrolyze all β-lactams except aztreonam (12).Their activity is inhibited by EDTA but not by clavulanic acid (12). Most MBL producers are hospital acquired and multidrug-resistant K. pneumoniae (2,12). Resistance levels to carbapenems of MBL producers may vary (Table 1). Death rates associated with MBL producers range from 18% to 67% (14).
Discovered in 2008 in Sweden from an Indian patient hospitalized previously in New Delhi (15), NDM-1–positive Enterobacteriaceae are now the focus of worldwide attention (15–17). Since mid-August 2010, NDM-1 producers have been identified on all continents except in Central and South America with, in most of the cases, a direct link with the Indian subcontinent (17) (Figure 4). Few cases have been reported from the United States and Canada (17). Recent findings suggest that the Balkan states and the Middle East may act as secondary reservoirs of NDM-1 producers (17) (Figure 4).
In contrast to several other carbapenemase genes, the blaNDM-1 gene is not associated with a single clone but rather with nonclonally related isolates and species (16,17). It has been identified mostly in E. coli and K. pneumoniae and to a lesser extent in other enterobacterial species (16,17). The level of resistance to carbapenems of NDM-1 producers may vary (Table 1). Plasmids carrying the blaNDM-1 gene are diverse and can harbor a high number of resistance genes associated with other carbapenemase genes (oxacillinase-48 [OXA-48] types, VIM types), plasmid-mediated cephalosporinase genes, ESBL genes, aminoglycoside resistance genes (16S RNA methylases), macrolide resistance genes (esterase), rifampin (rifampin-modifying enzymes) and sulfamethoxazole resistance genes as a source of multidrug resistance and pandrug resistance (16,17) (Figure 2, panel B). The association of such a high number of resistance genes in single isolates has been rarely observed, even among the other carbapenemase producers. Many NDM-1 producers remain susceptible only to tigecycline, colistin (Figure 2, panel B), and to a lesser extent fosfomycin (16,17).
Compared with other carbapenemases, NDM-1 has several characteristics that are deeply disconcerting for public health worldwide. These characteristics are 1) occurrence of the blaNDM-1 gene not in a single species but in many unrelated species and its spread in the environment, at least in the Indian subcontinent (18); 2) frequent acquisition by K. pneumoniae, a typical nosocomial pathogen, but also by E. coli that is by far the main (community-acquired) human pathogen; and 3) size of the reservoir—the Indian subcontinent has >1.4 billion persons. In certain areas in Pakistan, <20% of the population may carry NDM-1 producers (P. Nordmann, unpub. data).
Of particular concern, NDM-1 has been identified in E. coli ST-type 131 as a source of community-acquired infection (19), an ST type that is known to mobilize efficiently the ESBL CTX-M-15 worldwide (20). E. coli is the most common cause of diarrhea in children in India. Therefore, this organism may increase the risk of drug-resistant strains being released into the environment and further spread among humans. Accordingly, NDM-1 producers have been recently identified in tap and environmental water in New Delhi, among many unrelated gram-negative species (18).
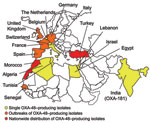
The first identified OXA-48 producer was from a K. pneumoniae strain isolated in Turkey in 2003 (21). Since then, OXA-48 producers have been extensively reported from Turkey as a source of nosocomial outbreaks (22–26). Their worldwide distribution now includes countries in Europe, in the southern and eastern part of the Mediterranean Sea, and Africa (21–26) (Figure 5). OXA-48 producers have not been reported from the United States and Canada. A point mutant analog of OXA-48, OXA-181, with similar carbapenemase activity, has been identified in strains from India or of Indian origin (27,28). There is an increasing trend of identification of OXA-48 producers in countries such as France, Germany, Spain, the Netherlands, and the United Kingdom through transfer of hospitalized patients from disease-endemic areas that are the source of hospital outbreaks (Figure 5).
