Volume 21, Number 5—May 2015
Research
Molecular Epidemiology of Plasmodium falciparum Malaria Outbreak, Tumbes, Peru, 2010–2012
Cite This Article
Citation for Media
Abstract
During 2010–2012, an outbreak of 210 cases of malaria occurred in Tumbes, in the northern coast of Peru, where no Plasmodium falciparum malaria case had been reported since 2006. To identify the source of the parasite causing this outbreak, we conducted a molecular epidemiology investigation. Microsatellite typing showed an identical genotype in all 54 available isolates. This genotype was also identical to that of parasites isolated in 2010 in the Loreto region of the Peruvian Amazon and closely related to clonet B, a parasite lineage previously reported in the Amazon during 1998–2000. These findings are consistent with travel history of index case-patients. DNA sequencing revealed mutations in the Pfdhfr, Pfdhps, Pfcrt, and Pfmdr1 loci, which are strongly associated with resistance to chloroquine and sulfadoxine/pyrimethamine, and deletion of the Pfhrp2 gene. These results highlight the need for timely molecular epidemiology investigations to trace the parasite source during malaria reintroduction events.
During the past decade, remarkable progress in malaria control has been achieved globally (1). As low-risk areas progress toward the preelimination phase of malaria elimination (http://www.who.int/malaria/areas/elimination/overview/en/), new challenges are posed by risk for reintroduction of parasites into areas where malaria transmission was interrupted (2). Human movement from malaria-endemic regions could facilitate outbreaks in areas where malaria had been eliminated (2,3). Molecular epidemiology tools have been used to investigate the sources of malaria reintroduction (4,5). Use of these tools enables rapid characterization of potentially pathogenic or multidrug-resistant strains before they become adapted and expand to other non–malaria-endemic areas where anopheline vectors are present (6–9).
In Peru, malaria reemerged in the 1990s and the number of cases peaked at ≈160,000 cases in 1998 (10). Most reported cases had occurred in the Amazon Basin (Loreto region) and areas in the northern Pacific coast of Peru, including the Tumbes and Piura regions. In vivo efficacy studies conducted during 1998–2000 revealed different patterns of drug resistance between parasites in the Amazon region and coastal areas (11,12). Although parasites from the eastern Amazon region were resistant to chloroquine and sulfadoxine/pyrimethamine, parasites from the northern Pacific coast were resistant to chloroquine but remained sensitive to sulfadoxine/pyrimethamine (11–13). In 2001, artesunate/mefloquine combination therapy was introduced in the Peruvian Amazon while artesunate–sulfadoxine/pyrimethamine remained in use in the northern Pacific coastal region (12).
After 2005, changes in drug policy and increased vector control efforts in Peru led to a drastic reduction in the number of malaria cases in the country. A major accomplishment was the interruption of Plasmodium falciparum transmission in the northern Pacific coast; no autochthonous malaria case has been reported since 2006. However, in October of 2010, the Regional Health Directorate in Tumbes received reports of 2 cases of P. falciparum malaria. An outbreak investigation confirmed the P. falciparum malaria epidemic in Tumbes. This outbreak continued to spread through 2012, when the last case of P. falciparum malaria was reported. Epidemiology investigations identified 2 index case-patients among military personnel stationed in Tumbes; surveillance activities conducted during the outbreak investigation suggested that these patients potentially acquired P. falciparum infection while in the Peruvian Amazon. We therefore hypothesized that a detailed genetic characterization of the parasite populations isolated during this outbreak might provide a better understanding of the source and main biological features of the parasite responsible for the reintroduction of malaria into Tumbes.
Previous genetic analyses of P. falciparum strains collected at the peak of the malaria epidemic, 1999–2000, revealed at least 5 distinct clonal lineages (clonets A–E), as defined by genotyping of 7 neutral microsatellite loci (14). These clonets, which were distributed in different areas of Peru, exhibited distinct patterns of mutations based on sequencing of the Pfcrt, Pfmdr1, Pfdhps, and Pfdhfr genes (14). Considering these historical data, we tested the following hypotheses. First, if the P. falciparum outbreak in Tumbes was caused by bottlenecked parasites from the coastal region, the parasites causing this outbreak would be genetically similar or closely related to clonet E, which was the only lineage found in the northern Pacific coast during 1999–2000. Second, if the parasite was introduced from the Peruvian Amazon, then the parasites causing this outbreak would be related to clonets A, B, C, or D. Third, if these parasites were introduced from outside Peru, they may have different molecular signatures.
