Volume 23, Number 4—April 2017
Research
Three Divergent Subpopulations of the Malaria Parasite Plasmodium knowlesi
Cite This Article
Citation for Media
Abstract
Multilocus microsatellite genotyping of Plasmodium knowlesi isolates previously indicated 2 divergent parasite subpopulations in humans on the island of Borneo, each associated with a different macaque reservoir host species. Geographic divergence was also apparent, and independent sequence data have indicated particularly deep divergence between parasites from mainland Southeast Asia and Borneo. To resolve the overall population structure, multilocus microsatellite genotyping was conducted on a new sample of 182 P. knowlesi infections (obtained from 134 humans and 48 wild macaques) from diverse areas of Malaysia, first analyzed separately and then in combination with previous data. All analyses confirmed 2 divergent clusters of human cases in Malaysian Borneo, associated with long-tailed macaques and pig-tailed macaques, and a third cluster in humans and most macaques in peninsular Malaysia. High levels of pairwise divergence between each of these sympatric and allopatric subpopulations have implications for the epidemiology and control of this zoonotic species.
Plasmodium knowlesi is a zoonotic malaria parasite that has only recently been recognized as a notable cause of malaria (1). Although cases have now been seen in most countries in Southeast Asia, the largest numbers have been reported in Malaysia (1–4). The extent to which this is a result of varying efforts in diagnosis is unclear, as specific molecular identification is required to discriminate P. knowlesi from other malaria parasite species. Moreover, although most reports are of cases presenting with clinical symptoms, asymptomatic infections may also occur (5).
The Plasmodium knowlesi parasite is transmitted by mosquitoes to humans from monkey reservoir hosts, with different Anopheles species of the Leucosphyrus group having been incriminated as potential vectors in different areas (1,6). Two macaque species, the long-tailed macaque (Macaca fascicularis) and the pig-tailed macaque (M. nemestrina), are the major reservoirs of infection (7,8). Human infections in Malaysian Borneo, the portion of Malaysia on the island of Borneo, have divergent genetic subpopulations that are seen in the different macaque species locally, indicating that 2 independent zoonoses may be occurring sympatrically (9). Noticeable geographic differentiation of parasites between Malaysian Borneo and peninsular Malaysia was also evident in microsatellite analysis; separate studies have revealed divergence between the 2 regions at unlinked genes encoding the normocyte binding protein (10–12) and the Duffy binding protein (13,14), as well as the 18S rRNA and mitochondrial cytochrome oxidase subunit 1 (15). Whole-genome sequencing has confirmed the presence of 2 divergent subpopulations of P. knowlesi in Malaysian Borneo and revealed a third divergent cluster of laboratory isolates maintained in laboratories since the 1960s; most of these were recorded to have originated from peninsular Malaysia (16).
To resolve the population structure in relation to host species and geography, a new collection of 182 P. knowlesi infection samples from humans and wild macaques living in diverse areas of Malaysia was genotyped at 10 microsatellite loci. We first analyzed the new dataset separately and then analyzed a combined dataset incorporating previous multilocus microsatellite data, using several independent and complementary statistical approaches to identify genetic substructure. All analyses revealed that 2 divergent genetic subpopulations of human cases occur sympatrically in Malaysian Borneo, detected separately in long-tailed macaques and pig-tailed macaques in the same region, whereas a third divergent genetic subpopulation occurs in humans and most macaques in peninsular Malaysia. This parasite species has undergone different sympatric and allopatric processes of divergence, which will affect its future adaptation to a changing environmental landscape. Current differences between the subpopulations need to be recognized in clinical and epidemiologic studies.
Study Sites and DNA Samples
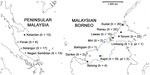
We obtained blood samples infected with P. knowlesi from human clinical cases at 7 sites and from macaque hosts at 8 sites across Malaysia (Figure 1). We extracted DNA from anticoagulated venous blood samples or dried blood spots, and tested the DNA for the presence of different malaria parasite species by species-specific PCR using methods described previously (7). Samples from 134 P. knowlesi–positive human cases collected during 2012–2014 that had sufficient DNA for multilocus genotyping originated from Kapit (n = 35), Betong (n = 4), and Lawas (n = 15) in Sarawak state, Malaysian Borneo; from Kudat (n = 20), Ranau (n = 25), and Tenom (n = 22) in Sabah state, Malaysian Borneo; and from Kelantan (n = 13) in peninsular Malaysia.