Several OXA-48–producing clones have been identified, and dissemination of this resistance trait is associated with a 62.5-kb plasmid (previously identified as a plasmid of ≈70 kb) (22). OXA-48/OXA-181 are peculiar because they weakly hydrolyze carbapenems and broad-spectrum cephalosporins, such as ceftazidime, and aztreonam (21,27), (Figure 2, panel C). Their activity is not inhibited by EDTA or clavulanic acid (resistance to amoxicillin/clavulanic acid; Figure 2, panel C). Although reported in various enterobacterial species, OXA-48 producers are mostly identified in K. pneumoniae and E. coli, and the level of resistance to carbapenems is usually higher when ESBL and permeability defects are associated (22–28), (Table 1). The OXA-48–type producers are likely the most difficult carbapenemase producers to be identified. Thus, their true prevalence could be underestimated. The attributed mortality rate from infections with OXA-48 producers is unknown.
The detection of carbapenemase producers in clinical infections is based first on susceptibility testing results obtained by disk diffusion or by automated systems (29). The Clinical and Laboratory Standards Institute (CLSI; Wayne, PA, USA) breakpoints of carbapenems have been lowered substantially in 2010 for a better detection of carbapenem-resistant isolates and carbapenemase producers (Table 2). The CLSI breakpoints of carbapenems are now lower than those of the European guidelines (Table 2). Applying the CLSI breakpoints is all that is needed for making treatment decisions according to CLSI recommendations. Special tests for carbapenemase detection are recommended for epidemiology and infection issues.
However, low-level resistance and even susceptibility to carbapenems have been observed for producers of any type of carbapenemases (Table 1). We believe, as do others (30), that the search for carbapenemase producers should be made for any enterobacterial isolates with decreased susceptibility to carbapenems. Our opinion is based on the paucity of clinical experience for treating infections caused by carbapenemase producers, on the unknown level of carbapenemase production in the site of the infection in vivo, and on the possibility of selecting in vivo for strains with increased levels of resistance to carbapenems and additional mechanisms of carbapenem resistance (carbapenemase, outer-membrane permeability defects).
Specific tests may help identify phenotypically a carbapenemase activity. The modified Hodge test based on in vivo production of carbapenemase has been suggested for detecting carbapenemase producers (29,31,32). However, this test is time consuming and may lack specificity (high-level AmpC producers) and sensitivity (weak detection of NDM producers) (27,29). This test may be useful for detecting KPC and OXA-48 producers (P. Nordmann, unpub. data). Boronic acid–based inhibition testing is reported to be specific for KPC detection in K. pneumoniae when performed with imipenem or meropenem but not with ertapenem if corresponding isolates co-produce a plasmid-mediated AmpC β-lactamase (29,30). The Etest MBL strip (bioMérieux, Solna, Sweden) is one of the methods advocated for detecting MBL producers on the basis of inhibition of MBL activity by EDTA (12). The Etest MBL, using imipenem and imipenem/EDTA, is efficient for detection of MBL producers with high resistance (12), but may be deficient for detecting MBL producers with low resistance to imipenem. No inhibition test is available for detection of OXA-48/OXA-181 producers.
Spectrophotometric assay is needed for detecting carbapenemase activity. However, this assay is time consuming, requires specific training, and does not easily discriminate between different types of carbapenemases.
The standard for identification of carbapenemases is based on use of molecular techniques, mostly PCR (29,33). A list of primers of the most prevalent carbapenemase genes identified in Enterobacteriaceae is shown in Table 3 (34). Standard conditions may be used for PCR-based detection (34). PCR performed on colonies may give results within 4–6 hours with excellent sensibility and specificity. Similarly, other molecular techniques, such as the Check-Points DNA technology, are useful for this purpose (35). Sequencing of PCR products may be of interest mostly for epidemiologic purposes. The main disadvantages of molecular-based technologies for detection of carbapenemases are their cost, the requirement of trained personal, and the absence of detection of any novel carbapenemase gene. Thus, there is an urgent need for an inexpensive, rapid, sensitive, and specific test for detection of carbapenemase activity.
The prevention of spread of carbapenemase producers relies on early detection of carriers (29,33). Patients who undergo screening should include patients who were hospitalized while abroad and then transferred to another country, and patients at risk (e.g., patients in intensive care units, transplant patients, immunocompromised patients). Screened patients should be kept in strict isolation before obtaining results of the screening (at least 24–48 hours). Because the reservoir of carbapenemase producers remains the intestinal flora, fecal and rectal swab specimens are adequate for performing this screening. Those specimens may be plated directly on screening media.
