Volume 22, Number 8—August 2016
Research
Virulence and Evolution of West Nile Virus, Australia, 1960–2012
Cite This Article
Citation for Media
Abstract
Worldwide, West Nile virus (WNV) causes encephalitis in humans, horses, and birds. The Kunjin strain of WNV (WNVKUN) is endemic to northern Australia, but infections are usually asymptomatic. In 2011, an unprecedented outbreak of equine encephalitis occurred in southeastern Australia; most of the ≈900 reported cases were attributed to a newly emerged WNVKUN strain. To investigate the origins of this virus, we performed genetic analysis and in vitro and in vivo studies of 13 WNVKUN isolates collected from different regions of Australia during 1960–2012. Although no disease was recorded for 1984, 2000, or 2012, isolates collected during those years (from Victoria, Queensland, and New South Wales, respectively) exhibited levels of virulence in mice similar to that of the 2011 outbreak strain. Thus, virulent strains of WNVKUN have circulated in Australia for >30 years, and the first extensive outbreak of equine disease in Australia probably resulted from a combination of specific ecologic and epidemiologic conditions.
West Nile virus (WNV) is a mosquito-transmitted flavivirus that causes encephalitis. Outbreaks of potentially fatal neurologic syndromes have occurred in Europe and Africa (1); recently, however, strains of WNV have caused large outbreaks of encephalitis in humans and horses in the Americas and Australia (2,3). The Kunjin strain of WNV (WNVKUN) is indigenous to Australia and historically has caused only relatively mild, nonfatal disease in humans and horses. However, in 2011, a large unprecedented outbreak of encephalitis in horses, involving ≈900 reported cases, occurred in southeastern Australia; a high proportion of cases were attributed to the emergence of a virulent strain of WNVKUN (3,4). WNVKUN has been shown to be enzootic to northern Australia and to have episodic activity in southern regions thought to be associated with periods of heavy rainfall (5). However, the epidemiology of WNVKUN seems to have changed over the past decade; virus activity has been detected in the absence of prior flooding and in areas where it was previously not detected (4).
Studies comparing the virulence of various WNV strains in mouse models have identified several motifs, residing in both structural and nonstructural genes as well as in the 5′ and 3′ untranslated regions (UTRs). These motifs were associated with enhanced viral invasion of the central nervous system and onset of neurologic disease (5–11).
To identify potential markers of virulence of WNVKUN in Australia, we investigated evolutionary mechanisms behind the emergence of virulent strain(s) by using established mouse models to compare the neuroinvasive properties of WNVKUN isolates collected from different regions of Australia during 1960–2012. To investigate known markers of WNV virulence, we conducted comparative analyses of viral genome sequences.
Cell Culture, Virus Production, and Titration
We used 13 WNVKUN strains isolated during 1960–2012 (Table 1) and African green monkey kidney (Vero) and Aedes albopictus mosquito (C6/36) cells, cultured as previously described (3). The 1960 prototype WNVKUN strain (WNVKUN1960) was used as the attenuated reference virus and was either an unknown passage of the original isolate (WNVMRM16) or derived from an infectious clone of a plaque-purified virus (WNVMRM61C) (12,13). These isolates were previously shown to be phenotypically identical (13,14). WNV strain NY99-4132 was obtained from the US Centers for Disease Control and Prevention (Fort Collins, CO, USA) and used as a virulent control. Virus stocks and methods for determination of infectious titers have been described (3).
Antigenic Analysis
We compared reactivity of WNVKUN isolates with a panel of monoclonal antibodies (mAbs) with that of reference strains WNVKUN1960 and WNVNY99. To do so, we used a fixed-cell ELISA, as previously described (3,5,15).
Virus Replication Kinetics
We performed growth kinetics analysis by infecting Vero and C6/36 cells at a multiplicity of infection of 1 at 37°C (Vero) or 28°C (C6/36) with WNVKUN. Culture supernatants were harvested at 0, 24, 48, and 72 h after infection (Vero) and 0, 24, 48, 72, 96, and 120 h after infection (C6/36) and titrated (3). Statistical significance from 3 independent experiments was determined by using 2-way analysis of variance following log transformation (16). Mean virus titers were compared between viruses by using the Tukey method for pairwise multiple comparisons (GraphPad Prism, version 6.0; GraphPad Software Inc., San Diego, CA, USA).