Samples that were collected during 2007–2014 from 48 P. knowlesi–positive macaques had sufficient DNA for multilocus genotyping. Most were from long-tailed macaques, sampled from Selangor (n = 17), Perak (n = 6), and Negeri Sembilan (n = 15) in peninsular Malaysia and from Balingian (n = 1), Limbang (n = 2), Miri (n = 2), and Sarikei (n = 2) in Sarawak; pig-tailed macaque samples were from Limbang (n = 1) and Kapit (n = 2) in Sarawak. We performed the sampling according to the protocols of the Department of Wildlife and National Parks in Malaysia. We included DNA of P. knowlesi strain Nuri (kindly provided by Clemens Kocken at the Biomedical Primate Research Centre, the Netherlands) in the genotyping as a control (17).
Microsatellite Genotyping of New Samples
We genotyped each of the P. knowlesi–positive DNA samples at 10 microsatellite loci (NC03_2, CD05_06, CD08_61, NC0AU: 9_1, NC10_1, CD11_157, NC12_2, NC12_4, CD13_61, CD13_107) using hemi-nested PCR assays specific for P. knowlesi, as described previously (9). We analyzed fluorescent dye-labeled PCR products by using capillary electrophoresis on the Genetic Analyzer 3730 (Applied Biosystems, Cheshire, UK), with GeneScan 500 LIZ internal size standards, following which we scored alleles and peak heights with GeneMapper version 4.0 software (Applied Biosystems).
The genotypic multiplicity of infection (MOI) was defined as the maximum number of alleles detected at any individual locus. Electrophoretic peak heights above 200 fluorescent units of the expected molecular sizes were scored as alleles, and secondary peaks within an infection sample were scored if they had a height of at least 25% relative to the predominant allele. We determined the multilocus genotype profile of each infection, and allele frequency counts for population samples, by counting the predominant allele at each locus within each infection.
Analysis of Microsatellite Genotypes from Previous Data
We retrieved whole genome sequence data of P. knowlesi samples from the European Nucleotide Archive (http://www.ebi.ac.uk/ena), and we obtained the reference genome sequence of strain H from GeneDB (http://www.genedb.org/Homepage/Pknowlesi). Most of the parasite genome short-read Illumina sequences available are from patients sampled in Malaysian Borneo (12,16), but a few are from older laboratory lines that originated from peninsular Malaysia, as well as 1 supposedly from the Philippines (16). Although genome sequences indicate some historical mislabeling or contamination of the laboratory lines, meaning that individual identities are in question, it is clear that most are from peninsular Malaysia (16). We aligned the raw short reads to P. knowlesi genome strain H by using the BWA-MEM alignment tool with default parameters (https://arxiv.org/pdf/1303.3997.pdf). We identified lists of indels using the SAMTools and VCFtools software (18,19) with the following parameters, described elsewhere (16): mpileup –B –Q 23 –d 2000 –C 50 -ugf; varFilter –d 10 –D 2000. Using ARTEMIS software (20), we determined the putative microsatellite allele size by inspecting the indels within the location of the PCR primers used for the second amplification PCR. We assessed the quality of the mapping within the microsatellite allele regions with the minimum depth of short-read coverage at 30-fold.
