Volume 26, Number 8—August 2020
Research
Population Genomic Structure and Recent Evolution of Plasmodium knowlesi, Peninsular Malaysia
Cite This Article
Citation for Media
Abstract
Most malaria in Malaysia is caused by Plasmodium knowlesi parasites through zoonotic infection from macaque reservoir hosts. We obtained genome sequences from 28 clinical infections in Peninsular Malaysia to clarify the emerging parasite population structure and test for evidence of recent adaptation. The parasites all belonged to a major genetic population of P. knowlesi (cluster 3) with high genomewide divergence from populations occurring in Borneo (clusters 1 and 2). We also observed unexpected local genetic subdivision; most parasites belonged to 2 subpopulations sharing a high level of diversity except at particular genomic regions, the largest being a region of chromosome 12, which showed evidence of recent directional selection. Surprisingly, we observed a third subpopulation comprising P. knowlesi infections that were almost identical to each other throughout much of the genome, indicating separately maintained transmission and recent genetic isolation. Each subpopulation could evolve and present a broader health challenge in Asia.
All endemic human malaria parasite species originated as zoonotic crossover infections from nonhuman primates (1–3) and now cause approximately half a million human deaths annually (4). Until recently, zoonotic malaria was considered to be very rare, but original findings in Malaysia (5,6) and subsequent surveys elsewhere have revealed that many human malaria cases in Southeast Asia are caused by the macaque parasite Plasmodium knowlesi (7). This parasite species now causes almost all malaria in Malaysia (4) and is responsible for clinical cases throughout Southeast Asia, where the distributions of macaque reservoir hosts and mosquito vectors overlap with human populations (8). As several countries in Southeast Asia are working toward eliminating malaria, P. knowlesi represents a special public health challenge. Because of the presence of wild reservoir hosts, elimination of P. knowlesi is unlikely, and the problem will deepen if the parasite adapts or environments change to enable more effective transmission between humans (9). Of particular concern, numbers of cases each year are continuing to increase (4), and intensive surveillance in particular areas indicates this increase is not attributable to ascertainment bias (10).
Population genetic studies are essential to determining whether recent parasite adaptation has occurred, which might reflect ongoing evolution that is likely to affect the epidemiology. The P. knowlesi parasite has a ≈25 megabase genome of 14 chromosomes (11,12), haploid in blood stage infections and recombining in a brief diploid stage after male and female parasites mate in the mosquito vector, so informative studies require analysis of loci throughout the genome. Understanding of P. knowlesi population genetics has been gained by microsatellite genotyping (13,14) and whole-genome sequencing (15,16). In Malaysian Borneo, P. knowlesi consists of 2 genetically divergent populations (termed clusters 1 and 2) associated with different reservoir hosts: cluster 1 with long-tailed macaques (Macaca fascicularis) and cluster 2 with pig-tailed macaques (M. nemestrina) (14). In Peninsular Malaysia, on the mainland of Asia, a different genetic subpopulation of P. knowlesi exists. This subpopulation was initially indicated by comparing genome sequences of a few old laboratory isolates from Peninsular Malaysia with genome sequences of recent clinical samples from Borneo (15), and by comparing sequences of 2 genes in clinical samples from both areas (17). Subsequent multilocus microsatellite analysis of recent clinical cases of P. knowlesi infection from Peninsular Malaysia has confirmed that all cases are attributable to the cluster 3 type (13).
Experimental studies have been conducted on only a few strains of P. knowlesi, isolated many years ago and maintained in laboratory monkeys (18). Genome sequencing has revealed these strains to be of the cluster 3 type (13,15), and one of them has been adapted to in vitro culture in human erythrocytes, using 2 independent approaches involving culture with mixtures of macaque and human erythrocytes before growth in human erythrocytes alone (19,20). The short-term adaptability of this single strain is further illustrated by selection for culture in long-tailed macaque erythrocytes, which was associated with the loss of a specific ligand gene needed for invading human erythrocytes (21). These examples from laboratory observations strongly suggest that highly diverse natural parasite populations are likely to adapt to changing conditions.
