Volume 15, Number 9—September 2009
Research
Susceptibilities of Nonhuman Primates to Chronic Wasting Disease
Cite This Article
Citation for Media
Abstract
Chronic wasting disease (CWD) is a transmissible spongiform encephalopathy, or prion disease, that affects deer, elk, and moose. Human susceptibility to CWD remains unproven despite likely exposure to CWD-infected cervids. We used 2 nonhuman primate species, cynomolgus macaques and squirrel monkeys, as human models for CWD susceptibility. CWD was inoculated into these 2 species by intracerebral and oral routes. After intracerebral inoculation of squirrel monkeys, 7 of 8 CWD isolates induced a clinical wasting syndrome within 33–53 months. The monkeys’ brains showed spongiform encephalopathy and protease-resistant prion protein (PrPres) diagnostic of prion disease. After oral exposure, 2 squirrel monkeys had PrPres in brain, spleen, and lymph nodes at 69 months postinfection. In contrast, cynomolgus macaques have not shown evidence of clinical disease as of 70 months postinfection. Thus, these 2 species differed in susceptibility to CWD. Because humans are evolutionarily closer to macaques than to squirrel monkeys, they may also be resistant to CWD.
Transmissible spongiform encephalopathies (TSEs), or prion diseases, are neurodegenerative diseases that affect many mammalian species. Some examples include bovine spongiform encephalopathy (BSE) in cattle, scrapie in sheep and goats, Creutzfeldt-Jakob disease (CJD) in humans, and chronic wasting disease (CWD) in cervids. CWD was first found in captive deer in Colorado in 1967 (1) and was later identified in several US states and Canadian provinces (2). Epidemiologic evidence suggests that CWD continues to spread among cervid populations in North America (3), creating concern that CWD may cross species barriers to infect humans or domestic animals that may be eaten by humans. Thus, the host range of CWD and the level of protection provided by species barriers should be determined.
Substantial progress has been made in testing species barriers for CWD by using transgenic mice expressing species-specific prion protein (PrP), by direct infection into new species, or by in vitro conversion assays. The most sensitive method for testing susceptibility to TSE agents is intracerebral injection. Unfortunately, this route does not mimic most natural situations and only enables assessment of whether the possibility of transmission exists. Hamir et al. infected cattle and sheep with CWD by the intracerebral route and found protease-resistant PrP (PrPres) in 5 of 13 cattle and 2 of 8 sheep, which indicated that these ruminant species can propagate CWD (4,5). However, oral exposure in these hosts apparently does not cause disease (2).
CWD cross-species transmission to nonagricultural and laboratory animals has shown variable levels of susceptibility depending on the route of transmission. For example, ferrets were 100% susceptible to CWD by intracerebral infection but were not susceptible to oral infection (6,7). Mink were only 25% susceptible to CWD by intracerebral infection and were not susceptible to oral infection (8). CWD has been successfully transmitted and adapted to laboratory rodents, including hamsters, transgenic mice expressing hamster PrP, and transgenic mice overexpressing mouse PrP (9,10). In contrast, transgenic mice expressing human PrP were not susceptible to CWD by intracerebral infection (11,12), a finding that provided evidence for a human species barrier against CWD infection. However, work started in 1980 and published in 2005 by Marsh et al. showed that 2 squirrel monkeys (Saimiri sciureus) infected by the intracerebral route with brain homogenate from a single CWD-affected mule deer became clinically sick at 31 and 34 months postinfection, and both were positive for PrPres (13). This evidence that at least 1 species of nonhuman primate was susceptible to CWD weakened the conclusion that humans may be protected from CWD by a species barrier.
We addressed 4 questions raised by the original observation that squirrel monkeys are susceptible to CWD (13). First, we compared intracerebral and oral routes of infection. This comparison was of interest because the oral route is likely to be an important natural route of disease transmission, and susceptibility is known to be lower by this route in most models. Second, we compared 2 species of nonhuman primates, cynomolgus macaques (Macaca fascicularis) and squirrel monkeys, each of which has previously shown susceptibility to various human prion diseases (14–16). However, humans are believed to be evolutionarily closer to cynomolgus macaques than to squirrel monkeys (17), and cynomolgus macaques may be a more accurate model for a human species barrier. Third, because only 1 CWD source was tested by Marsh et al. (13), we studied 8 different pools of CWD representing wild and captive cervids, including mule deer, white-tailed deer, and elk, from separate regions in the United States. Fourth, we tested the species tropism of CWD agent passaged in squirrel monkeys.
