Disclaimer: Early release articles are not considered as final versions. Any changes will be reflected in the online version in the month the article is officially released.
Volume 32, Number 5—May 2026
Synopsis
Borna Disease Virus 1 as Cause of Fatal Meningoencephalomyelitis in Wild Hedgehogs, Germany, 2022–2025
Figure 8
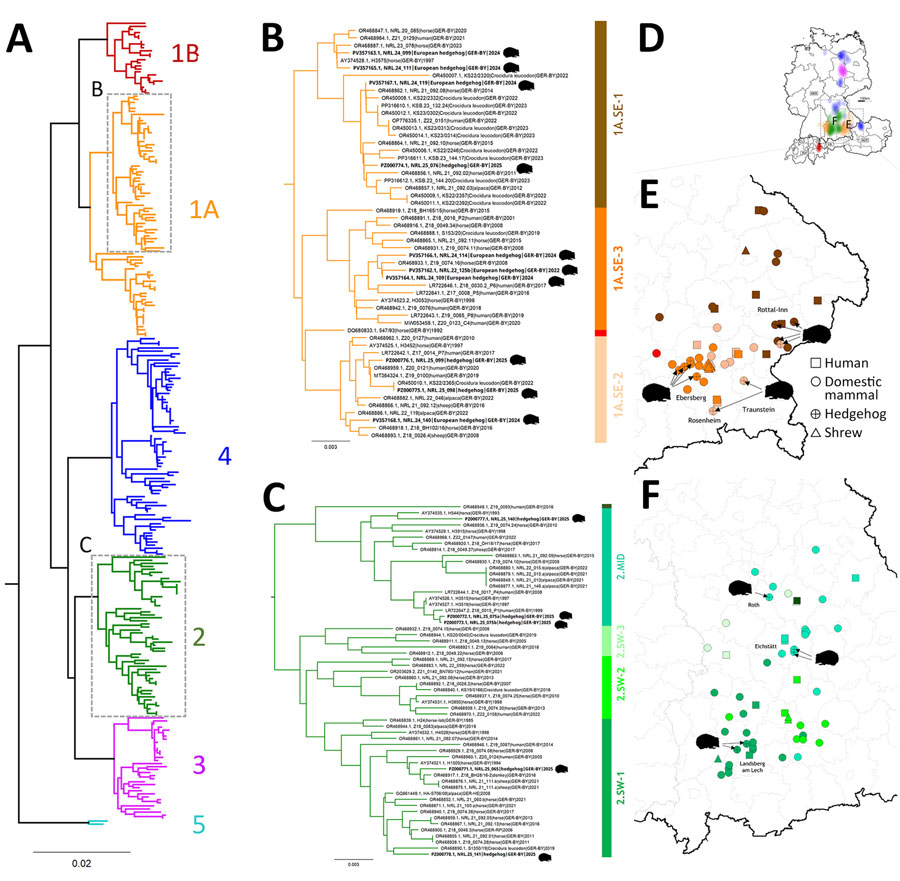
Figure 8. Phylogeographic analysis of Borna disease virus 1 (BoDV-1) infections in hedgehogs in Bavaria in study of the virus as cause of fatal meningoencephalomyelitis in wild hedgehogs, Germany, 2022–2025. A) Phylogenetic analysis of partial genomic BoDV-1 sequences (N, X, and P genes, encoding the nucleoprotein, accessory X protein, and phosphoprotein, respectively; 1,824 nucleotides, representing genome positions 54–1,877) of all 15 BoDV-1-infected hedgehogs in combination with 258 BoDV-1 sequences from naturally infected animals and humans available from public databases (1,9). Colors of tree branches represent BoDV-1 sequence clusters. Grey boxes mark the subtrees shown in detail in panels B and C. B, C) Detailed presentation of the subtree of cluster 1A, 1B (B), and of clusters 2–5 (C), which contain all 15 hedgehog-derived sequences (black text). Colors of vertical bars represent subclades of cluster 1A and 2 (1). D) BoDV-1-endemic area in Germany, Austria, Switzerland, and Liechtenstein (1). Colors represent the phylogenetic clusters shown in panel A. Licensed under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0). E, F) Detailed phylogeographic mapping of BoDV-1 cases from the phylogenetic subtree of cluster 1A (E) as shown in panel B or cluster 2 (F) as shown in panel C, to their distribution areas in Bavaria. Colors represent the different subclades. For data protection, human cases are mapped no more precisely than to the center of the administrative district of their origin.