All of the separately occurring P. knowlesi populations might evolve and emerge to present an even more serious public health challenge than already realized. To determine the population genetic substructure within P. knowlesi locally, we analyzed recent clinical samples from patients with P. knowlesi infection in Peninsular Malaysia by using whole-genome sequencing.
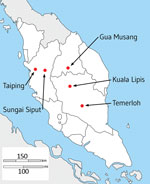
We collected heparinized venous blood samples of up to 10 mL from 56 patients with P. knowlesi malaria at 5 hospitals in Peninsular Malaysia during February 2016–January 2018 (Figure 1; Appendix 1 Table), after obtaining written informed consent from each patient. The study was approved by the Medical Research and Ethics Committee of the Malaysia Ministry of Health and by the Ethics Committee of the London School of Hygiene and Tropical Medicine.
We depleted leukocytes by passing each blood sample through a CF11 cellulose column to increase the proportion of parasite compared with host DNA. We extracted genomic DNA by using QIAamp DNA Mini kits (QIAGEN, https://www.qiagen.com) and confirmed that all contained only P. knowlesi by using nested PCR assays, testing for all locally known malaria parasite species (22). We lyophilized genomic DNA before transport to the United Kingdom, then dissolved the DNA in 30 μL of nuclease-free water and quantified it on a spectrophotometer by using the Quant-iT PicoGreen dsDNA Assay Kit (Thermo Fisher Scientific, https://www.thermofisher.com). We processed samples containing >300 ng of DNA for sequencing. We performed paired-end short-read genome sequencing by using Illumina MiSeq version 3 kits on the MiSeq platform (Illumina, https://www.illumina.com) with a read length of 300 bp, and aligned reads to the P. knowlesi PKNH 2.0 reference genome sequence (Appendix 1). After assembly and quality filtering, data from 28 clinical case samples were available for downstream analysis, representing samples from 5 hospitals in Peninsular Malaysia (Figure 1). For comparison with samples from elsewhere, we retrieved Illumina short reads from previous studies (15,16,23) and assembled them by using the identical pipeline as we had for the newly sequenced samples.
We masked from analysis parts of the genome to which short reads are difficult to uniquely map, including the subtelomeres and the multicopy kir and SICAvar gene families (Appendix 1). We called single-nucleotide polymorphisms (SNPs) by using a full repertoire of P. knowlesi genome sequences, including the 28 new sequenced infection samples from Peninsular Malaysia with high read coverage obtained in this study, as well as 74 previous samples from Malaysian Borneo (40 from cluster 1 and 34 from cluster 2) and 5 laboratory isolates (107 in total) (15,16,23). The procedures and parameters we used are comparable to those used for other original studies of P. knowlesi conducted previously (15,16) and to those used in population studies on endemic human malaria parasites, such as P. falciparum, that have much less diversity than P. knowlesi (15,24).
We used the packages adegenet (https://github.com/thibautjombart/adegenet/wiki) (25) and pegas (https://cran.r-project.org/web/packages/pegas/index.html) (26) in the R statistical framework to conduct principal component analysis and generate neighbor-joining trees by using an SNP-based pairwise genetic distance matrix. For population structure analysis, we used the package PopGenome (27) to calculate nucleotide diversity, within-population Tajima’s D indices, and between-population fixation indices (FST). For sliding-window analysis genomewide, we calculated nucleotide diversity in nonoverlapping 50-kb windows. To scan for genes that might be under balancing selection, we calculated Tajima’s D on a gene-by-gene basis and excluded genes with <3 SNPs from analysis. We calculated FST values between each of the major parasite clusters for all individual SNPs across the genome with a minor allele frequency of >10%, and we calculated mean values in all nonoverlapping sliding windows of 500 consecutive SNPs across the genome.
We performed a scan for loci under recent positive selection by identifying SNPs with an allele associated with extended haplotype homozygosity, using the R package rehh (28) and applying the default setting assumption that common alleles are more likely to be ancestral. We calculated the standardized integrated haplotype score (|iHS|) for biallelic SNPs with no missing calls and with a minor allele frequency of >10%. We set SNPs with |iHS| values in the top 0.01% as core SNPs, around which we identified putative windows of selection by using the extended haplotype homozygosity (EHH) score, plotted until the EHH signal declined to <0.05 on each side. We merged overlapping windows of EHH containing some of the same SNPs to produce a contiguous overall putative selection window for that region, and we considered any gaps of >20 kb between SNPs with elevated |iHS| values to break a putative window of selection.