Analyses of P. knowlesi Population Genetic Substructure
We evaluated population genetic structure by Bayesian clustering inference using STRUCTURE version 2.3.4 software (21), on samples for which there were no missing data at any locus. First, to allocate the probable ancestral assignment of a genotype into 1 or more K clusters, we set the parameters for the admixture model on the basis of correlated allele frequency, without providing the sample source information. However, the sensitivity for population structure analysis can be improved by providing population information, in which an algorithm assumes that the probability of an individual being part of a population varies among locations or sources of origins (22). For the second test, we set the parameter to LOCPRIOR. This parameter is informative when population structure signals are weak because of a close relationship between populations. We performed both LOCPRIOR and non-LOCPRIOR parameters in STRUCTURE runs separately with a burn-in period of 50,000 followed by 100,000 Markov chains (MCMC iterations). The simulations were replicated 20 times for K values ranging from 1 to 10. The optimal K value was calculated based on Evanno’s method of ΔK statistics implemented in the STRUCTURE HARVESTER webpage interface (23,24). For the optimum K, we aligned the 20-replicate runs at 10,000 permutations to determine the consensus of cluster scores using CLUMPP version 1.1.2 (25).
To evaluate population structure independently, we performed principal coordinate analysis (PCoA) using the GenAlEx package version 6 implemented in Microsoft Excel (26). We first generated a genetic distance matrix using the multilocus microsatellite dataset, and we plotted a 2-dimensional PCoA based on the first 2 highest eigenvalues. We calculated the K-means clusters using the first and second eigenvectors generated from the PCoA, and subsequently used them to assign each individual infection to the most probable cluster. In addition, we applied the discriminant analysis of principal component from the adegenet 2.0.0 packages in R to assess the population structure (27). In this procedure, we first transformed genotype data into 40 uncorrelated principal components, and then, using the discriminant function, we partitioned the variances into within-group and among-group components, while optimizing separations between groups.
We calculated pairwise differentiation (FST) between different subpopulations of P. knowlesi by using FSTAT software version 2.9.3.2 (28). We estimated the mean allelic diversity across loci, measured as expected heterozygosity (HE), using FSTAT software. We assessed multilocus linkage disequilibrium with the standardized index of association (IAS), calculated by LIAN version 3.7 (29), with Monte Carlo simulation of 10,000 data permutations.
Genotypic Diversity within P. knowlesi Infections
Of 182 P. knowlesi infections genotyped for this study (134 from humans, 45 from long-tailed macaques, and 3 from pig-tailed macaques), 166 (91.2%) yielded complete genotype data for the panel of 10 microsatellite loci, whereas the remainder were each genotyped for at least 7 of the loci (Table; Technical Appendix 1).
Among the human cases, single genotype infections were common, and the average number of genotypes per infection (MOI) was less than 2 at all sites sampled. This was expected when these samples were collectively reanalyzed with P. knowlesi infections of humans and macaques across Malaysia from previous studies (Technical Appendix 2 Table 1). We found no notable difference in numbers of genotypes per infection in Malaysian Borneo and peninsular Malaysia (mean MOI values of 1.50 and 1.77, respectively; p = 0.14 by Fisher exact test). In contrast, multiple genotype infections were more common in macaques both in Malaysian Borneo (mean MOI = 2.10, p = 6.7 × 10−3) and peninsular Malaysia (mean MOI = 2.39, p = 9.8 × 10−4) (Table; Figure 2). We counted the predominant allele at each locus per infection for subsequent statistical analyses on population structure.
Analysis of P. knowlesi Population Genetic Structure with New Samples
Bayesian clustering analyses using 2 admixture models on the new sample of 166 infections with complete genotype data for the full panel of 10 microsatellite loci identified 3 subpopulation clusters (K = 3; Figure 3; Technical Appendix 2 Figures 1 and 2, panel A), hereafter referred to as clusters 1–3. Human infections in Malaysian Borneo were assigned to clusters 1 and 2, whereas long-tailed macaque infections were all in cluster 1 and pig-tailed macaque infections were mostly in cluster 2 (1 pig-tailed macaque infection was assigned as intermediate between clusters 2 and 3), confirming the existence of 2 major sympatric subpopulations in Malaysian Borneo, as reported previously (9,12,16).
Among the samples from peninsular Malaysia, those from human cases were all assigned to cluster 3, along with most of the infections from wild long-tailed macaques sampled in Kelantan, although long-tailed macaque infections from the other 2 sites had more intermediate cluster assignments, suggesting some ancestral affinity with cluster 2. All the laboratory isolates, originating many years ago mainly from peninsular Malaysia, were clearly assigned to cluster 3, consistent with results of a recent whole genome sequence analysis (16).