Study Area and Sample Collection
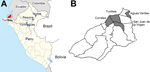
The Tumbes region is located in the Pacific northwestern part of Peru near the border with Ecuador (Figure 1, panel A). Tumbes is divided into 13 districts with a total surface of 4,670 km2 and a population of ≈228,227 (Figure 1, panel B). In the late 1990s, malaria transmitted by P. vivax and P. falciparum was highly endemic to the region (11,15). In Tumbes, Anopheles albimanus mosquitoes predominate, unlike in the Amazon region, where An. darlingi mosquitoes predominate. In the 2000s, malaria incidence was drastically reduced, and the parasite predominance shifted to P. vivax, which is seasonal in this area, peaking during the rainy season (February–June). The last autochthonous case of P. falciparum malaria in Tumbes was reported in 2006.
In October 2010, after the Regional Health Directorate received reports of 2 cases of P. falciparum malaria, an outbreak response team led by the Ministry of Health of Peru with the support of the US Naval Medical Research Unit No. 6 (NAMRU-6), the US Centers for Disease Control and Prevention (CDC), and the US Agency for International Development conducted an outbreak investigation and response. As part of these activities, malaria cases were detected by passive surveillance of febrile patients seeking treatment at local health facilities or at the regional referral hospital. Additional cases were detected by various case-finding activities conducted in areas where laboratory-confirmed P. falciparum malaria cases were found. For all patients, thick and thin smears stained with Giemsa 10% were examined for parasites. Slides were read at the local health facility and sent to the National Institute of Health in Peru (INS) and NAMRU-6 for species confirmation and quality control. Blood was spotted onto Whatman 3MM filter paper (GE Healthcare, Atlanta, GA, USA) and sent to INS and NAMRU-6. For some cases, whole blood was collected by venipuncture and shipped to INS and NAMRU-6. All biological samples were collected exclusively for the purpose of diagnosis, case investigation, and patient management as part of a public health intervention led by the Ministry of Health of Peru.
DNA Isolation and PCR Analysis
DNA was isolated from filter paper blood spots or whole blood samples by use of the QIAamp DNA Blood Mini Kit (QIAGEN, Valencia, CA, USA) as described elsewhere (16). Nested PCR was used to confirm P. falciparum infection for all patients in this study (17). Molecular analysis was performed at the NAMRU-6 laboratory in Lima, Peru. Selected samples were sent to CDC for further genetic characterization.
Microsatellite Analysis
Whole-genome amplified DNA (REPLI-g; QIAGEN) was used for microsatellite characterization. All P. falciparum–confirmed samples were assayed for 7 putatively neutral microsatellite loci. In studies in South America, 5 microsatellite loci have been used: TA1 (chromosome 6); poly α (chromosome 4); PfPK2 (chromosome 12); TA109 (chromosome 6), and 2490 (chromosome 10) (18–20). In addition to these markers, we also amplified the loci C2M34 (chromosome 2) and C3M69 (chromosome 3) (21). Primer sequences and PCR parameters for these loci have been described (14,16). Furthermore, all samples were assayed for 5 microsatellite loci that span ≈11 kb on chromosome 4 around Pfdhfr, 9 loci that span 17 kb on chromosome 8 around Pfdhps, 4 loci that span 11 kb on chromosome 7 around Pfcrt, and 6 loci that span 8 kb on chromosome 5 around Pfmdr1. The primers used to amplify microsatellite loci have been described (16,22). The amplification products were labeled with fluorescent dyes (HEX or FAM) and assayed for size on an 3130xl sequencer (Applied Biosystems, Foster City, CA, USA). The fragments were then scored by using GeneMapper software version 3.7 (Applied Biosystems) with default microsatellite settings, whereby bands of <500 relative fluorescence units were defined as background. Samples for which we obtained no amplification in some loci were reanalyzed to complete the haplotypes. Earlier, we had created haplotype identifiers for each of these genes on the basis of microsatellite loci that were nearby (16).
Genotyping of Markers of Drug Resistance
DNA isolates were sequenced for point mutations in Pfdhfr, Pfdhps, chloroquine resistance transporter (Pfcrt), and Pfmdr1. The methods used are described elsewhere (14,16,21,23,24).