Genomic Diversity of Different P. knowlesi Subpopulations
We successfully obtained paired-end short read Illumina genome sequences with high-read depth mapping to the P. knowlesi reference sequence from 28 P. knowlesi clinical case samples (Appendix 1) gathered from hospitals at 5 locations in Peninsular Malaysia (Figure 1). Analysis of these new infection samples together with previous sequences from infections in Malaysian Borneo enabled 994,761 SNPs to be initially called, of which 40,934 SNPs were removed because they were in genomic regions with generally unreliable short-read mapping (kir and SICAvar genes and subtelomeres), resulting in a total of 953,827 SNPs throughout the rest of the genome. After filtering out SNPs that had missing data in >10% of individual infection samples, we included 474,109 high-quality SNPs in subsequent analysis (Appendix 2 Datasheet 1).
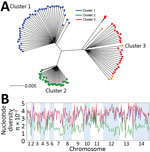
We generated a neighbor-joining tree by using pairwise genetic distances among individual P. knowlesi infection samples, which considered most nucleotide calls for all SNPs within each infection sample (Figure 2, panel A). The tree showed that all of the 28 samples from Peninsular Malaysia belonged to a genetic population (cluster 3) divergent from those in Malaysian Borneo (clusters 1 and 2). These new clinical samples from Peninsular Malaysia clustered with the old laboratory isolates (mostly from Peninsular Malaysia) that were sequenced previously and had initially indicated the existence of a third major genetic population within this species (15). The overall genomewide nucleotide diversity (π) among the new cluster 3 samples was 4.13 × 10−3 (the allele frequency spectrum is shown in Appendix 1 Figure 1), broadly similar to that for cluster 1 and higher than for cluster 2 (Figure 2, panel B). Differences between clusters 1 and 2 have been examined in detail in original analysis of parasites from Malaysian Borneo (16), and higher population genomic diversity within P. knowlesi has been noted in comparison with endemic malaria parasite species P. vivax and P. falciparum (15).
Low Levels of Diversity within Individual Clinical Infections
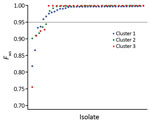
Although all of the P. knowlesi clinical isolates were genotypically distinct from each other as indicated by considerable pairwise differences, most of them contained minimal within-infection sequence diversity, as indicated by the high values of the genomewide within-isolate fixation index FWS (Figure 3). Of the 28 cluster 3 clinical isolates from Peninsular Malaysia in our study, only 4 were clearly mixed (with FWS values <0.95). This low proportion of mixed genotype infections was similar to that observed for the different P. knowlesi subpopulations (clusters 1 and 2) in Malaysian Borneo (Figure 3).
Population Genetic Substructure of P. knowlesi in Peninsular Malaysia
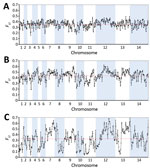
Analysis of SNP allele frequencies genomewide confirmed that the P. knowlesi cluster 3 population in Peninsular Malaysia is highly divergent from each of the separate clusters 1 and 2 in Malaysian Borneo (Figure 4). Comparison of cluster 3 with cluster 1 reveals a genomewide mean FST values of 0.32 (with 3,713 SNPs being at complete fixation), whereas comparison of cluster 3 with cluster 2 reveals a genomewide mean FST value of 0.42, (with 6,738 SNPs being at complete fixation) (Figure 4; Appendix 1 Figure 2). We observed a high consistency across the genome in the level of divergence when comparing cluster 3 with cluster 1 (Figure 4, panel A), but more variation across the genome was apparent in the comparison between cluster 3 and cluster 2 (Figure 4, panel B). This finding is attributable to a previously described mosaic pattern of diversity across the genome of cluster 2 (16), which contributes to greater genomewide heterogeneity in divergence between clusters 1 and 2 in Malaysian Borneo (Figure 4, panel C) than between either of these and cluster 3 in Peninsular Malaysia.