A description of the materials and methods used in this study follows. Additional details are available in the Technical Appendix
Animal Research
All monkeys and mice were housed at the Rocky Mountain Laboratories (Hamilton, MT, USA). Experimentation followed protocols approved by the National Institutes of Health Rocky Mountain Laboratories Animal Care and Use Committee.
CWD Pools for Infection of Primates
CWD-positive brain homogenates were provided by E.S.W. and M.W.M. Contents of each pool were as follows: MD-1, 6 free-ranging mule deer from Wyoming (18); MD-2, 4 captive mule deer from Colorado; MD-3, 28 captive mule deer from Wyoming and Colorado (2,19); WTD-1, 7 captive white-tailed deer from Wyoming and Colorado (18,20); WTD-2, 1 wild white-tailed deer from Wyoming; Elk-1, 2 free-ranging elk from Wyoming (18); Elk-2, 6 elk from a South Dakota game farm; and Elk-3, 10 captive elk from Wyoming and Colorado. Normal elk brain was a pool from 2 elk from Montana obtained from Lynn Creekmore of the US Department of Agriculture.
Inoculation of Monkeys
For intracerebral injections, squirrel monkeys received either 2 mg or 20 mg brain in a total volume of 200 μL, and cynomolgus macaques received 5 mg in a total volume of 500 μL. Oral doses of 200 mg brain/mL were given on 5 different days at 2–6 day intervals. Squirrel monkeys received 3-mL doses; most macaques received 4-mL doses. The inoculum was given to anesthetized animals through a rubber gastric tube.
Inoculation of Transgenic Mice
Brain homogenates diluted in phosphate buffered balanced solution containing 2% fetal bovine serum were inoculated intracerebrally into young adult mice. Volumes were 50 μL.
Generation of Transgenic Mice Expressing Human PrP
Mice expressing human PrP (tgRM and tg66) were generated by using a transgene, cosSHa.HumPrP, which was created by ligating the human PrP open reading frame into the cosSHa.Tet vector (21). The transgene was inoculated into eggs of FVBn–mouse PrP null mice in the laboratories of R.R. (tg66) and L.C. (tgRM). Each line of mice overexpressed human PrP as tested by Western blot with monoclonal antibody 3F4.
Analysis of Protease-sensitive PrP and PrPres by Immunoblot
Tissues were prepared by making a 20% (wt/vol) homogenate in 0.01 M Tris buffer, pH 7.4. Samples to be analyzed for protease-sensitive PrP (PrPsen) contained the following protease inhibitors: 10 μmol/L leupeptin, 1 μmol/L pepstatin A, and 1 μg/mL aprotinin. Samples were sonicated for 1 min and centrifuged at 5,000 rpm for 10 min. Supernatants were mixed 1:1 in 2× sample buffer and boiled for 3 min before electrophoresis.
Preparation of samples for PrPres analysis has been described (18). Removal of carbohydrate residues from PrPres was performed by digestion with peptide-N-glycosidase F (22).
After electrophoresis, proteins were transferred to Immobilon polyvinylidene difluoride–P membranes (Millipore, Billerica, MA, USA), and PrP bands were detected with antibodies 3F4 (residues 109–112) (23), D13 (residues 96–106) (24) (InPro Biotechnology, Inc., South San Francisco, CA, USA), or L42 (residues 145–163) (r-Biopharm, Darmstadt, Germany) (25). Bands were detected by using enhanced chemiluminescence substrate (GE Healthcare, Piscataway, NJ, USA).
Histopathologic and Immunohistochemical Analyses
Routine formalin fixation, embedding, and tissue-sectioning protocols were followed. Tissues were stained with hematoxylin and eosin and analyzed for pathologic changes. Immunohistochemical staining was performed by using an automated Nexus stainer (Ventana, Tucson, AZ, USA). Anti-PrP antibodies D13 and 3F4 were used for PrPres immunostaining as described (26,27).