Detection of P. falciparum Histidine-Rich Protein 2 Deletions
Two sets of primers were designed to amplify a 228-bp fragment of P. falciparum histidine-rich protein 2 (Pfhrp2) in a nested PCR. The outward forward primer was 5′-GGTTTCCTTCTCAAAAAATAAAG-3′, and the outward reverse primer was 5′-TCTACATGTGCTTGAGTTTCG-3′. The secondary reaction used 5′-GTATTATCCGCTGCCGTTTTTGCC-3′ (forward) and 5′-CTACACAAGTTATTATTAAATGCGGAA-3′ (reverse) primers. The cycling conditions were as follows: primary reaction at 95°C for 5 min; 30 cycles of 95°C for 30 s, 60°C for 30 s, 68°C for 30 s; and 68°C for 5 min; and secondary reaction at 95°C for 5 min; 30 cycles of 95°C for 30 s, 65°C for 30 s, 68°C for 30 s; and 68°C for 5 min.
Ethics Considerations
The activities were conducted in compliance with all applicable federal and international regulations governing the protection of human subjects. No informed consent was requested from the patients because all biological samples were collected as part of a public health intervention led by the Ministry of Health of Peru. All samples received by INS, CDC, or NAMRU-6 were coded, and no access to personal identifiable data was provided.
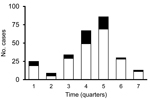
During October 2010–June 2012, a total of 210 cases of P. falciparum malaria were reported in Tumbes. Filter paper or whole blood samples were available for laboratory testing for 57 (27%) of the 210 patients. Figure 2 shows the temporal distribution of all 210 cases reported during the outbreak and the 57 cases that were included in this study. PCR confirmed P. falciparum monoinfection in 54 patients; P. vivax/P. falciparum mixed infections in the other 3 patients led to their exclusion from further analysis. To determine the clonal composition of the isolates, we genotyped 7 neutral microsatellite markers located in 6 different chromosomes. We observed virtually the same clonal lineage across all 54 isolates with the exception of 3 isolates that had alleles that were 3 bp shorter in loci TA1 and PfPK2 (Table 1). These results suggested that the outbreak originated from a single parasite source or from various sources of the same parasite population.
In a previous study, we found 5 distinct clonal lineages (clonets A–E) among P. falciparum isolates collected in the Peruvian Amazon (14). We compared the genotype of the outbreak parasite to genotypes in a historical microsatellite database and found that the genotype of the isolates from Tumbes closely resembled clonet B, a lineage found in the eastern Peruvian Amazon during 1999–2006 (14). However, the outbreak haplotype differed at 2 loci: C2M34 (232 vs. 226 bp) and C3M69 (134 vs. 149 bp) (Table 1; 14). On the basis of these results, we postulated that the isolates from Tumbes might be related to clonet B. Surveys and available patient records revealed that 7 malaria cases occurred among members of a military facility in Tumbes. At least 1 of these patients reported having traveled to Yurimaguas, a small town in the Peruvian Amazon ≈400 km southwest of the city of Iquitos. For this reason, we compared the genotype of the parasites from Tumbes with those from 23 samples collected during 2009–2010 from Yurimaguas (12 samples) and from the Requena District (11 samples), which is 250 km northeast of Yurimaguas in the Peruvian Amazon (Figure 1). For all 11 isolates from Requena and 1 from Yurimaguas, the microsatellite genotypes were identical to that of the isolates from Tumbes—we named this parasite BV1 variant (Table 1).
To gain further insight into the drug-resistance pattern of the P. falciparum BV1 variant, we performed DNA sequencing to characterize point mutations in Pfdhfr, Pfdhps, Pfcrt, and Pfmdr1, which have been associated with resistance to sulfadoxine/pyrimethamine and chloroquine. Similar to clonet B, all outbreak samples shared 437/540/581 Pfdhps, SVMNT Pfcrt, and 184/1034/1042/1246 Pfmdr1 genotypes. However, clonet B and the BV1 lineage differed in Pfdhfr haplotype. Whereas clonet B was 51I/108N/164L, the BV1 outbreak lineage and parasite isolate from Requena had the 50R/51I/108N Pfdhfr haplotype (Table 2; 14). Typing of microsatellite loci near the boundaries of the Pfdhfr and Pfmdr1 genes showed that the Bv1 variant belonged to the haplogroup DHFR-D1, which in a previous study was found to be a rare haplogroup in Iquitos in 2006–2007 (only found in 3 [5%] of 62 samples); and MDR-A1, which in a previous study was found to be abundant in Iquitos in 2006–2007 (frequency >50%) (16).
Because 40% of P. falciparum isolates from the Peruvian Amazon have major deletions of the genes coding for HRP2 and HRP3 (25), the most commonly used targets of rapid diagnostic tests (25), we investigated the presence of these deletions in these samples. All isolates in this outbreak lacked the Pfhrp2 gene (Table 2).