Virulence in Mice
Performance of all animal procedures was approved by The University of Queensland Animal Ethics Committee. To determine virus virulence in mice, we intraperitoneally inoculated 20 Swiss white outbred CD-1 mice (weanlings [18–19 days of age] and young adults [28 days of age]) with a range of doses (0.1–10,000 PFU) of each WNV strain (Table 1) (3). The significance of clinical differences between groups was calculated by Kaplan-Meier analysis and analyzed by log-rank test where noted (GraphPad Prism, version 6.0). A virus strain was designated as virulent if survival times for mice infected with this strain (both age groups) differed significantly from those of mice infected with the attenuated reference strain WNVKUN1960. All virus strains that did not meet this criterion for virulence were designated as attenuated.
Full-length Genome Sequencing
We sequenced 9 WNVKUN genomes (Technical Appendix Table 1) by using random primer sequencing on extracts of C6/36 cell cultures (17). Viral RNA was extracted by using a MagMAX-96 Viral RNA Isolation Kit (Ambion, Waltham, MA, USA) according to the manufacturer’s instructions. cDNA synthesis and random PCR amplification were conducted according to previously described methods (18), and resultant PCR amplicons were used for sequencing library preparation. DNA libraries were prepared by using a Nextera XT DNA Sample Preparation Kit (Illumina, San Diego, CA, USA) according to the manufacturer’s protocols. Paired-end sequencing of 150-bp fragments was performed by using a MiSeq Reagent Kit V2 (300 cycles) and MiSeq Sequencing System (Illumina). Sequencing data were analyzed by using CLC Genomics Workbench version 6.5.0 (http://www.clcbio.com). The sequence data were trimmed by using quality scores specified in CLC Genomics Workbench before performing read-mapping analysis. The genome sequences were assembled by read mapping against the reference WNVKUN strain genome (GenBank accession no. JX276662) with use of default parameters in the mapping algorithm. Where gaps in the genome sequence or low coverage (<10 reads/site) were observed, conventional Sanger sequencing was performed to complete or verify the sequence. Oligonucleotide primers sequences designed for these purposes are available upon request to A.A.K or R.A.H.
Bioinformatics Analysis
We used MUSCLE, as implemented in MEGA6 (19), to align complete open reading frame (ORF) (10,320 nt) and partial (402 nt) envelope (E) gene nucleotide sequences of the newly sequenced WNVKUN strains, together with those available for 6 other WNVKUN strains and selected WNV isolates, representing different lineages. We estimated maximum-likelihood phylogenetic trees by using PhyML version 3.0 (20) and by using substitution models and rates among sites selected with JModelTest version 2.1.5 (21). We tested reliability of the inferred trees by using the bootstrap method with 1,000 replicates. All trees were rooted with analogous ORF sequences from Murray Valley encephalitis virus (GenBank accession no. NC000943) and Japanese encephalitis virus (GenBank accession no. EF571853) and visualized by using FigTree version 1.4.0 (http://tree.bio.ed.ac.uk/software/figtree/). Pairwise distances were determined at the nucleotide and amino acid levels by using the p-distance model in MEGA6.
WNVKUN Strains
A panel of previously characterized mAbs (5,15,22–24) was used to antigenically type 13 WNVKUN strains. The binding profiles of these mAbs confirmed that all WNVKUN isolates closely resembled the prototype WNVKUN1960 strain, including strong recognition by mAb 10A1, known to be specific for WNVKUN strains (Table 2) (2,24). Only WNVKUN strains isolated before 2000 were recognized by mAb 5H1, which binds a linear epitope in the αA3 motif (residues 39–53) in the methyltransferase domain of nonstructural (NS) protein 5 (15). Lack of 5H1 binding was associated with a substitution at residue 49 (Ile-Val) in αA3 (Table 3); this finding was consistent with previous mutagenesis study findings that a substitution of Ile to Ala at this residue was associated with abolition of 5H1 binding (26).
Glycosylated E Proteins
All analyzed WNVKUN isolates collected after 1960 contain glycosylated E proteins. The sequence analysis of the E gene of WNVKUN isolates revealed the presence of a conserved potential N-linked glycosylation site at residue 154 in all but the prototype WNVKUN1960 isolate (Table 3). To confirm that this site was indeed glycosylated on the viral E protein, each virus was assessed for recognition in ELISA by mAbs 17D7 and 3.101C (Table 2), which specifically recognize glycosylated and unglycosylated WNVKUN E proteins, respectively (5,23). The results supported the predictions from our sequencing data; all WNVKUN strains except the prototype WNVKUN1960 strain were recognized by mAb 17D7 but not by 3.101C.