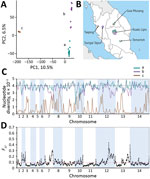
The distance matrix-based neighbor-joining tree indicated internal branching of the P. knowlesi cluster 3 clinical samples from Peninsular Malaysia into 3 different subclusters (Figure 2, panel A). To examine this branching further, we focused the principal component analysis on the clinical samples from Peninsular Malaysia alone, which showed that they formed 3 groups (Figure 5, panel A). The smallest group contained 3 of the samples (GMK03, TPK03, and KLK12) separated from the rest along principal component 1 (which explained 10.5% of overall variation), whereas the 2 other groups were less tightly separated along principal component 2 (which explained 6.5% of overall variation) (Figure 5, panel A). These 3 groups existing within P. knowlesi cluster 3 in Peninsular Malaysia are considered as subpopulations, designated in this article as subclusters A (15 of the infections), B (10 infections), and C (3 infections), which are also apparent in neighbor-joining analysis of the cluster 3 samples alone (Appendix 1 Figure 3). These different parasite genetic subclusters are not separated geographically within Peninsular Malaysia, each being detected from multiple sites (Figure 5, panel B), and the hospital with the largest sample size (in Kuala Lipis) had infections of all 3 subclusters. Moreover, infections of the different subcluster types were not temporally aggregated (Appendix 1 Table).
The most divergent of these types (subcluster C) consisted of infections that were highly related to each other, virtually identical in many parts of the genome (Figure 5, panel C). This finding is remarkable because each of these cases were sampled from different states within Peninsular Malaysia (Figure 4, panel B). Although subclusters A and B had similar levels of nucleotide diversity to each other, sliding-window analysis indicated a few genomic regions in which subcluster B has lower diversity than subcluster A (e.g., in a region covering half of chromosome 12) (Figure 5, panel C). Genomewide scan of differentiation between subclusters A and B by sliding-window FST analysis showed peaks of high differentiation against a background of low differentiation in most of the genome (Figure 5, panel D). The regions that showed differences in levels of diversity are also the most differentiated between the subclusters, most notably the large region of chromosome 12, which has many windows with FST values exceeding 0.2 (Figure 5, panel D) and contains some individual SNPs with fixed differences (Appendix 1 Figure 4).
Identification of Genomic Loci under Recent Selection in Peninsular Malaysia
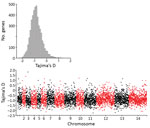
To scan for loci that might be under different selection pressures in P. knowlesi in Peninsular Malaysia, we summarized nucleotide site allele frequency spectra by calculating Tajima’s D index for all 4,742 genes with >3 SNPs. Overall, values were negatively skewed (mean −0.86) (Figure 6, panel A); only 215 genes had values >0, of which only 8 had values >1.0 (Figure 6, panel B). This genomewide pattern is consistent with expectations if long-term population size expansion had occurred. Individual genes with unusually high Tajima’s D values (Figure 6, panel B) might be under balancing selection and might be examined separately (Appendix 2 Datasheet 2). Some genes with high values have orthologs in other malaria parasite species that are likely targets of immunity, including a tryptophan-rich protein (PKNH_1472400, D = 0.98), a 6-cysteine protein (PKNH_1254400, D = 0.61), an exported protein PHIST (PKNH_0808500, D = 0.57), and an MSP7-like protein (PKNH_1265900, D = 1.15). However, we found that some other genes with orthologs considered to be targets of immunity in other malaria parasite populations had negative Tajima’s D values, including the circumsporozoite protein (csp) gene, which had the highest Tajima’s D value genomewide in cluster 1 P. knowlesi in Malaysian Borneo (15), as well as the apical membrane antigen 1 gene (ama1, D = −1.35), the Duffy binding protein α (DBPα, D = −0.89), and the normocyte binding protein gene (NBPXa, D = −0.42). These findings indicate that the mode or strength of selection on orthologous targets is not uniform in all malaria parasite populations, including among different P. knowlesi subpopulations.