Our molecular epidemiology investigation identified a possible source of the P. falciparum parasites causing a major outbreak of malaria in Tumbes, Peru, a region that had been free of falciparum malaria since 2006. Our results suggested that this outbreak was caused by a single introduction of a parasite population that originated in the Loreto region of the Peruvian Amazon. These parasites had a chloroquine- and sulfadoxine/pyrimethamine–resistant mutation pattern. This study illustrates the value of molecular epidemiology tools during malaria outbreak investigations and malaria reintroduction events.
All P. falciparum samples available for testing belonged to a single genetic lineage, according to their nearly identical microsatellite genotypes. For 2 isolates, 2 alleles were slightly different sizes (169 vs. 172 bp). This variation could represent microevolution of parasites during this outbreak, which has been observed during outbreaks involving other microsporidia parasites (26).
Because one of the first patients reported in October 2010 in Tumbes had a history of travel to Loreto, we hypothesized that the parasite population causing this outbreak could be associated with parasites from Loreto. The genotype of the parasites from Tumbes was identical to that of 11 of 11 P. falciparum isolates collected from the Requena District in Loreto in 2010 and 1 of 12 P. falciparum isolates collected from Yurimaguas (V. Udhayakumar, pers. comm.). Further analysis revealed that the Tumbes genotype was closely related to clonet B, which we previously reported to have been introduced to Loreto from Brazil before the major malaria epidemic in the 1990s (14,16). Furthermore, the parasite from Tumbes was highly unrelated to clonet E, which was the only parasite population found in the northern coast of Peru before falciparum malaria was eliminated in this region (14). These results demonstrate that the outbreak of falciparum malaria in Tumbes was most likely caused by a single event, the introduction of a parasite population from Loreto.
Consistent with the neutral microsatellite findings, all other genetic markers tested supported the aforementioned conclusion. Included were the chloroquine-resistant SVMNT genotype, pyrimethamine-resistant genotype (Pfdhfr 50R, 51I, and 108N), sulfadoxine-resistant genotype (437G, 540E, and 581G), and deletion of the Pfhrp2 gene.
Several observations are relevant to the recent resurgence of malaria in Tumbes. First, this lineage has a new Pfdhfr triple mutant genotype (50R, 51I, 108N) that was mainly found outside of Peru in South America (14,16,21,27). This genotype seems to have been introduced into Peru during or after peak transmission of malaria within Peru because such genotypes were only reported in, not before, 2006 (14,16). Second, in the northern coast of Peru, artesunate–sulfadoxine/pyrimethamine combination therapy was being used for the treatment of P. falciparum malaria, while this newly introduced parasite strain had mutation patterns consistent with resistance to chloroquine and sulfadoxine/pyrimethamine. Coincidentally, malaria cases reported during this outbreak were treated with artesunate and mefloquine to decrease the risk for treatment failure. Therefore, the use of artesunate–sulfadoxine/pyrimethamine for the treatment of P. falciparum in the northern coast of Peru, as is currently recommended by treatment guidelines published by the Ministry of Health of Peru, may not be appropriate in this region because of the risk for malaria reintroduction from the Amazon region. Third, a region with no documented evidence of Pfhrp2-deleted parasites suddenly became populated with such a parasite strain, thereby making HRP2-based rapid tests inadequate diagnostic tools for this investigation. Misdiagnosis could have occurred if HRP2-based rapid tests were used as the primary diagnostic tool in this region.
In summary, this study provides experimental evidence of the value of timely molecular epidemiology investigations for pinpointing the source of P. falciparum reintroduction in areas working toward malaria elimination. Additionally, these data point out that future screening of military recruits (or any other migrant population from malaria-endemic areas such as the Amazon region) for the presence of malaria and provision of appropriate treatment can help prevent future reintroduction of malaria in areas from which it had been eliminated.
Dr. Baldeviano is the head of the Immunology and Vaccine Development Unit, Department of Parasitology, NAMRU-6. His research focuses on the use of genetic, immunologic, and high-throughput tools for the control and prevention of malaria.
Acknowledgments
We are grateful to all the personnel of the Ministry of Health in Tumbes and Lima who were involved in the response to the outbreak and who assisted with collection of patient information and samples. We also thank Jaime Chang and Guillermo Gonzalvez for their guidance and contributions to the control of the outbreak, and we acknowledge the assistance provided by the Center for Global Health of the Universidad Peruana Cayetano Heredia, which provided valuable logistical support to this study.