Analysis of Population Genetic Structure Incorporating New and Previously Acquired Microsatellite Data
To further evaluate the population structure of P. knowlesi, we collated the dataset in this study with data from samples analyzed previously (9). This yielded a total of 758 P. knowlesi infections with the complete panel of 10 microsatellite loci genotyped. This total comprises 166 samples from the present study (Table), 556 previously genotyped samples, 29 samples that had undergone repeat genotyping for all 10 loci completed here (Technical Appendix 2 Figure 3), and 7 derived from Illumina short-read sequence data.
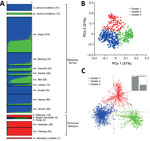
The admixture STRUCTURE analysis without the LOCPRIOR model identified 2 subpopulation clusters (K = 2; Technical Appendix 2 Figure 2, panel B, and 4). This was consistent with a previous analysis showing that human cases in the Malaysian Borneo group fell into 2 different genotype clusters, which are also respectively seen in long-tailed and pig-tailed macaque infections, although the current analysis assigned samples from peninsular Malaysia to cluster 2 (previously, they had been grouped into cluster 1). However, incorporation of the LOCPRIOR model showed 3 subpopulation clusters (K = 3; Figure 4, panel A; Technical Appendix 2 Figure 4), with most of the isolates from peninsular Malaysia belonging to cluster 3, as also seen with the analysis based solely on the new samples. Overall, this confirms that human P. knowlesi infections in Malaysian Borneo are divided into 2 different genetic subpopulations that are associated with different macaque reservoir host species, whereas human infections in peninsular Malaysia belong to a third subpopulation that is also seen in long-tailed macaques at 1 of the sites in peninsular Malaysia.
Robustness and Divergence of Subpopulation Clusters
Using an a priori designation of 3 subpopulation clusters (K = 3), we independently assigned all 758 infections into clusters using PCoA (Figure 4, panel B) and discriminant analysis (Figure 4, panel C), and compared the results with those derived from the STRUCTURE analysis (Figure 4, panel A). These showed highly concordant results (Technical Appendix 1). PCoA indicated that infections in humans were strongly associated with infections in local macaque reservoir hosts for both Malaysian Borneo and peninsular Malaysia (online Technical Appendix 2 Figure 5). Discriminant analysis also showed clear clustering, with only minimal overlap among the inertia ellipses for the 3 major clusters.
To test the consistency and robustness of cluster assignment for all 758 infections, across the different methods used (Bayesian analysis using STRUCTURE, principal coordinates analysis, and discriminant analysis), we assessed a consensus for each individual (Technical Appendix 1; Technical Appendix 2 Table 2). A large majority (86.4%) of infections were assigned into the same cluster by all 3 methods (cluster 1, n = 384; cluster 2, n = 175; cluster 3, n = 96). Most of the remainder (12.9% of the total) had an agreed assignment for 2 of the methods (cluster 1, n = 65; cluster 2, n = 16; cluster 3, n = 17), whereas only 5 (0.7%) showed no agreement across the methods. Omitting the few infections that did not show agreement for 2 or more methods yielded a dataset of 753 P. knowlesi infections that grouped into 3 major subpopulation clusters (cluster 1, n = 449; cluster 2, n = 191; cluster 3, n = 113; Technical Appendix 2 Table 3). We estimated values of allelic diversity (HE) between 0.51 and 0.83 among different sites at each of the subpopulation clusters (Technical Appendix 2 Table 4), and we observed similar patterns even without separating the infections by subpopulation cluster assignments (Technical Appendix 2 Table 5). The index of multilocus linkage disequilibrium yielded various degrees (IAS range from −0.007 to 0.305), with loss of significance at majority of the sites among the 3-subpopulation clusters (Technical Appendix 2 Table 4). However, the degree of significance increased when all infections were not assigned into subpopulation clusters (IAS range from −0.002 to 0.242 with p<0.01 at most sites; Technical Appendix 2 Table 5). Analyses of allele frequencies across all 10 microsatellite loci confirmed strong genetic differentiation among these clusters (FST = 0.184 between clusters 1 and 2; FST = 0.152 between clusters 1 and 3; FST = 0.201 between clusters 2 and 3; p<3.3 × 10−4 for each comparison using 3,000 randomized permutations). This indicates deep divergence among the 3 major parasite subpopulations that infect humans, 2 of which are sympatric and predominantly associated with different reservoir hosts (long-tailed and pig-tailed macaques in Malaysian Borneo), and 1 of which is allopatric in a different geographic region (peninsular Malaysia).