We used the standardized integrated haplotype score |iHS| index as a means of scanning for evidence of genomic regions that are likely to have been affected by recent positive directional selection. Analyzing the full population sample of clinical isolates from Peninsular Malaysia, we observed that 11 SNPs had standardized |iHS| values in the top 0.01%, and examination of the ranges of their extended haplotype homozygosity identified 4 distinct genomic windows of extended haplotypes (Figure 7; Appendix 2 Datasheet 3). Two of these (in chromosomes 1 and 9) spanned across genomic SICAvar and kir genes that had been masked from SNP calling and analysis. The other 2 windows of extended haplotype homozygosity did not include SICAvar or kir genes but covered ≈28 kb (11 genes) on chromosome 9 and ≈315 kb (81 genes) on chromosome 12. The large region of elevated |iHS| values on chromosome 12 coincides with the region having the highest genomic divergence between cluster 3 population subclusters A and B (Figure 5, panel D), indicating that recent selection on this region has affected part of the P. knowlesi population and contributed to the local genetic substructure in Peninsular Malaysia.
Genomewide sequence analysis of new clinical isolates has revealed unexpected parasite population structure and evidence of recent selection in P. knowlesi in Peninsular Malaysia. On the basis of previous multilocus microsatellite analysis, we expected that genome sequencing of samples from Peninsular Malaysia would reveal a parasite population distinct from those previously described in Malaysian Borneo. This distinction was indeed clearly shown; all samples from Peninsular Malaysia belonged to a genetic population (cluster 3) that is highly divergent genomewide from both of the clusters 1 and 2 in Malaysian Borneo (mean FST values of 0.32 for cluster 1 and 0.42 for cluster 2). However, the cluster 3 clinical samples from Peninsular Malaysia themselves constituted 3 distinct subpopulations, and the cause of this local population genetic structure needs to be determined. No geographic separation is apparent; each of the three cluster 3 subpopulations was found in overlapping locations, and all were detected from among cases in the hospital that had most samples analyzed. The population structure might reflect >1 local zoonotic transmission cycle or could be a sign of recent selection and emergence of a subpopulation of P. knowlesi transmitted more effectively between humans.
In Malaysian Borneo, long-tailed macaques are reservoir hosts for the cluster 1 population of P. knowlesi, whereas pig-tailed macaques are reservoir hosts for the cluster 2 population (13,14), but whether different reservoir hosts contribute to the parasite population structure we have shown within Peninsular Malaysia is unknown. Microsatellite analysis of P. knowlesi in long-tailed macaques from Peninsular Malaysia has indicated that most of them belong to cluster 3, although some samples from long-tailed macaques had indeterminate cluster assignments (13). Our findings underscore the need to sample and genotype parasites from pig-tailed macaques in Peninsular Malaysia, as well as to analyze more samples from long-tailed macaques, to investigate whether the parasite subclusters have different reservoir host species locally.
Genetic subpopulations of P. knowlesi in Peninsular Malaysia might also be transmitted by different mosquito species. P. knowlesi is transmitted by the Anopheles Leucosphyrus group of mosquitoes, which contains a diverse array of species found throughout Southeast Asia (29), including An. latens, An. cracens, An. introlatus, and An. hackeri, in which P. knowlesi has been detected in Peninsular Malaysia, as well as other species that have been shown to be infected elsewhere (30). Anopheles Leucosphyrus group mosquitoes predominantly inhabit forested areas (31,32), so changes to forest areas and ongoing deforestation will affect human exposure. The potential vector species vary in relative abundance among different sampling sites in Peninsular Malaysia (33–35), but more surveys are required to determine the relative extent to which they transmit P. knowlesi and whether they transmit different populations of the parasite (36).
Genomewide scanning revealed discrete regions of divergence between subclusters A and B of P. knowlesi cluster 3, in particular a large region on chromosome 12. Interestingly, this region had the strongest evidence of recent directional selection, as indicated by the integrated haplotype score analysis. Moreover, this genomic region did not show evidence of recent selection in Malaysian Borneo (15), so the signature is specific to Peninsular Malaysia and indicates selection to be operating locally.