This work was funded by the US Department of Defense Armed Forces Health Surveillance Center, Global Emerging Infectious Systems Division, through the Malaria Pillar Sustainment grant awarded to A.G.L. In addition, this investigation was partly supported by the Amazon Malaria Initiative, which is financially supported by the US Agency for International Development. S.A. was supported by the American Society of Microbiology/CDC Postdoctoral Fellowship Program. The participation of A.G.L. in this investigation was sponsored by training grant NIH/FIC 2D43 TW007393 awarded to NAMRU-6 by the Fogarty International Center of the US National Institutes of Health.
References
- World Health Organization. Malaria; country profiles 2012 [cited 2014 Dec 1] http://www.who.int/malaria/publications/country-profiles/en/index.html
- Cotter C, Sturrock HJ, Hsiang MS, Liu J, Phillips AA, Hwang J, The changing epidemiology of malaria elimination: new strategies for new challenges. Lancet. 2013;382:900–11. DOIPubMedGoogle Scholar
- Webster-Kerr K, Peter Figueroa J, Weir PL, Lewis-Bell K, Baker E, Horner-Bryce J, Success in controlling a major outbreak of malaria because of Plasmodium falciparum in Jamaica. Trop Med Int Health. 2011;16:298–306. DOIPubMedGoogle Scholar
- Hanna JN, Ritchie SA, Eisen DP, Cooper RD, Brookes DL, Montgomery BL. An outbreak of Plasmodium vivax malaria in Far North Queensland, 2002. Med J Aust. 2004;180:24–8 .PubMedGoogle Scholar
- Arez AP, Snounou G, Pinto J, Sousa CA, Modiano D, Ribeiro H, A clonal Plasmodium falciparum population in an isolated outbreak of malaria in the Republic of Cabo Verde. Parasitology. 1999;118:347–55. DOIPubMedGoogle Scholar
- Djimdé AA, Dolo A, Ouattara A, Diakite S, Plowe CV, Doumbo OK. Molecular diagnosis of resistance to antimalarial drugs during epidemics and in war zones. J Infect Dis. 2004;190:853–5. DOIPubMedGoogle Scholar
- Samudio F, Santamaria AM, Obaldia N III, Pascale JM, Bayard V, Calzada JE. Prevalence of Plasmodium falciparum mutations associated with antimalarial drug resistance during an epidemic in Kuna Yala, Panama, Central America. Am J Trop Med Hyg. 2005;73:839–41 .PubMedGoogle Scholar
- Laserson KF, Petralanda I, Almera R, Barker RH Jr, Spielman A, Maguire JH, Genetic characterization of an epidemic of Plasmodium falciparum malaria among Yanomami Amerindians. J Infect Dis. 1999;180:2081–5. DOIPubMedGoogle Scholar
- Legrand E, Volney B, Lavergne A, Tournegros C, Florent L, Accrombessi D, Molecular analysis of two local falciparum malaria outbreaks on the French Guiana coast confirms the msp1 B-K1/varD genotype association with severe malaria. Malar J. 2005;4:26. DOIPubMedGoogle Scholar
- Aramburú Guarda J, Ramal Asayag C, Witzig R. Malaria reemergence in the Peruvian Amazon region. Emerg Infect Dis. 1999;5:209–15 . DOIPubMedGoogle Scholar
- Durand S, Marquino W, Cabezas C, Utz G, Fiestas V, Cairo J, Unusual pattern of Plasmodium falciparum drug resistance in the northwestern Peruvian Amazon region. Am J Trop Med Hyg. 2007;76:614–8 .PubMedGoogle Scholar
- Ruebush TK II, Neyra D, Cabezas C. Modifying national malaria treatment policies in Peru. J Public Health Policy. 2004;25:328–45. DOIPubMedGoogle Scholar
- Magill AJ, Zegarra J, Garcia C, Marquino W, Ruebush TK II. Efficacy of sulfadoxine-pyrimethamine and mefloquine for the treatment of uncomplicated Plasmodium falciparum malaria in the Amazon basin of Peru. Rev Soc Bras Med Trop. 2004;37:279–81. DOIPubMedGoogle Scholar
- Griffing SM, Mixson-Hayden T, Sridaran S, Alam MT, McCollum AM, Cabezas C, South American Plasmodium falciparum after the malaria eradication era: clonal population expansion and survival of the fittest hybrids. PLoS ONE. 2011;6:e23486. DOIPubMedGoogle Scholar