Sequencing
Primate genomic DNA was purified from whole blood, and PCR products were amplified by using PuRe Taq Ready-To-Go PCR beads (GE Healthcare). Two primers from the extreme outer ends of the open reading frame, including the previously published forward primer HM-1 (28) with mPrP-780R (5′-TCCCACTATCAGGAAGATGAGG-3′) or a combination of outer primers with internal primers mPrP-397F (5′-CCTTGGTGGCTACATGCTG-3′) and mPrP-416R (5′-CCAGCATGTAGCCACCAAG-3′), were used. Assembly comparisons were made against human, elk, mule deer, cynomolgus macaque, and squirrel monkey by using Sequencher version 4.6 (Gene Codes, Ann Arbor, MI, USA).
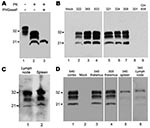
Infectivity Levels in CWD Pools
When the 8 pools of CWD (representing both wild and captive deer and elk) used as inocula were analyzed by immunoblot, PrPres in the 8 pools showed similar electrophoretic mobilities and glycoform patterns (Figure 1, panel A), but PrPres levels differed when quantitatively compared (Figure 1, panel C). To measure the level of infectivity in these pools, we titered each pool in transgenic mice expressing deer PrP (line 33; tgDeerPrP) (18). A typical endpoint dilution titration is shown in Figure 1, panel B. The 8 pools had 50% infectious dose (ID50) titers ranging from 6.3 × 107 to 5.0 × 108 ID50/g of brain homogenate (Figure 1, panel C). Comparison of titers with PrPres levels showed a partial correlation (Figure 1, panel C). For example, the CWD pool with the lowest infectivity titer (MD-2) was also the pool with the lowest PrPres level. However, for some pools, these tests showed discrepant values.
Intracerebral Infection of Squirrel Monkeys
To test susceptibility to CWD, we inoculated squirrel monkeys with each of the 8 CWD pools described above. Of 13 squirrel monkeys, 11 became symptomatic (33–53 mo postinfection [mpi]) (Table 1). The most consistent and reliable clinical finding was a severe wasting syndrome. Weight loss (average decrease of 33%) was most pronounced in the final few months of infection. Affected monkeys also had rough, poor-quality coats despite continuing to eat and drink. In the final 3–5 weeks, monkeys became weak and less active and spent most of their time hunched at the bottom of their cage. When the monkeys were encouraged to move, they did so slowly and deliberately. In the terminal stage of disease, a few had muscle tremors, excessive salivation, and mild ataxia. Fine, coordinated movement such as eating food was rarely affected. Monkeys were euthanized when terminal-stage weakness and wasting compromised their mobility and ability to eat and drink.
No clear correlation between incubation period and amount of agent inoculated was noted (Table 1). For example, 3 pairs of monkeys received the same inocula but in amounts that differed by 10-fold (Elk-1, Elk-3, and MD-1). Two pairs that received the lower dose became clinically sick first (Elk-1 and Elk-3). Both members of the third pair (MD-1) were euthanized after 36 months (Table 1). Two animals received the same dose of WTD-1 pool, yet to date, only 1 animal has become clinically sick. Animals that received the CWD pool with the lowest titer (MD-2) had incubation periods similar to those receiving much higher titered inocula (Table 1).
In all monkeys with clinical signs, CWD was confirmed by Western blot detection of PrPres in brain (Figure 2, panels A, B). The glycoform pattern of PrPres was similar for all affected monkeys inoculated with different CWD pools (Figure 2, panel B). Because PrPres deposition may also occur outside the central nervous system, we also tested peripheral lymphoid tissues. For 3 of 11 monkeys that had PrPres in brain, PrPres was also found in spleen and lymph nodes (Figure 2, panel C). In general, PrPres levels were much lower in lymphoid tissues than in brain and were often not detected by Western blot. All nonlymphatic tissues tested (cardiac muscle, skeletal muscle, duodenum, jejunum, ileum, colon, salivary gland, kidney, and lung) were negative for PrPres by immunoblot.