There is no universal screening medium able to detect all types of carbapenemase producers with high sensitivity and high specificity, however. Agar plates containing imipenem at a concentration of 1 mg/L have been proposed for screening only KPC producers (36). We have demonstrated that a culture medium designed to screen for ESBL producers (ChromID ESBL; bioMérieux, La-Balme-Les-Grotte, France) may be used also for screening carbapenemase producers. Although this medium may lack specificity (co-detection of ESBL producers), its sensitivity is higher than a culture medium designed to screen for carbapenemase producers (CHROMagar KPC; CHROMagar, Paris, France) (33,37). The main problem remains detection of OXA-48 producers that are susceptible to cephalosporins and have low-level resistance to carbapenems when not co-producing an ESBL (Figure 2, panel C) (37). None of these culture media detect those OXA-48 producers (37).
After this screening procedure, carbapenemase producers may be identified according to the techniques described above (antibacterial drug susceptibility testing, molecular techniques). Recently, PCR-based techniques performed directly on fecal specimens have been proposed for detection of KPC and NDM-1 producers.
Carbapenemase producers in Enterobacteriaceae are not the source of specific types of clinical infections. The role of these bacteria is related to the difficult-to-treat infections rather than to expression of specific virulence traits.
We believe we are now at the edge of 2 concomitant epidemics of carbapenemase producers worldwide. The first epidemic will be caused mainly by carbapenemase producers in E. coli as a source of community-acquired infections. These carbapenemases are thus far primarily of the NDM and of the OXA-48 types. A few published reports of community-acquired infections caused by carbapenemase producers are available, but it is more likely that the numbers of cases in disease-endemic areas are already high. The example of the spread of ESBL producers in the community within the past 10 years shows us that a high rate of carbapenemase producers in E. coli may be reached rapidly worldwide. As opposed to a viral epidemic, such as pandemic (H1N1) 2009, the epidemic of carbapenemase producers cannot stop spontaneously. Such community-based outbreaks will be difficult to control. Modulation of the factors that enhance spread of carbapenemase producers in the community is difficult because these factors are multiple and are associated with lack of hygiene, overuse and over-the-counter use of antibacterial drugs, and increased worldwide travel. In addition, many carbapenemase producers carry unrelated drug-resistance determinants. Therefore, selection pressure with structurally unrelated antibacterial drugs (not only β-lactams) may contribute to their spread.
We cannot predict either the speed of diffusion of those carbapenemase producers in the community or their prevalence at a steady state (5%–50%?). The actual prevalence of carbapenemase producers is still unknown because many countries that are likely to be their main reservoirs have not established any search protocol for their detection. The prevalence may substantially differ, depending on the country, as known with the current prevalence rate of ESBL producers in E. coli. The prevalence is estimated to be 3%–5% in France and >80% in India (38).
The second epidemic will likely be caused mainly by nosocomial carbapenemase producers in K. pneumoniae of all types (KPC, IMP, VIM, NDM, and OXA-48). It is likely that in certain countries high rates of different types of carbapenemase producers may already exist, for example, in Greece (VIM and KPC) and in the Indian subcontinent (NDM, KPC, OXA-181). K. pneumoniae will play a major role because it has been repeatedly identified to be the most common enterobacterial species for spreading ESBL genes in health care facilities during the past 30 years. It may play the same role for spreading carbapenemase producers in patients with identical risk factors (patients receiving broad-spectrum antibiotherapy, patients in intensive care units, immunocompromised patients, transplant patients, surgical patients). Early identification of carbapenemase producers in clinical infections, at the carriage state, or both, is therefore mandatory to prevent development of those hospital-based outbreaks. We believe we still can efficiently prevent emergence of hospital-based outbreaks of carbapenemase producers. A similar strategy has been implemented in northern European countries for containment of hospital-acquired methicillin-resistant Staphylococcus aureus, which has been useful.
The dearth of novel antibacterial drugs in the pipeline means that we must conserve the efficacy of existing antibacterial drugs as much as possible. Carbapenemase producers in Enterobacteriaceae are different from other multidrug-resistant bacteria in that they are susceptible to few (if any) antibacterial drugs (39).