- Marquiño W, Ylquimiche L, Hermenegildo Y, Palacios AM, Falconi E, Cabezas C, Efficacy and tolerability of artesunate plus sulfadoxine-pyrimethamine and sulfadoxine-pyrimethamine alone for the treatment of uncomplicated Plasmodium falciparum malaria in Peru. Am J Trop Med Hyg. 2005;72:568–72 .PubMedGoogle Scholar
- Bacon DJ, McCollum AM, Griffing SM, Salas C, Soberon V, Santolalla M, Dynamics of malaria drug resistance patterns in the Amazon basin region following changes in Peruvian national treatment policy for uncomplicated malaria. Antimicrob Agents Chemother. 2009;53:2042–51. DOIPubMedGoogle Scholar
- Singh B, Bobogare A, Cox-Singh J, Snounou G, Abdullah MS, Rahman HA. A genus- and species-specific nested polymerase chain reaction malaria detection assay for epidemiologic studies. Am J Trop Med Hyg. 1999;60:687–92 .PubMedGoogle Scholar
- Anderson TJ, Su XZ, Bockarie M, Lagog M, Day KP. Twelve microsatellite markers for characterization of Plasmodium falciparum from finger-prick blood samples. Parasitology. 1999;119:113–25. DOIPubMedGoogle Scholar
- Anderson TJ, Haubold B, Williams JT, Estrada-Franco JG, Richardson L, Mollinedo R, Microsatellite markers reveal a spectrum of population structures in the malaria parasite Plasmodium falciparum. Mol Biol Evol. 2000;17:1467–82. DOIPubMedGoogle Scholar
- Echeverry DF, Nair S, Osorio L, Menon S, Murillo C, Anderson TJ. Long term persistence of clonal malaria parasite Plasmodium falciparum lineages in the Colombian Pacific region. BMC Genet. 2013;14:2. DOIPubMedGoogle Scholar
- McCollum AM, Mueller K, Villegas L, Udhayakumar V, Escalante AA. Common origin and fixation of Plasmodium falciparum dhfr and dhps mutations associated with sulfadoxine-pyrimethamine resistance in a low-transmission area in South America. Antimicrob Agents Chemother. 2007;51:2085–91. DOIPubMedGoogle Scholar
- Nair S, Nash D, Sudimack D, Jaidee A, Barends M, Uhlemann AC, Recurrent gene amplification and soft selective sweeps during evolution of multidrug resistance in malaria parasites. Mol Biol Evol. 2007;24:562–73. DOIPubMedGoogle Scholar
- Griffing S, Syphard L, Sridaran S, McCollum AM, Mixson-Hayden T, Vinayak S, pfmdr1 amplification and fixation of pfcrt chloroquine resistance alleles in Plasmodium falciparum in Venezuela. Antimicrob Agents Chemother. 2010;54:1572–9. DOIPubMedGoogle Scholar
- Mejia Torres RE, Banegas EI, Mendoza M, Diaz C, Bucheli ST, Fontecha GA, Efficacy of chloroquine for the treatment of uncomplicated Plasmodium falciparum malaria in Honduras. Am J Trop Med Hyg. 2013;88:850–4. DOIPubMedGoogle Scholar
- Gamboa D, Ho MF, Bendezu J, Torres K, Chiodini PL, Barnwell JW, A large proportion of P. falciparum isolates in the Amazon region of Peru lack pfhrp2 and pfhrp3: implications for malaria rapid diagnostic tests. PLoS ONE. 2010;5:e8091. DOIPubMedGoogle Scholar
- Hunter PR, Wilkinson DC, Lake IR, Harrison FC, Syed Q, Hadfield SJ, Microsatellite typing of Cryptosporidium parvum in isolates from a waterborne outbreak. J Clin Microbiol. 2008;46:3866–7. DOIPubMedGoogle Scholar
- Zhou Z, Griffing SM, de Oliveira AM, McCollum AM, Quezada WM, Arrospide N, Decline in sulfadoxine-pyrimethamine-resistant alleles after change in drug policy in the Amazon region of Peru. Antimicrob Agents Chemother. 2008;52:739–41. DOIPubMedGoogle Scholar
Figures
Tables
Cite This ArticleTable of Contents – Volume 21, Number 5—May 2015
| EID Search Options |
|---|
|
|
|
|
|
|
Please use the form below to submit correspondence to the authors or contact them at the following address:
G. Christian Baldeviano, US Naval Medical Research Unit No. 6, Av. Venezuela, Cuadra 36 S/N, Callao 2, Peru
Top