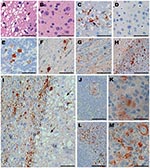
Tissues from squirrel monkeys euthanized after intracerebral injection with CWD (Table 1) were also examined by histopathologic analysis, including staining with hematoxylin and eosin and immunohistochemical detection of PrPres. All monkeys examined had spongiosis in the cerebral cortex, caudate, putamen, and thalamus (Figure 3, panel A). In addition, PrPres deposition was observed in many brain regions with large PrPres-positive plaques in the thalamus, cerebellum, and spinal cord (Figure 3, panels C, E, F) and in smaller plaques spread out in the gray matter of the internal capsule and white matter of the corpus callosum (Figure 3, panels G, H). The most abundant and consistent location for PrPres staining was found in the frontal cortex and in the fiber tracts of the claustrum (Figure 3, panel I). The adjacent caudate had severe spongiosis and astrocytosis but minimal PrPres (Figure 3, panel I). PrPres was also detected in lymph nodes and spleen, within follicles, in areas resembling follicular dendritic cells (Figure 3, panels K, M). Immunohistochemical analysis showed no PrPres in heart, kidney, adrenal gland, skeletal muscle, salivary gland, tongue, pancreas, white fat, and all regions of the gastrointestinal tract.
Oral Infection of Squirrel Monkeys
To test a more natural route of infection, we exposed squirrel monkeys orally to CWD. Of the 15 exposed squirrel monkeys, 1 (no. 345) was found dead in its cage at 69 mpi; it had shown no neurologic signs or weakness. Western blot results indicated PrPres in brain, spleen, and lymph nodes (Figure 2, panel D). The level of PrPres in the brain of monkey 345 was comparable with that in end-stage intracerebrally inoculated monkeys; body weight at necropsy indicated a 33% decrease over the final 10 months. The high levels of PrPres and the severe wasting indicate that CWD infection could have been the cause of death. A second monkey, 303, was euthanized at 69 mpi because of suspicion of TSE after 2 weeks of progressive weakness, wasting, and eventual anorexia. PrPres analysis confirmed PrPres in brain (Figure 2, panel D), spleen, and lymph nodes. For monkeys 303 and 345, levels of PrPres in the lymph nodes and spleens were 10–100-fold lower than those in brain.
Two other orally infected monkeys were euthanized during the first 69 mpi (Table 2). Monkey 301 was euthanized at 39 mpi, after rapid onset of lethargy and anorexia that led to severe dehydration. Results of Western blot analysis for PrPres were negative in brain (Figure 1, panel B), spleen, lymph nodes, heart, skeletal muscle, duodenum, jejunum, ileum, colon, salivary gland, kidney, lung, and tonsil. However, immunohistochemical analysis detected PrPres in the spleen and 1 mesenteric lymph node from this monkey, indicating a low level of infection (Figure 3, panels J,K). Monkey 614 was euthanized at 44 mpi because it did not recover from anesthesia related to routine tuberculosis screening. Neither Western blot nor immunohistochemical analysis detected PrPres in brain, spleen, or lymph nodes of this monkey.
Infection of Cynomolgus Macaques
We inoculated cynomolgus macaques both orally and intracerebrally with 3 CWD inocula representing elk, mule deer, and white-tailed deer (Table 3). Of the cynomolgus macaques, 1 (no. 609) was euthanized at 48 mpi after it became aggressive. Brain (Figure 2, panel B), spinal cord, spleen, and lymph nodes were negative for PrPres by Western blot and immunohistochemical analysis. All remaining CWD-inoculated cynomolgus monkeys are currently (at 70 mpi) neurologically asymptomatic and have stable or increased body weights.
Sequences
Amino acid substitutions in PrP can alter susceptibility to TSE agents, including CWD (18,29,30). To determine whether the lack of susceptibility in several intracerebrally inoculated squirrel monkeys (Table 1) was caused by PrP gene polymorphisms, we sequenced the PrP genes from 23 squirrel monkeys. When compared with published squirrel monkey sequences (28,31), variation was seen at residue 164, in the number of octapeptide repeats, and at residue 19 of the signal peptide (Table 4). However, these genetic differences in PrP did not appear to account for the lack of susceptibility of monkey 310, which was genotype A, because this genotype was also found in 5 of the CWD-positive monkeys. Because we were not able to sequence PrP of monkey 628, we could not assess the role of PrP variation in the lack of disease.