Growth Kinetics and Plaque Morphology of WNVKUN Strains
Infection of Vero cells at 24 h postinfection demonstrated that WNVKUN1960, WNVK2499, WNVK6453, and WNVK68967 isolates yielded significantly lower titers than did WNVNY99 (p<0.05) (Figure 1, panel A). However, by 48 h postinfection, similar titers were reached for all WNV isolates except WNVKUN1960.
A similar trend was observed in C6/36 cells, in which viral titers of WNVKUN1960, WNVK2499, WNVK68967, and WNVGU1009 were significantly lower than those for WNVNY99, WNVNSW2011, and WNVBoort at 48 h after infection (p<0.05) and titers for WNVK6453, WNVK74015, and WNVGU0631 were intermediate. By 96 h after infection, the titers of all WNV isolates except WNVKUN1960 were similar.
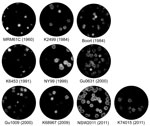
In terms of plaque morphology of WNVKUN strains, in Vero cells, WNVNSW2011 and WNVK74015 produced large plaques (average size 4.3 ± 0.63 and 4.3 ± 0.77 mm, respectively), a size similar to those produced by the WNVNY99 strain (average size 4.8 ± 0.45 mm). The prototype virus, WNVKUN1960, produced very small plaques (average size 2.7 ± 0.47 mm), which differed significantly from those of all other viruses tested during this study (p<0.0001). The remaining isolates produced intermediate-sized plaques (average size 3.5–3.9 ± 0.45–0.84 mm) (Figure 2) (5). Plaques formed by WNVK6453, WNVK74015, WNVGu1009, and WNVGu0631 were less well defined than those formed by WNVNY99, WNVNSW2011, WNVK2499, and WNVBoort.
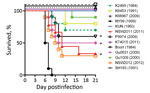
Virulence of WNVKUN Strains in Mice
We previously demonstrated that differentiation between virulent and attenuated strains of WNV can be detected in weanling and young adult mice (3,27). In this study, we found that in addition to WNVNSW2011, 3 other WNVKUN isolates (WNVBoort, WNVGu0631, WNVNSW2012) were neuroinvasive in both mouse models (Figure 3; Technical Appendix Table 2). The WNVBoort strain, obtained from the spinal cord of a symptomatic horse during a small outbreak of equine disease in southeastern Australia in 1984, was neuroinvasive in young adult mice (40% mortality rate at 1,000 PFU); this finding did not statistically differ in this respect from that for WNVNSW2011 (p = 0.3218) (Figure 3). Two other isolates obtained from mosquitoes, 1 from the Gulf of Carpentaria in 2000 (WNVGu0631) and 1 from southeastern Australia in 2012 (WNVNSW2012), also exhibited levels of neuroinvasive properties in young adult mice similar to those caused by WNVNSW2011 (Figure 3). In weanling mice, the virulence of WNVBoort, WNVGu0631, and WNVNSW2012 was also similar to that of WNVNSW2011; mortality rates, 50% lethal dose, or time to death did not differ significantly (Table 4). Of note, WNVGu1009 isolated at the same time and from the same region as WNVGU0631 was significantly less virulent in young adult mice (Figure 3; Technical Appendix Table 2). The remaining isolates were relatively attenuated in both young adult (Figure 3) and weanling (Technical Appendix Table 2) mice and did not differ significantly from the attenuated prototype WNV KUN1960 strain (p>0.05).