References
- Ebinger A, Santos PD, Pfaff F, Dürrwald R, Kolodziejek J, Schlottau K, et al. Lethal Borna disease virus 1 infections of humans and animals—in-depth molecular epidemiology and phylogeography. Nat Commun. 2024;15:7908. DOIPubMedGoogle Scholar
- Malbon AJ, Dürrwald R, Kolodziejek J, Nowotny N, Kobera R, Pöhle D, et al. New World camelids are sentinels for the presence of Borna disease virus. Transbound Emerg Dis. 2022;69:451–64. DOIPubMedGoogle Scholar
- Schulze V, Große R, Fürstenau J, Forth LF, Ebinger A, Richter MT, et al. Borna disease outbreak with high mortality in an alpaca herd in a previously unreported endemic area in Germany. Transbound Emerg Dis. 2020;67:2093–107. DOIPubMedGoogle Scholar
- Vahlenkamp TW, Konrath A, Weber M, Müller H. Persistence of Borna disease virus in naturally infected sheep. J Virol. 2002;76:9735–43. DOIPubMedGoogle Scholar
- Schlottau K, Forth L, Angstwurm K, Höper D, Zecher D, Liesche F, et al. Fatal encephalitic Borna disease virus 1 in solid-organ transplant recipients. N Engl J Med. 2018;379:1377–9. DOIPubMedGoogle Scholar
- Korn K, Coras R, Bobinger T, Herzog SM, Lücking H, Stöhr R, et al. Fatal encephalitis associated with Borna disease virus 1. N Engl J Med. 2018;379:1375–7. DOIPubMedGoogle Scholar
- Niller HH, Angstwurm K, Rubbenstroth D, Schlottau K, Ebinger A, Giese S, et al. Zoonotic spillover infections with Borna disease virus 1 leading to fatal human encephalitis, 1999–2019: an epidemiological investigation. Lancet Infect Dis. 2020;20:467–77. DOIPubMedGoogle Scholar
- Weissenböck H, Bagó Z, Kolodziejek J, Hager B, Palmetzhofer G, Dürrwald R, et al. Infections of horses and shrews with Bornaviruses in upper Austria: a novel endemic area of Borna disease. Emerg Microbes Infect. 2017;6:
e52 . DOIPubMedGoogle Scholar - Böhmer MM, Haring VC, Schmidt B, Saller FS, Coyer L, Chitimia-Dobler L, et al. One Health in action: investigation of the first detected local cluster of fatal borna disease virus 1 (BoDV-1) encephalitis, Germany 2022. J Clin Virol. 2024;171:
105658 . DOIPubMedGoogle Scholar - Rubbenstroth D, Schlottau K, Schwemmle M, Rissland J, Beer M. Human bornavirus research: back on track! PLoS Pathog. 2019;15:
e1007873 . DOIPubMedGoogle Scholar - Nobach D, Bourg M, Herzog S, Lange-Herbst H, Encarnação JA, Eickmann M, et al. Shedding of infectious Borna disease virus-1 in living bicolored white-toothed shrews. PLoS One. 2015;10:
e0137018 . DOIPubMedGoogle Scholar - Hilbe M, Herrsche R, Kolodziejek J, Nowotny N, Zlinszky K, Ehrensperger F. Shrews as reservoir hosts of Borna disease virus. Emerg Infect Dis. 2006;12:675–7. DOIPubMedGoogle Scholar
- Dürrwald R, Kolodziejek J, Weissenböck H, Nowotny N. The bicolored white-toothed shrew Crocidura leucodon (HERMANN 1780) is an indigenous host of mammalian Borna disease virus. PLoS One. 2014;9:
e93659 . DOIPubMedGoogle Scholar - Ruszkowski JJ, Hetman M, Turlewicz-Podbielska H, Pomorska-Mól M. Hedgehogs as a potential source of zoonotic pathogens—a review and an update of knowledge. Animals (Basel). 2021;11:1754. DOIPubMedGoogle Scholar
- Schönbächler K, Hatt J, Silaghi C, Merz N, Fraefel C, Bachofen C. Confirmation of tick-borne encephalitis virus in a European hedgehog (Erinaceus europaeus) [in German]. Schweiz Arch Tierheilkd. 2019;161:23–31. DOIPubMedGoogle Scholar
- Faragó Z. Rabid hedgehog in inner-city area of Budapest [in Hungarian]. Orv Hetil. 1997;138:2231–2.PubMedGoogle Scholar
- Duque-Valencia J, Sarute N, Olarte-Castillo XA, Ruíz-Sáenz J. Evolution and interspecies transmission of canine distemper virus—an outlook of the diverse evolutionary landscapes of a multi-host virus. Viruses. 2019;11:582. DOIPubMedGoogle Scholar
- Matiasek K, Pfaff F, Weissenböck H, Wylezich C, Kolodziejek J, Tengstrand S, et al. Mystery of fatal ‘staggering disease’ unravelled: novel rustrela virus causes severe meningoencephalomyelitis in domestic cats. Nat Commun. 2023;14:624. DOIPubMedGoogle Scholar