No vaccines are readily available for preventing infections with carbapenemase producers. This finding is particularly true for E. coli, which is part of the human intestinal flora. Therefore, everything must be done to prevent infections as common as pyelonephritis from becoming life threatening because of the lack of any effective treatment.
Dr Nordmann is professor of medical microbiology at South-Paris Medical School, chief of the Department of Clinical Microbiology at Bicêtre Hospital, and head of National Institute of Health and Medical Research (INSERM) research unit 914, Emerging Antibiotic Resistance, Le Kremlin-Bicêtre, France. His research focuses on molecular mechanisms of antibiotic resistance and their clinical implication.
Acknowledgments
We thank Sandrine Barnabeu, Amélie Carrër, and Gaëlle Cuzon for collecting much of the data presented in this review.
This work was funded by grants from the National Institute of Health and Medical Research (INSERM) research unit 914 and by the Ministère de l’Education Nationale et de la Recherche (UPRES-EA3539), Université Paris XI, Paris, France.
References
- Pitout JD, Laupland KB. Extended-spectrum β-lactamase–producing Enterobacteriaceae: an emerging public-health concern. Lancet Infect Dis. 2008;8:159–66. DOIPubMedGoogle Scholar
- Queenan AM, Bush K. Carbapenemases: the versatile β-lactamases. Clin Microbiol Rev. 2007;20:440–58. DOIPubMedGoogle Scholar
- Naas T, Nordmann P. Analysis of a carbapenem-hydrolyzing class A β-lactamase from Enterobacter cloacae and of its LysR-type regulatory protein. Proc Natl Acad Sci U S A. 1994;91:7693–7. DOIPubMedGoogle Scholar
- Giske CG, Sundsfjord AS, Kahlmeter G, Woodford N, Nordmann P, Paterson DL, Redefining extended-spectrum β-lactamase: balancing science and clinical need. J Antimicrob Chemother. 2009;63:1–4. DOIPubMedGoogle Scholar
- Yigit H, Queenan AM, Anderson GJ, Domenech-Sanchez A, Biddle JW, Steward CD, Novel carbapenem-hydrolyzing β-lactamase KPC-1 from a carbapenem-resistant strain of Klebsiella pneumoniae. Antimicrob Agents Chemother. 2001;45:1151–61. DOIPubMedGoogle Scholar
- Nordmann P, Cuzon G, Naas T. The real threat of Klebsiella pneumoniae carbapenemase-producing bacteria. Lancet Infect Dis. 2009;9:228–36. DOIPubMedGoogle Scholar
- Navon-Venezia S, Leavitt A, Schwaber MJ, Rasheed JK, Srinivasan A, Patel JB, First report on a hyperepidemic clone of KPC-3–producing Klebsiella pneumoniae in Israel genetically related to a strain causing outbreaks in the United States. Antimicrob Agents Chemother. 2009;53:818–20. DOIPubMedGoogle Scholar
- Cuzon G, Naas T, Truong H, Villegas MV, Wisell KT, Carmeli Y, Worldwide diversity of Klebsiella pneumoniae that produce β-lactamase blaKPC-2 gene. Emerg Infect Dis. 2010;16:1349–56. DOIPubMedGoogle Scholar
- Borer A, Saidel-Odes L, Riesenberg K, Eskira S, Peled N, Nativ R, Attributable mortality rate for carbapenem-resistant Klebsiella pneumoniae bacteremia. Infect Control Hosp Epidemiol. 2009;30:972–6. DOIPubMedGoogle Scholar
- Patel G, Huprikar S, Factor SH, Jenkins SG, Calfee DP. Outcomes of carbapenem-resistant Klebsiella pneumoniae infection and the impact of antimicrobial and adjunctive therapies. Infect Control Hosp Epidemiol. 2008;29:1099–106. DOIPubMedGoogle Scholar