Sequence of Viral Genomes
We sequenced WNVKUN isolates to analyze their relationship to the prototype WNVKUN strains from 1960 (WNVMRM16, WNVMRM61C); the 2011 outbreak strains (WNVNSW2011, WNVSA2011, WNVV11-03, and WNVV11-07); and exotic strains of WNV known to be virulent in humans and horses (WNVNY99) or representing different WNV lineages. Phylogenetic analysis of the ORF sequences demonstrated that the WNVKUN strains form a single genetically homogeneous clade within lineage 1 (Figure 4), as previously recognized (24); nucleotide and amino acid identities between strains were 96.1%–99.4% and 98.5%–100%, respectively. As expected, the most recent isolates, including the 2011 outbreak strain, were the most divergent, and the early prototype strains (WNVMRM61C and WNVMRM16) occupied the basal lineage of this clade. Recent strains isolated in 2011 and 2012 from southeastern Australian states clustered together and shared high levels of nucleotide (98.6%–100%) and amino acid (99.6%–100%) identities, indicating transmission of a genetically homogeneous virus population during this period. These strains were either virulent for horses or shown in this study to be virulent in mice (Figure 3; Technical Appendix Table 2). No other association between phylogenetic relationships and virulence was found; other virulent strains clustered closely and interspersed with the attenuated strains identified in this study. An expanded phylogenetic analysis that used 45 partial E gene sequences (402 nt) and a larger range of reference WNVKUN strains showed a similar pattern of relationships (Technical Appendix Figure).
Of note is the very close relationship (99.9% aa identity) between a virulent 2011 strain isolated from a horse and an isolate obtained from Culex annulirostris mosquitoes trapped in New South Wales, Australia, in 2012 (WNVNSW2012). Only 3 nonconservative changes were identified between WNVNSW2011 and WNVNSW2012, located in NS1 (Lys33Arg), NS3 (Phe509Leu), and NS4A (Phe92Leu). These results suggest that the virulent strain either had persisted in New South Wales after the end of the 2011 outbreak or had been reintroduced to the area.
Analyses of predicted gene products from the complete ORF sequence of each WNVKUN isolate revealed that, in addition to the glycosylation site at residues 154–156 in the E protein, all strains isolated after 1960 contained a Phe residue at position 653 in the NS5 protein, which has previously been shown to play a role in resistance to antiviral activity of interferon-α/β (10) (Table 3). In contrast, WNVKUN1960 contained a Ser residue at position 653 in NS5 (3,5,24,28). The Pro residue at position 249 in the NS3 protein, previously shown to be present in WNV strains and associated with increased virulence in birds of some species (25), was not present in any of the WNVKUN isolates, which all contained an Ala residue at this position (Table 3).
In addition to an Ile→Val substitution at position 49 in NS5 of WNVKUN isolates collected after 2009, analysis of more contemporary WNVKUN isolates also revealed a consistent 8-nt deletion in the 3′ UTR, just downstream from the ORF stop codon. This deletion was identified in WNVGu1009 and all isolates collected after 2000. In contrast, this deletion was not present in isolates collected before 2000 (Table 3) or in another isolate from Gulf of Carpentaria collected in 2000 (WNVGu0631). We suggest that these 2 features (Ile→Val 49 residue in NS5 and an 8-nt deletion in the 3′ UTR) can be considered as potential evolutionary markers.
In addition to the genetic variability described above, sequence analysis between virulent and attenuated WNVKUN strains identified other nucleotide differences between isolates, located throughout the viral genome. These differences result in amino acid substitutions (Table 4) and may contribute to observed phenotypic differences.
We also sequenced WNVKUN viral RNA extracted directly from mosquito saliva expectorated onto sugar-soaked nucleic acid preservation cards placed in mosquito traps in Darwin, Northern Territory, in 2012 (WNVNT2012) (29,30). When partial sequences from E, NS5, and the 3′ UTR from this RNA were aligned, we observed a high level (99.7%) of identity with the WNVKUN1960 strains, indicating that viruses genetically homologous to the prototype virus are still circulating in some regions of Australia (online Technical Appendix Figure). Closer analysis revealed a lack of E glycosylation, similar to that found in the prototype strain. However, Phe was identified at position 653 of NS5, similar to that found in recent isolates.
Historically, WNVKUN has been associated with only mild disease in humans and rare cases of disease in horses, consistent with data from mouse virulence studies that revealed a relatively attenuated phenotype (3,8,27). Thus, the emergence of an equid-virulent strain of WNVKUN, responsible for ≈900 cases of encephalitis in horses in southeastern Australia, was unprecedented.
Although most WNVKUN isolates examined in this study exhibited an attenuated phenotype, similar to that of the prototype WNVKUN1960, we identified an additional 3 strains with neuroinvasive properties in mice similar to those reported for WNVNSW2011 (3). The first, WNVBoort, was isolated from the spinal cord of a horse with nonsuppurative encephalomyelitis and severe neurologic symptoms in northern Victoria in 1984 (31). At that time, 53 animals in the same area were clinically affected. However, a high incidence of Ross River virus–specific antibody in these animals implicated that virus rather than WNVKUN as the primary etiologic agent (31). Our results are also supported by another recent study showing virulence of WNVBoort in 18–19-day-old mice (32).