Even more unexpected is the observation of a separate P. knowlesi cluster 3 subpopulation (subcluster C), represented by 3 infections highly related to each other throughout most of the genome. Although less common, clinical cases with this parasite type were identified in different hospitals in 3 different states in Peninsular Malaysia. Population genetic substructure also has been observed in the endemic malaria parasites P. falciparum (37) and P. vivax (38) in Malaysia, although that observation has been interpreted as indicating fragmented populations that are close to being eliminated. Notable substructure of P. falciparum populations also has been observed in Cambodia, probably because of strong selection on locally emerging drug-resistant types in areas where transmission was low (39). Zoonotic P. knowlesi populations are probably substructured for other reasons, as previously observed in Malaysian Borneo, where the 2 divergent parasite genetic populations observed in human cases are associated with different reservoir host species (13,14).
Population genomic analysis of P. knowlesi so far has mainly focused on parasites from Malaysia, where most reported cases of P. knowlesi malaria have been identified. However, cases of P. knowlesi malaria in humans have now been reported from all Southeast Asia countries. Whether other local zoonotic subpopulations exist throughout the region or whether all parasites belong to the major genetic populations observed in Malaysia is unknown. Our findings highlight the importance of monitoring population genetic changes in Malaysia and conducting comparable analysis in other areas where P. knowlesi has only very recently been realized to occur in humans (40,41).
Dr. Hocking completed a PhD in the London Interdisciplinary Doctoral Training Programme (http://lido-dtp.ac.uk). Her thesis focuses on sequencing of genomes and transcriptomes to investigate population structure and biology of malaria parasites.
Acknowledgments
We thank the hospital directors for their cooperation, the medical and nursing staff for obtaining consent and blood samples from malaria patients, and the medical laboratory staff for processing the samples from the Malaysia Ministry of Health hospitals at Kuala Lipis, Gua Musang, Temerloh, Sungai Siput, and Taiping. We thank the Director General of Health in Malaysia for permission to publish this paper. We are grateful to Alfred Amambua-Ngwa for comments on the draft manuscript.
This study was supported by funds from Universiti Malaysia Sarawak (grant no. F05/SpTDG/1447/2016/4), European Research Council (grant no. AdG-2011-294428), and the Biotechnology and Biological Sciences Research Council (London Interdisciplinary Doctoral Training Programme PhD studentship support for S.E.H.).
References
- Liu W, Li Y, Learn GH, Rudicell RS, Robertson JD, Keele BF, et al. Origin of the human malaria parasite Plasmodium falciparum in gorillas. Nature. 2010;467:420–5. DOIPubMedGoogle Scholar
- Loy DE, Plenderleith LJ, Sundararaman SA, Liu W, Gruszczyk J, Chen YJ, et al. Evolutionary history of human Plasmodium vivax revealed by genome-wide analyses of related ape parasites. Proc Natl Acad Sci U S A. 2018;115:E8450–9. DOIPubMedGoogle Scholar
- Rutledge GG, Böhme U, Sanders M, Reid AJ, Cotton JA, Maiga-Ascofare O, et al. Plasmodium malariae and P. ovale genomes provide insights into malaria parasite evolution. Nature. 2017;542:101–4. DOIPubMedGoogle Scholar
- WHO. World malaria report 2018 [cited 2019 Jun 13]. https://www.who.int/malaria/publications/world-malaria-report-2018
- Cox-Singh J, Davis TM, Lee KS, Shamsul SS, Matusop A, Ratnam S, et al. Plasmodium knowlesi malaria in humans is widely distributed and potentially life threatening. Clin Infect Dis. 2008;46:165–71. DOIPubMedGoogle Scholar