Three major subpopulations of P. knowlesi have been demonstrated in natural human infections in Malaysia. These subpopulations show profound divergence, with pairwise FST values of ≈0.2, suggesting minimal or no current gene flow between parasites in Malaysian Borneo and peninsular Malaysia, nor between parasites in long-tailed and pig-tailed macaque hosts within Malaysian Borneo.
The existence of 3 divergent clusters was initially indicated from whole genome sequence-based single nucleotide polymorphism analysis of P. knowlesi clinical isolates and laboratory lines (16). Whereas 2 of the clusters of genome sequences (clusters 1 and 2) had been seen in clinical infections in Malaysian Borneo, the third (cluster 3) was seen only in old laboratory lines that were originally isolated mostly from peninsular Malaysia. Using microsatellite scoring obtained from genome sequences and combined with genotyping of infections from humans and macaques in the current study, we confirmed that the cluster 3 subpopulation is widespread in peninsular Malaysia. Furthermore, it is divergent from clusters 1 and 2, which account for all infections in Malaysian Borneo and apparently a minority of wild macaque infections in peninsular Malaysia. With smaller numbers of samples, recent studies on sequence diversity in genes encoding the normocyte binding protein (Pknbpxa) (10) and the Duffy binding protein (PkDBP) (30), as well as the 18S rRNA gene and the mitochondrial Cox1 gene, have suggested that parasites in peninsular Malaysia had probably diverged from those in Malaysian Borneo.
It is likely that allopatric divergence occurred as a result of the ocean barrier between Borneo and mainland Southeast Asia, established at the end of the last ice age ≈13,000 years ago, which prevents the movement of wild macaque reservoir hosts (31). However, one of the old laboratory lines that was recently sequenced is labeled as having originally been isolated from a long-tailed macaque in “Philippines,” and this sequence is clearly assigned to cluster 3 along with the parasites from peninsular Malaysia (16), although the islands of the Philippines have never been connected to peninsular Malaysia or any other part of mainland Southeast Asia (32). Unless there was a historical mislabeling or previous mixup of parasite material, this finding suggests that wider sampling of P. knowlesi in wild macaques will give a more complete understanding of divergence within this zoonotic parasite species (31,33–35). Similarly, the observation that a minority of P. knowlesi parasites in long-tailed macaques from peninsular Malaysia are assigned to cluster 2, which has otherwise been seen only in samples from Malaysian Borneo, indicates that additional sampling of macaques from different areas may uncover more features of the parasite population structure.
The sympatric differentiation between cluster 1 and cluster 2 parasites in Malaysian Borneo supports the idea that parasite subpopulations are transmitted independently in long-tailed and pig-tailed macaque populations (36,37). Although pig-tailed macaques occur mostly in forested areas, long-tailed macaques have a broader habitat range in both forested and nonforested areas (38). Because of the absence of parasite samples from pig-tailed macaques in peninsular Malaysia, it is unknown whether there is divergence in P. knowlesi between the different macaque host species in this region.
Analysis of genome sequences to derive the frequency distribution of single-nucleotide polymorphism alleles indicates that the cluster 1 subpopulation of P. knowlesi has undergone long-term population growth (16). It is unknown whether parasites of cluster 2 and cluster 3 subpopulations have a similar demographic history, but genome sequencing of more samples within these subpopulations should be able to address this in the future.