Infectivity of CWD-infected Squirrel Monkey Tissues in PrP Transgenic Mice
To determine whether passage of CWD in squirrel monkeys altered the tropism of the infectious agent, we inoculated tgDeerPrP mice and tg mice expressing human PrP (lines 66 and RM) intracerebrally with tissue homogenates from 3 CWD-positive squirrel monkeys (nos. 322, 308, and 301) with PrPres and from an intracerebrally inoculated cynomolgus macaque (no. 609). Clinical disease did not develop in any tgDeerPrP, tg66, or tgRM mice during 600–700 days (Table 5). The lack of transmission to tgDeerPrP mice from the 3 squirrel monkeys with detectable CWD PrPres indicated that either the infectivity levels were low in these squirrel monkeys or that the original cervid species tropism was altered by the passage in squirrel monkeys. Similarly, the lack of transmission to tg mice expressing human PrP implied that passage through squirrel monkeys did not facilitate adaptation to an agent with increased tropism for humans.
As new CWD foci continue to emerge among cervid populations, the risk for CWD transmission to humans needs to be assessed. We used 2 monkey species and 2 routes of inoculation to test the susceptibility of primates to 8 different pools of CWD. To date, we have verified CWD in 11 of 13 intracerebrally inoculated squirrel monkeys; average incubation period was 41 months (range 33–53 months). Using a single CWD pool, Marsh et al. noted infection in 2 of 2 squirrel monkeys 31–34 months after intracerebral inoculation (13). Intracerebral inoculation of squirrel monkeys with other TSE agents, including agents of kuru, variant CJD, sporadic CJD, and sheep scrapie, had incubation periods of ≈24 months and attack rates of ≈100% (14,15,32). The extended incubation periods and lower attack rates for our squirrel monkeys may result from a partial species barrier to CWD.
The signs of wasting syndrome in CWD-infected monkeys were similar to those of CWD infection in cervids, in which loss of body condition is nearly always a major component of infection and neurologic deficits vary (2). The correlation of clinical signs between CWD in cervids and squirrel monkeys suggests that CWD might affect a common brain region in each species. We observed PrPres deposition in squirrel monkeys primarily in the frontal lobe of the cerebral cortex, claustrum, putamen, and thalamus. Cervids typically have the most abundant and predictable PrPres in the dorsal motor vagus nucleus (obex), olfactory cortex, and diencephalon (including thalamus, hypothalamus, metathalamus, and epithalamus) (2,33). A plausible hypothesis could be that disruption of regions within the hypothalamus and thalamus leads to a metabolic imbalance, resulting in a severe wasting syndrome.
We did not observe a strong correlation between infectivity titer inoculated and attack incidence or incubation period (Table 1). One potential explanation is that the variation in speed of disease progression might not be relevant given the low number of animals in each group. A second possibility is that our squirrel monkeys varied at PrP alleles that may affect CWD susceptibility. However, analysis of 23 squirrel monkeys showed no PrP sequence differences correlating with susceptibility to CWD (Tables 1, 2, 4). A third possibility is that genes other than the gene for PrP might influence CWD susceptibility.
For humans, eating infected or contaminated tissue is a likely route of CWD exposure. In other animal models, oral transmission of TSE is generally 1,000-fold less effective than direct intracerebral challenge and results in longer incubation periods and lower efficiency of disease transmission. In our oral transmission experiments, we found evidence of CWD infection in 3 monkeys; 2 at 69 mpi had abundant PrPres in brain and lower levels in spleen and lymph nodes, and 1 euthanized at 39 mpi had PrPres in lymphatic tissues only. Thus, transmission seems to be slower by the oral route than by the intracerebral route, and other orally infected monkeys may be affected in the future.
Cynomolgus macaques are evolutionarily closer to humans than are squirrel monkeys (17). At nearly 6 years postinoculation, no macaques have shown clinical signs of CWD. Intracerebral inoculation of cynomolgus macaques with BSE causes disease in 3 years; human variant CJD requires 2–3 years, and human sporadic CJD requires 5 years (16,34). However, oral inoculation of cynomolgus macaques with BSE agent required a minimum of 5 years before clinical disease was observed (35). Therefore, we cannot rule out CWD transmission to cynomolgus macaques.