- Schwaber MJ, Klarfeld-Lidji S, Navon-Venezia S, Schwartz D, Leavitt A, Carmeli Y. Predictors of carbapenem-resistant Klebsiella pneumoniae acquisition among hospitalized adults and effect of acquisition on mortality. Antimicrob Agents Chemother. 2008;52:1028–33. DOIPubMedGoogle Scholar
- Walsh TR, Toleman MA, Poirel L, Nordmannn P. Metallo-β-lactamases: the quiet before the storm? Clin Microbiol Rev. 2005;18:306–25. DOIPubMedGoogle Scholar
- Ito H, Arakawa Y, Ohsuka S, Wacharotayankun R, Kato N, Ohta M. Plasmid-mediated dissemination of the metallo-β-lactamase gene blaIMP among clinically isolated strains of Serratia marcescens. Antimicrob Agents Chemother. 1995;39:824–9.PubMedGoogle Scholar
- Daikos GL, Petrikkos P, Psichogiou M, Kosmidis C, Vryonis E, Skoutelis A, Prospective observational study of the impact of VIM-1 metallo-β-lactamase on the outcome of patients with Klebsiella pneumoniae bloodstream infections. Antimicrob Agents Chemother. 2009;53:1868–73. DOIPubMedGoogle Scholar
- Yong D, Toleman MA, Giske CG, Cho HS, Sundman K, Lee K, Characterization of a new metallo-β-lactamase gene, blaNDM-1, and a novel erythromycin esterase gene carried on a unique genetic structure in Klebsiella pneumoniae sequence type 14 from India. Antimicrob Agents Chemother. 2009;53:5046–54. DOIPubMedGoogle Scholar
- Kumarasamy KK, Toleman MA, Walsh TR, Bagaria J, Butt F, Balakrishnan R, Emergence of a new antibiotic resistance mechanism in India, Pakistan, and the UK: a molecular, biological, and epidemiological study. Lancet Infect Dis. 2010;10:597–602. DOIPubMedGoogle Scholar
- Nordmann P, Poirel L, Toleman MA, Walsh TR. Does broad-spectrum β-lactam resistance due to NDM-1 herald the end of the antibiotic era for treatment of infections caused by Gram-negative bacteria? J Antimicrob Chemother. 2011;66:689–92. DOIPubMedGoogle Scholar
- Walsh TR, Weeks J, Livermore DM, Toleman MA. Dissemination of NDM-1 positive bacteria in the New Delhi environment and its implications for human health: an environmental point prevalence study. Lancet Infect Dis. 2011;11:355–62. DOIPubMedGoogle Scholar
- Poirel L, Hombrouk-Alet C, Freneaux C, Bernabeu S, Nordmann P. Global spread of New Delhi metallo-β-lactamase 1. Lancet Infect Dis. 2010;10:832. DOIPubMedGoogle Scholar
- Coque TM, Novais A, Carattoli A, Poirel L, Pitout J, Peixe L, Dissemination of clonally related Escherichia coli strains expressing extended-spectrum β-lactamase CTX-M-15. Emerg Infect Dis. 2008;14:195–200. DOIPubMedGoogle Scholar
- Poirel L, Héritier C, Tolün V, Nordmann P. Emergence of oxacillinase-mediated resistance to imipenem in Klebsiella pneumoniae. Antimicrob Agents Chemother. 2004;48:15–22. DOIPubMedGoogle Scholar
- Carrër A, Poirel L, Yilmaz M, Akan OA, Feriha C, Cuzon G, Spread of OXA-48–encoding plasmid in Turkey and beyond. Antimicrob Agents Chemother. 2010;54:1369–73. DOIPubMedGoogle Scholar
- Cuzon G, Ouanich J, Gondret R, Naas T, Nordmann P. Outbreak of OXA-48–positive carbapenem-resistant Klebsiella pneumoniae isolates in France. Antimicrob Agents Chemother. 2011;55:2420–3. DOIPubMedGoogle Scholar
- Moquet O, Bouchiat C, Kinana A, Seck A, Arouna O, Bercion R, Class D OXA-48 carbapenemase in multidrug-resistant enterobacteria, Senegal. Emerg Infect Dis. 2011;17:143–4. DOIPubMedGoogle Scholar
- Benouda A, Touzani O, Khairallah MT, Araj GF, Matar GM. First detection of oxacillinase-mediated resistance to carbapenems in Klebsiella pneumoniae from Morocco. Ann Trop Med Parasitol. 2010;104:327–30. DOIPubMedGoogle Scholar