The observation that most infections in all macaque populations are polyclonal, whereas most human cases contain single parasite genotypes, probably reflects a higher intensity of transmission among macaques than from macaques to humans (9). It is not yet known whether there are any substantial differences in the clinical course of infections caused by the 3 major subpopulations of P. knowlesi; this question should be investigated in a manner that accounts for any confounding variables between different study sites. In any case, recognition of these divergent subpopulations provides a more accurate basis on which to understand and potentially control the transmission of this zoonosis. Furthermore, obtaining whole-genome sequence data from more clinical samples belonging to each of the 3 major types should enable a more thorough investigation of the genomic divergence, and identify loci at which there are signals of recent adaptation that may relate to differences in virulence or transmission.
Mr. Divis is a lecturer at the Malaria Research Centre in Universiti Malaysia Sarawak, Malaysia, and a PhD student at the London School of Hygiene and Tropical Medicine, United Kingdom. His research interests include molecular epidemiology, population genetics, and genomics of Plasmodium knowlesi.
Acknowledgments
We thank colleagues in Malaria Research Centre, Universiti Malaysia Sarawak, and the London School of Hygiene and Tropical Medicine for laboratory assistance and the Director General of Health in Malaysia for permission to publish this article. We also thank Sarawak Forestry Department, Sarawak Biodiversity Centre, and Department of Wildlife and National Parks Peninsular Malaysia for the permission to collect samples from macaques. We are also grateful to Clemens Kocken for providing P. knowlesi DNA control samples.
This study is supported by postgraduate scholarship from the Ministry of Higher Education in Malaysia and grants from UNIMAS (grant nos. 01/(TD03)/1003/2012(01) and F05/SpTDG/1447/2016/4), and from the European Research Council (AdG-2011-294428).
References
- Singh B, Daneshvar C. Human infections and detection of Plasmodium knowlesi. Clin Microbiol Rev. 2013;26:165–84. DOIPubMedGoogle Scholar
- Setiadi W, Sudoyo H, Trimarsanto H, Sihite BA, Saragih RJ, Juliawaty R, et al. A zoonotic human infection with simian malaria, Plasmodium knowlesi, in Central Kalimantan, Indonesia. Malar J. 2016;15:218. DOIPubMedGoogle Scholar
- Yusof R, Lau YL, Mahmud R, Fong MY, Jelip J, Ngian HU, et al. High proportion of knowlesi malaria in recent malaria cases in Malaysia. Malar J. 2014;13:168. DOIPubMedGoogle Scholar
- William T, Rahman HA, Jelip J, Ibrahim MY, Menon J, Grigg MJ, et al. Increasing incidence of Plasmodium knowlesi malaria following control of P. falciparum and P. vivax Malaria in Sabah, Malaysia. PLoS Negl Trop Dis. 2013;7:e2026. DOIPubMedGoogle Scholar
- Fornace KM, Nuin NA, Betson M, Grigg MJ, William T, Anstey NM, et al. Asymptomatic and submicroscopic carriage of Plasmodium knowlesi malaria in household and community members of clinical cases in Sabah, Malaysia. J Infect Dis. 2016;213:784–7. DOIPubMedGoogle Scholar
- Vythilingam I, Wong ML, Wan-Yussof WS. Current status of Plasmodium knowlesi vectors: a public health concern? Parasitology. 2016;1–9. DOIPubMedGoogle Scholar
- Lee KS, Divis PC, Zakaria SK, Matusop A, Julin RA, Conway DJ, et al. Plasmodium knowlesi: reservoir hosts and tracking the emergence in humans and macaques. PLoS Pathog. 2011;7:e1002015. DOIPubMedGoogle Scholar