The second virulent strain identified in this study, WNVGu0631, was isolated from Cx. annulirostris mosquitoes collected from Normanton, Gulf of Carpentaria, in April 2000. Of note, this virus was isolated in the absence of any reported disease outbreak, as part of a survey for the presence of Japanese encephalitis virus in northern Queensland (33). The second Gulf of Carpentaria isolate, WNVGu1009, was also collected in April 2000, from the town of Karumba, which is ≈30 km from Normanton. However, WNVGU1009 is genetically distinct and attenuated to the same degree as the prototype WNVKUN1960 in 28-day-old mice (Figure 4). These observations demonstrated that virulent WNVKUN strains might co-circulate with attenuated strains in some regions of Australia. Furthermore, the circulation of neuroinvasive strains may often appear in the absence of disease outbreaks. This suggestion is consistent with our finding that WNVNSW2012 was genetically almost identical to the WNVNSW2011 and exhibited similar levels of neuroinvasiveness in mice. However, no cases of disease in equids were associated with WNVKUN infection during the 2012 season (3,4,34). This lack of cases suggests that the persistence of virulent strains in southeastern Australia is not the sole determinant for initiating disease outbreaks and that specific climatic and ecologic conditions, perhaps influencing mosquito populations and viral transmission, are also required.
A similar scenario occurred in North America, where an unusually high number of cases in humans (5,387), most in Texas, USA, were reported in 2012. However, sequence analysis of WNV isolates from 2012 revealed that the strains circulating in Texas were virulent and attenuated, and no specific virulence determinants responsible for the increase in cases could be identified (35). Instead, other factors, including temperature and changes in mosquito or bird populations, were speculated to have contributed to the magnitude of the 2012 outbreak (36).
To identify a phylogenetic association with virulence and to identify potential virulence determinants encoded in the genome of WNVKUN strains, we also performed full-length sequencing of the ORF of several of the viruses studied. Although recent virulent strains were phylogenetically closely related, no other association between phylogenetic grouping and virulence phenotype was found (Figure 4; Technical Appendix Figure). One notable change in the genome that was clearly associated with the temporal distribution of these viruses was a highly conserved 8-base deletion in the 3′ UTR, just downstream of the ORF stop codon. Isolates from samples collected after 2000, including the virulent WNVNSW2011 and attenuated strains, invariably contained this deletion. This finding suggests that the deletion is an evolutionary marker but is not directly associated with virulence. This finding is also consistent with our observation that the neuroinvasive 2000 Gulf of Carpentaria isolate, WNVGu0631, did not have this deletion but that the co-circulating attenuated isolate, WNVGu1009, collected from the same region at the same time, did have this deletion.
An additional evolutionary change was observed in the α-A3 motif of the methyltransferase domain of the NS5 protein. Isolates obtained before 2009, including the prototype WNVKUN1960, contained a conserved Ile residue at position 49. However, all isolates collected after 2009 displayed an Ile→Val substitution at this position. Coincidentally, this substitution abolished the binding of a mAb (5H1) that recognizes a linear epitope comprising the α-A3 peptide (15).
Initial comparisons between the virulent isolate WNVNSW2011 from a horse and the attenuated prototype WNVKUN1960 revealed that several previously identified WNV virulence markers were detected in the former but not in the latter isolate (3). These markers included the conserved N-linked glycosylation of the E protein (7) and the Phe residue at position 653 in the NS5 protein, associated with resistance to antiviral activity of interferon α/β (8). Although these initial observations suggested the involvement of these motifs in the enhanced neuroinvasive properties of the isolate collected from a horse in 2011, our study revealed that, with the exception of WNVKUN1960, all strains examined contain both of these markers, regardless of virulence phenotype in mouse models. Thus, it seems that, although these motifs contribute to virulence in mice, they are not likely to be solely responsible for enhancing the neuroinvasive properties of some WNVKUN strains and, hence, not likely to be markers of evolving virulence in recent isolates of WNVKUN.