- Poirel L, Ros A, Carrër A, Fortineau N, Carricajo A, Berthelot P, Cross-border transmission of OXA-48–producing Enterobacter cloacae from Morocco to France. J Antimicrob Chemother. 2011;66:1181–2. DOIPubMedGoogle Scholar
- Castanheira M, Deshpande LM, Mathai D, Bell JM, Jones RN, Mendes RE. Early dissemination of NDM-1– and OXA-181–producing Enterobacteriaceae in Indian hospitals: report from the SENTRY Antimicrobial Surveillance Program, 2006–2007. Antimicrob Agents Chemother. 2011;55:1274–8. DOIPubMedGoogle Scholar
- Kalpoe JS, Al Naiemi N, Poirel L, Nordmann P. Detection of an Ambler class D OXA-48–type β-lactamase in a Klebsiella pneumoniae strain in The Netherlands. J Med Microbiol. 2011;60:677–8. DOIPubMedGoogle Scholar
- Miriagou V, Cornaglia G, Edelstein M, Galani I, Giske CG, Gniadkowski M, Acquired carbapenemases in Gram-negative bacterial pathogens: detection and surveillance issues. Clin Microbiol Infect. 2010;16:112–22. DOIPubMedGoogle Scholar
- Thomson KS. Extended-spectrum β-lactamase, AmpC and carbapenemase issues. J Clin Microbiol. 2010;48:1019–25. DOIPubMedGoogle Scholar
- Centers for Disease Control and Prevention. Guidance for control of infections with carbapenem-resistant or carbapenemase-producing Enterobacteriaceae in acute care facilities. MMWR Morb Mortal Wkly Rep. 2009;58:256–60.PubMedGoogle Scholar
- Galani I, Rekatsina PD, Hatzaki D, Plachouras D, Souli M, Giamarellou H. Evaluation of different laboratory tests for the detection of metallo-β-lactamase production in Enterobacteriaceae. J Antimicrob Chemother. 2008;61:548–53. DOIPubMedGoogle Scholar
- Nordmann P, Poirel L, Carrër A, Toleman MA, Walsh TR. How to detect NDM-1 producers. J Clin Microbiol. 2011;49:718–21. DOIPubMedGoogle Scholar
- Poirel L, Walsh TR, Cuvillier V, Nordmann P. Multiplex PCR for detection of acquired carbapenemase genes. Diagn Microbiol Infect Dis. 2011;70:119–23. DOIPubMedGoogle Scholar
- Naas T, Cuzon G, Bogaerts P, Glupczynski Y, Nordmann P. Evaluation of a DNA microarray (Check-MDR CT102) for rapid detection of TEM, SHV, and CTX-M extended-spectrum β-lactamases and of KPC, OXA-48, VIM, IMP, and NDM-1 carbapenemases. J Clin Microbiol. 2011;49:1608–13. DOIPubMedGoogle Scholar
- Adler A, Navon-Venezia S, Moran-Gilad J, Marcos E, Schwartz D, Carmeli Y. Laboratory and clinical evaluation of screening agar plates for the detection of carbapenem-resistant enterobacteriaceae from surveillance rectal swabs. J Clin Microbiol. 2011;49:2239–42. DOIPubMedGoogle Scholar
- Carrër A, Fortineau N, Nordmann P. Use of ChromID ESBL medium for detecting carbapenemase-producing Enterobacteriaceae. J Clin Microbiol. 2010;48:1913–4. DOIPubMedGoogle Scholar
- Jean SS, Hsueh PR. High burden of antibimicrobial resistance in Asia. Int J Antimicrob Agents. 2011;37:291–5. DOIPubMedGoogle Scholar
- Falagas ME, Karageorgopoulos DE, Nordmann P. Therapeutic options with Enterobacteriaceae producing carbapenem-hydrolyzing enzymes. Future Microbiol. 2011;6:653–6. DOIPubMedGoogle Scholar
Figures
Tables
Cite This ArticleTable of Contents – Volume 17, Number 10—October 2011
| EID Search Options |
|---|
|
|
|
|
|
|

Please use the form below to submit correspondence to the authors or contact them at the following address:
Patrice Nordmann, Service de Bactériologie-Virologie, Hôpital de Bicêtre, 78 Rue du Général Leclerc, 94275 K-Bicêtre, France
Top