- Vythilingam I, Noorazian YM, Huat TC, Jiram AI, Yusri YM, Azahari AH, et al. Plasmodium knowlesi in humans, macaques and mosquitoes in peninsular Malaysia. Parasit Vectors. 2008;1:26. DOIPubMedGoogle Scholar
- Divis PC, Singh B, Anderios F, Hisam S, Matusop A, Kocken CH, et al. Admixture in humans of two divergent Plasmodium knowlesi populations associated with different macaque host species. PLoS Pathog. 2015;11:e1004888. DOIPubMedGoogle Scholar
- Ahmed MA, Fong MY, Lau YL, Yusof R. Clustering and genetic differentiation of the normocyte binding protein (nbpxa) of Plasmodium knowlesi clinical isolates from Peninsular Malaysia and Malaysia Borneo. Malar J. 2016;15:241. DOIPubMedGoogle Scholar
- Ahmed AM, Pinheiro MM, Divis PC, Siner A, Zainudin R, Wong IT, et al. Disease progression in Plasmodium knowlesi malaria is linked to variation in invasion gene family members. PLoS Negl Trop Dis. 2014;8:e3086. DOIPubMedGoogle Scholar
- Pinheiro MM, Ahmed MA, Millar SB, Sanderson T, Otto TD, Lu WC, et al. Plasmodium knowlesi genome sequences from clinical isolates reveal extensive genomic dimorphism. PLoS One. 2015;10:e0121303. DOIPubMedGoogle Scholar
- Fong MY, Lau YL, Chang PY, Anthony CN. Genetic diversity, haplotypes and allele groups of Duffy binding protein (PkDBPαII) of Plasmodium knowlesi clinical isolates from Peninsular Malaysia. Parasit Vectors. 2014;7:161. DOIPubMedGoogle Scholar
- Fong MY, Rashdi SA, Yusof R, Lau YL. Distinct genetic difference between the Duffy binding protein (PkDBPαII) of Plasmodium knowlesi clinical isolates from North Borneo and Peninsular Malaysia. Malar J. 2015;14:91. DOIPubMedGoogle Scholar
- Yusof R, Ahmed MA, Jelip J, Ngian HU, Mustakim S, Hussin HM, et al. Phylogeographic evidence for 2 genetically distinct zoonotic Plasmodium knowlesi parasites, Malaysia. Emerg Infect Dis. 2016;22:1371–80. DOIPubMedGoogle Scholar
- Assefa S, Lim C, Preston MD, Duffy CW, Nair MB, Adroub SA, et al. Population genomic structure and adaptation in the zoonotic malaria parasite Plasmodium knowlesi. Proc Natl Acad Sci U S A. 2015;112:13027–32. DOIPubMedGoogle Scholar
- Kocken CH, Ozwara H, van der Wel A, Beetsma AL, Mwenda JM, Thomas AW. Plasmodium knowlesi provides a rapid in vitro and in vivo transfection system that enables double-crossover gene knockout studies. Infect Immun. 2002;70:655–60. DOIPubMedGoogle Scholar
- Li H, Handsaker B, Wysoker A, Fennell T, Ruan J, Homer N, et al.; 1000 Genome Project Data Processing Subgroup. The Sequence Alignment/Map format and SAMtools. Bioinformatics. 2009;25:2078–9. DOIPubMedGoogle Scholar
- Danecek P, Auton A, Abecasis G, Albers CA, Banks E, DePristo MA, et al.; 1000 Genomes Project Analysis Group. The variant call format and VCFtools. Bioinformatics. 2011;27:2156–8. DOIPubMedGoogle Scholar
- Rutherford K, Parkhill J, Crook J, Horsnell T, Rice P, Rajandream MA, et al. Artemis: sequence visualization and annotation. Bioinformatics. 2000;16:944–5. DOIPubMedGoogle Scholar
- Pritchard JK, Stephens M, Donnelly P. Inference of population structure using multilocus genotype data. Genetics. 2000;155:945–59.PubMedGoogle Scholar
- Hubisz MJ, Falush D, Stephens M, Pritchard JK. Inferring weak population structure with the assistance of sample group information. Mol Ecol Resour. 2009;9:1322–32. DOIPubMedGoogle Scholar