Additional markers of WNV virulence identified in WNV strains from North America were not detected in any of the WNVKUN isolates. This finding is consistent with our repeated observations that even the equid-virulent WNVNSW2011 is substantially less neuroinvasive than WNVNY99 in young adult mice (3). These motifs may include Val 22 and Ser 72 residues in the premembrane, which enhance mouse neuroinvasiveness when introduced into the prototype WNVKUN1960 (11), and the Pro residue at position 249 in NS3, which is associated with enhanced virulence in birds (25). The absence of the latter motif in all WNVKUN strains is also consistent with the perceived lack of illness and death among birds in Australia, notably during the 2011 outbreak among equids. Some isolates included in this study (including WNVKUN1960, WNVSH183, WNVBoort, and WNVHu6774) have an unknown passage history. Extensive passage through cells is known to occasionally lead to passage-adapted mutations, and care should be taken when interpreting sequencing data from these virus strains.
WNVKUN is thought to be endemic to the tropical areas of northern Australia, suggesting that virulent viruses emerging in southeastern Australia probably originate from northern Australia. However, WNVKUN recently isolated from mosquitoes in northern Australia, including the 2011 Kimberley isolate WNVK74015, were more attenuated than WNVNSW2011. This finding suggests a different explanation for the evolution of virulent WNVKUN viruses, which may be associated with the adaption of WNVKUN to different hosts (avian and terrestrial) or different vector species in temperate regions. In this context, virulence in equids may be just a coincidental outcome of the constraints placed on virus fitness in different geographic locations (35–37).
Overall, our results show that virulent strains of WNVKUN have been circulating in Australia for >30 years and that the first extensive outbreak of disease among horses in Australia in 2011 probably resulted from a combination of ecologic and epidemiologic conditions rather than the emergence of a novel, more virulent strain. Further studies evaluating viral fitness of West Nile virus quasispecies in terms of population-dependent host–virus interactions, are warranted.
Dr. Prow is a postdoctoral research fellow at the QIMR Berghofer Medical Research Institute and is engaged in research to characterize the pathogenesis and virulence of WNV strains in murine models. She also has a special interest in understanding the neuroinvasive properties of neurotrophic flaviviruses.
Acknowledgments
We thank Mary Carr for providing clinical material used for the isolation of WNVSA2011, Melissa Sanchez and Robert Doms for providing mAb 17D7, and the Western Australian Department of Health for funding and assisting with the detection of WNVKUN isolates in Western Australia. We also thank Richard Russell for providing the WNVNSW2012 isolate.
This work was supported by the Australian Research Council Linkage Project grant LP120100686 and the National Health and Medical Research Council project grant APP1045188.
References
- Murgue B, Zeller H, Deubel V. The ecology and epidemiology of West Nile virus in Africa, Europe and Asia. Curr Top Microbiol Immunol. 2002;267:195–221. DOIPubMedGoogle Scholar
- Lanciotti RS, Roehrig JT, Deubel V, Smith J, Parker M, Steele K, Origin of the West Nile virus responsible for an outbreak of encephalitis in the northeastern United States. Science. 1999;286:2333–7. DOIPubMedGoogle Scholar
- Frost MJ, Zhang J, Edmonds JH, Prow NA, Gu X, Davis R, Characterization of virulent West Nile virus Kunjin strain, Australia, 2011. Emerg Infect Dis. 2012;18:792–800. DOIPubMedGoogle Scholar
- Roche SE, Wicks R, Garner MG, East IJ, Paskin R, Moloney BJ, Descriptive overview of the 2011 epidemic of arboviral disease in horses in Australia. Aust Vet J. 2013;91:5–13. DOIPubMedGoogle Scholar