The PrP gene sequence can influence cross-species transmission of prion disease. Therefore, we compared squirrel monkey and cynomolgus macaque PrP gene sequences to look for differences that might account for different susceptibilities of these monkeys to CWD. In the PrP gene excluding the signal peptide, deer differed from squirrel monkeys at 17 residues and from cynomolgus macaques at 16 residues, but 14 of these differing residues were identical in squirrel monkeys and macaques (Figure 4). Therefore, there are only 2 residues in cynomolgus macaques (100 and 108) and 3 residues in squirrel monkeys (56, 159 and 182) at which these monkeys differ from deer and also from each other. These residues might play a role in susceptibility differences seen in our study.
Human exposure to CWD-infected cervids in past decades is likely. The highest levels of prion infectivity are present in the central nervous system and lymphatic tissues of CWD-infected cervids; contamination of knives, saws, and muscles with these tissues can easy occur when processing game. Despite the likelihood of exposures, epidemiologic studies of humans living in CWD-endemic areas of Colorado and Wyoming during 1979–2001 have not shown any increases in human TSE cases (36,37). Ongoing studies by the Colorado Department of Public Health and Environmental Human Prion Disease Surveillance Program, in conjunction with the University of Colorado School of Medicine, have also concluded that no convincing cases of CWD transmission to humans have been detected in Colorado (38). However, if CWD in humans appears like a wasting syndrome similar to that observed in the squirrel monkeys in our study, affected persons might receive a diagnosis of a metabolic disorder and never be tested for TSE. Fortunately, additional laboratory data are consistent with the epidemiologic data, and these results support the conclusion that a species barrier protects humans from CWD infection (11–13,20,36,37).
Dr Brent Race is a staff scientist in the Laboratory of Persistent Viral Diseases, National Institute of Allergy and Infectious Diseases. His primary research interests are infectious diseases of humans and livestock, especially transmissible spongiform encephalopathies.
Acknowledgments
We thank John Portis, Byron Caughey, and Kristin McNally for critical review of the manuscript; Lori Lubke and Rebecca Rosenke for assistance with histologic analysis; Rocky Rivera and Ed Schreckendgust for animal husbandry; Anita Mora and Gary Hettrick for graphics assistance; Serguei Soukharev for assistance in creating transgenic mice expressing human PrP; Robert Rohwer for tg66 mice and sporadic CJD inocula; Terry Kreeger, Jean Jewell, and Lynn Creekmore for CWD-positive and CWD-negative cervid tissues; and Pier-Luigi Gambetti for a sample from a patient with sporadic CJD.
This study was supported by the National Institute of Allergy and Infectious Diseases, Division of Intramural Research.
References
- Williams ES, Young S. Chronic wasting disease of captive mule deer: a spongiform encephalopathy. J Wildl Dis. 1980;16:89–98.PubMedGoogle Scholar
- Miller MW, Williams ES. Chronic wasting disease of cervids. Curr Top Microbiol Immunol. 2004;284:193–214.PubMedGoogle Scholar
- Hamir AN, Kunkle RA, Cutlip RC, Miller JM, O’Rourke KI, Williams ES, Experimental transmission of chronic wasting disease agent from mule deer to cattle by the intracerebral route. J Vet Diagn Invest. 2005;17:276–81.PubMedGoogle Scholar
- Hamir AN, Kunkle RA, Cutlip RC, Miller JM, Williams ES, Richt JA. Transmission of chronic wasting disease of mule deer to Suffolk sheep following intracerebral inoculation. J Vet Diagn Invest. 2006;18:558–65.PubMedGoogle Scholar