- Earl DA, vonHoldt BM. STRUCTURE HARVESTER: a website and program for visualizing STRUCTURE output and implementing the Evanno method. Conserv Genet Resour. 2012;4:359–61. DOIGoogle Scholar
- Evanno G, Regnaut S, Goudet J. Detecting the number of clusters of individuals using the software STRUCTURE: a simulation study. Mol Ecol. 2005;14:2611–20. DOIPubMedGoogle Scholar
- Jakobsson M, Rosenberg NA. CLUMPP: a cluster matching and permutation program for dealing with label switching and multimodality in analysis of population structure. Bioinformatics. 2007;23:1801–6. DOIPubMedGoogle Scholar
- Peakall R, Smouse PE. GENALEX 6: genetic analysis in Excel. Population genetic software for teaching and research. Mol Ecol Resour. 2006;6:288–95. DOIGoogle Scholar
- Jombart T, Devillard S, Balloux F. Discriminant analysis of principal components: a new method for the analysis of genetically structured populations. BMC Genet. 2010;11:94. DOIPubMedGoogle Scholar
- Goudet J. FSTAT (Version 1.2): A computer program to calculate F-statistics. J Hered. 1995;86:485–6. DOIGoogle Scholar
- Haubold B, Hudson RR. LIAN 3.0: detecting linkage disequilibrium in multilocus data. Linkage Analysis. Bioinformatics. 2000;16:847–8. DOIPubMedGoogle Scholar
- Putaporntip C, Kuamsab N, Jongwutiwes S. Sequence diversity and positive selection at the Duffy-binding protein genes of Plasmodium knowlesi and P. cynomolgi: Analysis of the complete coding sequences of Thai isolates. Infect Genet Evol. 2016;44:367–75. DOIPubMedGoogle Scholar
- Liedigk R, Kolleck J, Böker KO, Meijaard E, Md-Zain BM, Abdul-Latiff MA, et al. Mitogenomic phylogeny of the common long-tailed macaque (Macaca fascicularis fascicularis). BMC Genomics. 2015;16:222. DOIPubMedGoogle Scholar
- Voris HK. Maps of Pleistocene sea levels in Southeast Asia: shorelines, river systems and time durations. J Biogeogr. 2000;27:1153––67. DOIGoogle Scholar
- Esselstyn JA, Widmann P, Heaney LR. The mammals of Palawan Island, Philippines. Proc Biol Soc Wash. 2004;117:271–302.
- Meijaard E. Mammals of south-east Asian islands and their Late Pleistocene environments. J Biogeogr. 2003;30:1245–57. DOIGoogle Scholar
- Smith DG, Ng J, George D, Trask JS, Houghton P, Singh B, et al. A genetic comparison of two alleged subspecies of Philippine cynomolgus macaques. Am J Phys Anthropol. 2014;155:136–48. DOIPubMedGoogle Scholar
- Muehlenbein MP, Pacheco MA, Taylor JE, Prall SP, Ambu L, Nathan S, et al. Accelerated diversification of nonhuman primate malarias in Southeast Asia: adaptive radiation or geographic speciation? Mol Biol Evol. 2015;32:422–39. DOIPubMedGoogle Scholar
- Ziegler T, Abegg C, Meijaard E, Perwitasari-Farajallah D, Walter L, Hodges JK, et al. Molecular phylogeny and evolutionary history of Southeast Asian macaques forming the M. silenus group. Mol Phylogenet Evol. 2007;42:807–16. DOIPubMedGoogle Scholar
- Moyes CL, Shearer FM, Huang Z, Wiebe A, Gibson HS, Nijman V, et al. Predicting the geographical distributions of the macaque hosts and mosquito vectors of Plasmodium knowlesi malaria in forested and non-forested areas. Parasit Vectors. 2016;9:242. DOIPubMedGoogle Scholar
Figures
Table
Cite This ArticleTable of Contents – Volume 23, Number 4—April 2017
| EID Search Options |
|---|
|
|
|
|
|
|


Please use the form below to submit correspondence to the authors or contact them at the following address:
Paul C.S. Divis, Malaria Research Centre, Universiti Malaysia Sarawak, 94300 Kota Samarahan, Sarawak, Malaysia
Top