- Adams SC, Broom AK, Sammels LM, Hartnett AC, Howard MJ, Coelen RJ, Glycosylation and antigenic variation among Kunjin virus isolates. Virology. 1995;206:49–56. DOIPubMedGoogle Scholar
- Audsley M, Edmonds JH, Liu W, Mokhonov V, Mokhonova E, Melian EB, Virulence determinants between New York 99 and Kunjin strains of West Nile virus. Virology. 2011;414:63–73. DOIPubMedGoogle Scholar
- Beasley DW, Whiteman MC, Zhang S, Huang CY, Schneider BS, Smith DR, Envelope protein glycosylation status influences mouse neuroinvasion phenotype of genetic lineage 1 West Nile virus strains. J Virol. 2005;79:8339–47. DOIPubMedGoogle Scholar
- Daffis S, Lazear HM, Liu WJ, Audsley M, Engle M, Khromykh AA, The naturally attenuated Kunjin strain of West Nile virus shows enhanced sensitivity to the host type I interferon response. J Virol. 2011;85:5664–8 .DOIPubMedGoogle Scholar
- Keller BC, Fredericksen BL, Samuel MA, Mock RE, Mason PW, Diamond MS, Resistance to alpha/beta interferon is a determinant of West Nile virus replication fitness and virulence. J Virol. 2006;80:9424–34. DOIPubMedGoogle Scholar
- Laurent-Rolle M, Boer EF, Lubick KJ, Wolfinbarger JB, Carmody AB, Rockx B, The NS5 protein of the virulent West Nile virus NY99 strain is a potent antagonist of type I interferon-mediated JAK-STAT signaling. J Virol. 2010;84:3503–15. DOIPubMedGoogle Scholar
- Setoh YX, Prow NA, Hobson-Peters J, Lobigs M, Young PR, Khromykh AA, Identification of residues in West Nile virus pre-membrane protein that influence viral particle secretion and virulence. J Gen Virol. 2012;93:1965–75. DOIPubMedGoogle Scholar
- Doherty RL, Carley JG. Studies of arthropod-borne virus infections in Queensland. II. Serological investigations of antibodies to dengue and Murray Valley encephalitis in eastern Queensland. Aust J Exp Biol Med Sci. 1960;38:427–39. DOIPubMedGoogle Scholar
- Khromykh AA, Kenney MT, Westaway EG. Trans-complementation of flavivirus RNA polymerase gene NS5 by using Kunjin virus replicon–expressing BHK cells. J Virol. 1998;72:7270–9.PubMedGoogle Scholar
- Westaway EG. Assessment and application of a cell line from pig kidney for plaque assay and neutralization tests with twelve group B arboviruses. Am J Epidemiol. 1966;84:439–56.PubMedGoogle Scholar
- Hall RA, Tan SE, Selisko B, Slade R, Hobson-Peters J, Canard B, Monoclonal antibodies to the West Nile virus NS5 protein map to linear and conformational epitopes in the methyltransferase and polymerase domains. J Gen Virol. 2009;90:2912–22. DOIPubMedGoogle Scholar
- Wang GP, Bushman FD. A statistical method for comparing viral growth curves. J Virol Methods. 2006;135:118–23. DOIPubMedGoogle Scholar
- Warrilow D, Hall-Mendelin S, Hobson-Peters J, Prow NA, Allcock R, Hall RA. Complete coding sequences of three members of the kokobera group of flaviviruses. Genome Announc. 2014;2:e00890–14.DOIPubMedGoogle Scholar
- Palacios G, Quan PL, Jabado OJ, Conlan S, Hirschberg DL, Liu Y, Panmicrobial oligonucleotide array for diagnosis of infectious diseases. Emerg Infect Dis. 2007;13:73–81. DOIPubMedGoogle Scholar
- Tamura K, Stecher G, Peterson D, Filipski A, Kumar S. MEGA6: Molecular Evolutionary Genetics Analysis version 6.0. Mol Biol Evol. 2013;30:2725–9. DOIPubMedGoogle Scholar
- Guindon S, Gascuel O. A simple, fast, and accurate algorithm to estimate large phylogenies by maximum likelihood. Syst Biol. 2003;52:696–704. DOIPubMedGoogle Scholar
- Darriba D, Taboada GL, Doallo R, Posada D. jModelTest 2: more models, new heuristics and parallel computing. Nat Methods. 2012;9:772. DOIPubMedGoogle Scholar
- Hall RA, Kay BH, Burgess GW, Clancy P, Fanning ID. Epitope analysis of the envelope and non-structural glycoproteins of Murray Valley encephalitis virus. J Gen Virol. 1990;71:2923–30. DOIPubMedGoogle Scholar