- Sigurdson CJ, Mathiason CK, Perrott MR, Eliason GA, Spraker TR, Glatzel M, Experimental chronic wasting disease (CWD) in the ferret. J Comp Pathol. 2008;138:189–96. DOIPubMedGoogle Scholar
- Bartz JC, Marsh RF, McKenzie DI, Aiken JM. The host range of chronic wasting disease is altered on passage in ferrets. Virology. 1998;251:297–301. DOIPubMedGoogle Scholar
- Harrington RD, Baszler TV, O’Rourke KI, Schneider DA, Spraker TR, Liggitt HD, A species barrier limits transmission of chronic wasting disease to mink (Mustela vison). J Gen Virol. 2008;89:1086–96. DOIPubMedGoogle Scholar
- Raymond GJ, Raymond LD, Meade-White KD, Hughson AG, Favara C, Gardner D, Transmission and adaptation of chronic wasting disease to hamsters and transgenic mice: evidence for strains. J Virol. 2007;81:4305–14. DOIPubMedGoogle Scholar
- Sigurdson CJ, Manco G, Schwarz P, Liberski P, Hoover EA, Hornemann S, Strain fidelity of chronic wasting disease upon murine adaptation. J Virol. 2006;80:12303–11. DOIPubMedGoogle Scholar
- Kong Q, Huang S, Zou W, Vanegas D, Wang M, Wu D, Chronic wasting disease of elk: transmissibility to humans examined by transgenic mouse models. J Neurosci. 2005;25:7944–9. DOIPubMedGoogle Scholar
- Tamguney G, Giles K, Bouzamondo-Bernstein E, Bosque PJ, Miller MW, Safar J, Transmission of elk and deer prions to transgenic mice. J Virol. 2006;80:9104–14. DOIPubMedGoogle Scholar
- Marsh RF, Kincaid AE, Bessen RA, Bartz JC. Interspecies transmission of chronic wasting disease prions to squirrel monkeys (Saimiri sciureus). J Virol. 2005;79:13794–6. DOIPubMedGoogle Scholar
- Brown P, Gibbs CJ Jr, Rodgers-Johnson P, Asher DM, Sulima MP, Bacote A, Human spongiform encephalopathy: the National Institutes of Health series of 300 cases of experimentally transmitted disease. Ann Neurol. 1994;35:513–29. DOIPubMedGoogle Scholar
- Williams L, Brown P, Ironside J, Gibson S, Will R, Ritchie D, Clinical, neuropathologial and immunohistochemical features of sporadic and variant forms of Creutzfeldt-Jakob disease in the squirrel monkey (Saimiri scuireus). J Gen Virol. 2007;88:688–95. DOIPubMedGoogle Scholar
- Lasmezas CI, Deslys JP, Demaimay R, Adjou KT, Lamoury F, Dormont D, BSE transmission to macaques. Nature. 1996;381:743–4. DOIPubMedGoogle Scholar
- Hayasaka K, Gojobori T, Horai S. Molecular phylogeny and evolution of primate mitochondrial DNA. Mol Biol Evol. 1988;5:626–44.PubMedGoogle Scholar
- Meade-White K, Race B, Trifilo M, Bossers A, Favara C, Lacasse R, Resistance to chronic wasting disease in transgenic mice expressing a naturally occurring allelic variant of deer prion protein. J Virol. 2007;81:4533–9. DOIPubMedGoogle Scholar
- Sigurdson CJ, Williams ES, Miller MW, Spraker TR, O’Rourke KI, Hoover EA. Oral transmission and early lymphoid trophism of chronic wasting disease PrPres in mule deer fawns (Odocoileus hemionus). J Gen Virol. 1999;80:2757–64.PubMedGoogle Scholar
- Raymond GJ, Bossers A, Raymond LD, O’Rourke KI, McHolland LE, Bryant PK III, Evidence of a molecular barrier limiting susceptibility of humans, cattle and sheep to chronic wasting disease. EMBO J. 2000;19:4425–30. DOIPubMedGoogle Scholar
- Scott MR, Kohler R, Foster D, Prusiner SB. Chimeric prion protein expression in cultured cells and transgenic mice. Protein Sci. 1992;1:986–97. DOIPubMedGoogle Scholar
- Race BL, Meade-White KD, Ward A, Jewell J, Miller MW, Williams ES, Levels of abnormal prion protein in deer and elk with chronic wasting disease. Emerg Infect Dis. 2007;13:824–30.PubMedGoogle Scholar
- Kascsak RJ, Rubenstein R, Merz PA, Tonna-DeMasi M, Fersko R, Carp RI, Mouse monoclonal and polyclonal antibody to scrapie-associated fibril proteins. J Virol. 1987;61:3688–93.PubMedGoogle Scholar