- Douady CJ, Chatelier PI, Madsen O, de Jong WW, Catzeflis F, Springer MS, et al. Molecular phylogenetic evidence confirming the Eulipotyphla concept and in support of hedgehogs as the sister group to shrews. Mol Phylogenet Evol. 2002;25:200–9. DOIPubMedGoogle Scholar
- Steininger P, Ensser A, Knöll A, Korn K. Results of tick-borne encephalitis virus (TBEV) diagnostics in an endemic area in southern Germany, 2007 to 2022. Viruses. 2023;15:2357. DOIPubMedGoogle Scholar
- Thilén E, Rubbenstroth D, Tengstrand S, Pfaff F, Wensman JJ, Ley C. Evidence of rustrela virus-associated feline staggering disease in Sweden since the 1970s. Acta Vet Scand. 2024;66:59. DOIPubMedGoogle Scholar
- Li Z, Feng Z, Ye H. Rabies viral antigen in human tongues and salivary glands. J Trop Med Hyg. 1995;98:330–2.PubMedGoogle Scholar
- Haines DM, Martin KM, Chelack BJ, Sargent RA, Outerbridge CA, Clark EG. Immunohistochemical detection of canine distemper virus in haired skin, nasal mucosa, and footpad epithelium: a method for antemortem diagnosis of infection. J Vet Diagn Invest. 1999;11:396–9. DOIPubMedGoogle Scholar
- Haas B, Becht H, Rott R. Purification and properties of an intranuclear virus-specific antigen from tissue infected with Borna disease virus. J Gen Virol. 1986;67:235–41. DOIPubMedGoogle Scholar
- Wang F, Flanagan J, Su N, Wang L-C, Bui S, Nielson A, et al. RNAscope: a novel in situ RNA analysis platform for formalin-fixed, paraffin-embedded tissues. J Mol Diagn. 2012;14:22–9. DOIPubMedGoogle Scholar
- Garman RH. Histology of the central nervous system. Toxicol Pathol. 2011;39:22–35. DOIPubMedGoogle Scholar
- Liesche F, Ruf V, Zoubaa S, Kaletka G, Rosati M, Rubbenstroth D, et al. The neuropathology of fatal encephalomyelitis in human Borna virus infection. Acta Neuropathol. 2019;138:653–65. DOIPubMedGoogle Scholar
- Fürstenau J, Richter MT, Erickson NA, Große R, Müller KE, Nobach D, et al. Borna disease virus 1 infection in alpacas: comparison of pathological lesions and viral distribution to other dead-end hosts. Vet Pathol. 2024;61:62–73. DOIPubMedGoogle Scholar
- Bilzer T, Planz O, Lipkin WI, Stitz L. Presence of CD4+ and CD8+ T cells and expression of MHC class I and MHC class II antigen in horses with Borna disease virus-induced encephalitis. Brain Pathol. 1995;5:223–30. DOIPubMedGoogle Scholar
- Ellenberger C, Heenemann K, Vahlenkamp TW, Grothmann P, Herden C, Heinrich A. Borna disease in an adult free-ranging Eurasian beaver (Castor fiber albicus). J Comp Pathol. 2024;209:31–5. DOIPubMedGoogle Scholar
- Rubbenstroth D. Avian Bornavirus research—a comprehensive review. Viruses. 2022;14:1513. DOIPubMedGoogle Scholar
- Carbone KM, Moench TR, Lipkin WI. Borna disease virus replicates in astrocytes, Schwann cells and ependymal cells in persistently infected rats: location of viral genomic and messenger RNAs by in situ hybridization. J Neuropathol Exp Neurol. 1991;50:205–14. DOIPubMedGoogle Scholar
- Werner-Keišs N, Garten W, Richt JA, Porombka D, Algermissen D, Herzog S, et al. Restricted expression of Borna disease virus glycoprotein in brains of experimentally infected Lewis rats. Neuropathol Appl Neurobiol. 2008;34:590–602. DOIPubMedGoogle Scholar
- Coras R, Korn K, Kuerten S, Huttner HB, Ensser A. Severe bornavirus-encephalitis presenting as Guillain-Barré-syndrome. Acta Neuropathol. 2019;137:1017–9. DOIPubMedGoogle Scholar
- Shankar V, Kao M, Hamir AN, Sheng H, Koprowski H, Dietzschold B. Kinetics of virus spread and changes in levels of several cytokine mRNAs in the brain after intranasal infection of rats with Borna disease virus. J Virol. 1992;66:992–8. DOIPubMedGoogle Scholar
- Enbergs HK, Vahlenkamp TW, Kipar A, Müller H, Haimo K. Enbergs, Thomas W Vahlenkam. Experimental infection of mice with Borna disease virus (BDV): replication and distribution of the virus after intracerebral infection. J Neurovirol. 2001;7:272–7. DOIPubMedGoogle Scholar
1These senior authors contributed equally to this article.