- Hobson-Peters J, Toye P, Sanchez MD, Bossart KN, Wang LF, Clark DC, A glycosylated peptide in the West Nile virus envelope protein is immunogenic during equine infection. J Gen Virol. 2008;89:3063–72. DOIPubMedGoogle Scholar
- Scherret JH, Poidinger M, Mackenzie JS, Broom AK, Deubel V, Lipkin WI, The relationships between West Nile and Kunjin viruses. Emerg Infect Dis. 2001;7:697–705. DOIPubMedGoogle Scholar
- Brault AC, Huang CY, Langevin SA, Kinney RM, Bowen RA, Ramey WN, A single positively selected West Nile viral mutation confers increased virogenesis in American crows. Nat Genet. 2007;39:1162–6. DOIPubMedGoogle Scholar
- Tan CS, Hobson-Peters JM, Stoermer MJ, Fairlie DP, Khromykh AA, Hall RA. An interaction between the methyltransferase and RNA dependent RNA polymerase domains of the West Nile virus NS5 protein. J Gen Virol. 2013;94:1961–71. DOIPubMedGoogle Scholar
- Prow NA, Setoh YX, Biron RM, Sester DP, Kim KS, Hobson-Peters J, The West Nile virus-like flavivirus Koutango is highly virulent in mice due to delayed viral clearance and the induction of a poor neutralizing antibody response. J Virol. 2014;88:9947–62. DOIPubMedGoogle Scholar
- Wright PJ, Warr HM, Westaway EG. Synthesis of glycoproteins in cells infected by the flavivirus Kunjin. Virology. 1981;109:418–27. DOIPubMedGoogle Scholar
- Hall-Mendelin S, Ritchie SA, Johansen CA, Zborowski P, Cortis G, Dandridge S, Exploiting mosquito sugar feeding to detect mosquito-borne pathogens. Proc Natl Acad Sci U S A. 2010;107:11255–9. DOIPubMedGoogle Scholar
- van den Hurk AF, Hall-Mendelin S, Townsend M, Kurucz N, Edwards J, Ehlers G, Applications of a sugar-based surveillance system to track arboviruses in wild mosquito populations. Vector Borne Zoonotic Dis. 2014;14:66–73. DOIPubMedGoogle Scholar
- Badman R, Campbell J, Aldred J. Arbovirus infection in horses—Victoria. Commun Dis Intell. 1984;17:5–6.
- Bingham J, Payne J, Harper J, Frazer L, Eastwood S, Wilson S, Evaluation of a mouse model for the West Nile virus group for the purpose of determining viral pathotypes. J Gen Virol. 2014;95:1221–32. DOIPubMedGoogle Scholar
- van den Hurk AF, Nisbet DJ, Foley PN, Ritchie SA, Mackenzie JS, Beebe NW. Isolation of arboviruses from mosquitoes (Diptera: Culicidae) collected from the Gulf Plains region of northwest Queensland, Australia. J Med Entomol. 2002;39:786–92. DOIPubMedGoogle Scholar
- Prow NA. The changing epidemiology of Kunjin virus in Australia. Int J Environ Res Public Health. 2013;10:6255–72. DOIPubMedGoogle Scholar
- Coffey LL, Forrester N, Tsetsarkin K, Vasilakis N, Weaver SC. Factors shaping the adaptive landscape for arboviruses: implications for the emergence of disease. Future Microbiol. 2013;8:155–76. DOIPubMedGoogle Scholar
- Duggal NK, D’Anton M, Xiang J, Seiferth R, Day J, Nasci R, Sequence analyses of 2012 West Nile virus isolates from Texas fail to associate viral genetic factors with outbreak magnitude. Am J Trop Med Hyg. 2013;89:205–10. DOIPubMedGoogle Scholar
- van den Hurk AF, Hall-Mendelin S, Webb CE, Tan CS, Frentiu FD, Prow NA, Role of enhanced vector transmission of a new West Nile virus strain in an outbreak of equine disease in Australia in 2011. Parasit Vectors. 2014;7:586.DOIPubMedGoogle Scholar
Figures
Tables
Cite This Article1Current affiliation: QIMR Berghofer Medical Research Institute, Herston, Queensland, Australia.
2These authors contributed equally to the major technical aspects of the work.
3Current affiliation: Willamette University, Salem, Oregon, USA.
4These authors served as joint senior authors.
Table of Contents – Volume 22, Number 8—August 2016
| EID Search Options |
|---|
|
|
|
|
|
|


Please use the form below to submit correspondence to the authors or contact them at the following address:
Roy A. Hall or Alexander A. Khromykh, The University of Queensland, St. Lucia, 4072, Queensland, Australia; or
Top