- Matsunaga Y, Peretz D, Williamson A, Burton D, Mehlhorn I, Groth D, Cryptic epitopes in N–terminally truncated prion protein are exposed in the full-length molecule: dependence of conformation on pH. Proteins. 2001;44:110–8. DOIPubMedGoogle Scholar
- Vorberg I, Buschmann A, Harmeyer S, Saalmuller A, Pfaff E, Groschup MH. A novel epitope for the specific detection of exogenous prion proteins in transgenic mice and transfected murine cell lines. Virology. 1999;255:26–31. DOIPubMedGoogle Scholar
- Race B, Meade-White K, Oldstone MB, Race R, Chesebro B. Detection of prion infectivity in fat tissues of scrapie-infected mice. PLoS Pathog. 2008;4:e1000232. DOIPubMedGoogle Scholar
- Kercher L, Favara C, Striebel JF, Lacasse R, Chesebro B. Prion protein expression differences in microglia and astroglia influence scrapie-induced neurodegenration in the retina and brain of transgenic mice. J Virol. 2007;81:10340–51. DOIPubMedGoogle Scholar
- Schätzl HM, Da Costa M, Taylor L, Cohen FE, Prusiner SB. Prion protein variation among primates. J Mol Biol. 1995;245:362–74. DOIPubMedGoogle Scholar
- O’Rourke KI, Spraker TR, Hamburg LK, Besser TE, Brayton KA, Knowles DP. Polymorphisms in prion precursor functional gene but not the pseudogene are associated with susceptibility to chronic wasting disease in white-tailed deer. J Gen Virol. 2004;85:1339–46. DOIPubMedGoogle Scholar
- Fox KA, Jewell JE, Williams ES, Miller MW. Patterns of PrPCWD accumulation during the course of chronic wasting disease infection in orally inoculated mule deer (Odocoileus hemionus). J Gen Virol. 2006;87:3451–61. DOIPubMedGoogle Scholar
- Schneider I, Schneider H, Schneider MP, Silva A. The prion protein and New World primate phylogeny. Genet Mol Biol. 2004;27:505–10. DOIGoogle Scholar
- Gibbs CJ Jr, Gajdusek DC. Experimental subacute spongiform virus encephalopathies in primates and other laboratory animals. Science. 1973;182:67–8. DOIPubMedGoogle Scholar
- Spraker TR, Zink RR, Cummings BA, Wild MA, Miller MW, O’Rourke KI. Comparison of histological lesions and immunohistochemical staining of proteinease-resistant prion protein in a naturally occurring spongiform encephalopathy of free-ranging mule deer (Odocoileus hemionus) with those of chronic wasting disease of captive mule deer. Vet Pathol. 2002;39:110–9. DOIPubMedGoogle Scholar
- Herzog C, Riviere J, Lescoutra-Etchegaray N, Charbonnier A, Leblanc V, Sales N, PrPTSE distribution in a primate model of variant, sporadic, and iatrogenic Creutzfeldt-Jakob disease. J Virol. 2005;79:14339–45. DOIPubMedGoogle Scholar
- Lasmezas CI, Comoy E, Hawkins S, Herzog C, Mouthon F, Konold T, Risk of oral infection with bovine spongiform encephalopathy agent in primates. Lancet. 2005;365:781–3.PubMedGoogle Scholar
- Belay ED, Maddox RA, Williams ES, Miller MW, Gambetti P, Schonberger LB. Chronic wasting disease and potential transmission to humans. Emerg Infect Dis. 2004;10:977–84.PubMedGoogle Scholar
- Mawhinney S, Pape WJ, Forster JE, Anderson CA, Bosque P, Miller MW. Human prion disease and relative risk associated with chronic wasting disease. Emerg Infect Dis. 2006;12:1527–35.PubMedGoogle Scholar
- Anderson CA, Bosque P, Filley CM, Arciniegas DB, Kleinschmidt-Demasters BK, Pape WJ, Colorado surveillance program for chronic wasting disease transmission to humans: lessons from 2 highly suspicious but negative cases. Arch Neurol. 2007;64:439–41. DOIPubMedGoogle Scholar
Figures
Tables
Cite This ArticleTable of Contents – Volume 15, Number 9—September 2009
| EID Search Options |
|---|
|
|
|
|
|
|

Please use the form below to submit correspondence to the authors or contact them at the following address:
Brent Race, Laboratory of Persistent Viral Diseases, Rocky Mountain Laboratories, 903 South 4th St, Hamilton, MT 59840, USA;
Top