- Singh B, Kim Sung L, Matusop A, Radhakrishnan A, Shamsul SS, Cox-Singh J, et al. A large focus of naturally acquired Plasmodium knowlesi infections in human beings. Lancet. 2004;363:1017–24. DOIPubMedGoogle Scholar
- Shearer FM, Huang Z, Weiss DJ, Wiebe A, Gibson HS, Battle KE, et al. Estimating geographical variation in the risk of zoonotic Plasmodium knowlesi infection in countries eliminating malaria. PLoS Negl Trop Dis. 2016;10:
e0004915 . DOIPubMedGoogle Scholar - Singh B, Daneshvar C. Human infections and detection of Plasmodium knowlesi. Clin Microbiol Rev. 2013;26:165–84. DOIPubMedGoogle Scholar
- William T, Jelip J, Menon J, Anderios F, Mohammad R, Awang Mohammad TA, et al. Changing epidemiology of malaria in Sabah, Malaysia: increasing incidence of Plasmodium knowlesi. Malar J. 2014;13:390. DOIPubMedGoogle Scholar
- Cooper DJ, Rajahram GS, William T, Jelip J, Mohammad R, Benedict J, et al. Plasmodium knowlesi malaria in Sabah, Malaysia, 2015–2017: ongoing increase in incidence despite near-elimination of the human-only Plasmodium species. Clin Infect Dis. 2020;70:361–7.PubMedGoogle Scholar
- Lapp SA, Geraldo JA, Chien JT, Ay F, Pakala SB, Batugedara G, et al. MaHPIC consortium. PacBiol assembly of a Plasmodium knowlesi genome sequence with Hi-C correction and manual annotation of the SICAvar gene family. Parasitology. 2018;145:71–84. DOIPubMedGoogle Scholar
- Pain A, Böhme U, Berry AE, Mungall K, Finn RD, Jackson AP, et al. The genome of the simian and human malaria parasite Plasmodium knowlesi. Nature. 2008;455:799–803. DOIPubMedGoogle Scholar
- Divis PC, Lin LC, Rovie-Ryan JJ, Kadir KA, Anderios F, Hisam S, et al. Three divergent subpopulations of the malaria parasite Plasmodium knowlesi. Emerg Infect Dis. 2017;23:616–24. DOIPubMedGoogle Scholar
- Divis PC, Singh B, Anderios F, Hisam S, Matusop A, Kocken CH, et al. Admixture in humans of two divergent Plasmodium knowlesi populations associated with different macaque host species. PLoS Pathog. 2015;11:
e1004888 . DOIPubMedGoogle Scholar - Assefa S, Lim C, Preston MD, Duffy CW, Nair MB, Adroub SA, et al. Population genomic structure and adaptation in the zoonotic malaria parasite Plasmodium knowlesi. Proc Natl Acad Sci U S A. 2015;112:13027–32. DOIPubMedGoogle Scholar
- Divis PCS, Duffy CW, Kadir KA, Singh B, Conway DJ. Genome-wide mosaicism in divergence between zoonotic malaria parasite subpopulations with separate sympatric transmission cycles. Mol Ecol. 2018;27:860–70. DOIPubMedGoogle Scholar
- Yusof R, Ahmed MA, Jelip J, Ngian HU, Mustakim S, Hussin HM, et al. Phylogeographic evidence for 2 genetically distinct zoonotic Plasmodium knowlesi parasites, Malaysia. Emerg Infect Dis. 2016;22:1371–80. DOIPubMedGoogle Scholar
- Coatney GR, Collins WE, McWilson W, Contacos PG. The primate malarias. Atlanta: US Department of Health and Human Services; 1971.
- Moon RW, Hall J, Rangkuti F, Ho YS, Almond N, Mitchell GH, et al. Adaptation of the genetically tractable malaria pathogen Plasmodium knowlesi to continuous culture in human erythrocytes. Proc Natl Acad Sci U S A. 2013;110:531–6. DOIPubMedGoogle Scholar
- Lim C, Hansen E, DeSimone TM, Moreno Y, Junker K, Bei A, et al. Expansion of host cellular niche can drive adaptation of a zoonotic malaria parasite to humans. Nat Commun. 2013;4:1638. DOIPubMedGoogle Scholar
- Moon RW, Sharaf H, Hastings CH, Ho YS, Nair MB, Rchiad Z, et al. Normocyte-binding protein required for human erythrocyte invasion by the zoonotic malaria parasite Plasmodium knowlesi. Proc Natl Acad Sci U S A. 2016;113:7231–6. DOIPubMedGoogle Scholar
- Lee KS, Divis PC, Zakaria SK, Matusop A, Julin RA, Conway DJ, et al. Plasmodium knowlesi: reservoir hosts and tracking the emergence in humans and macaques. PLoS Pathog. 2011;7:
e1002015 . DOIPubMedGoogle Scholar - Pinheiro MM, Ahmed MA, Millar SB, Sanderson T, Otto TD, Lu WC, et al. Plasmodium knowlesi genome sequences from clinical isolates reveal extensive genomic dimorphism. PLoS One. 2015;10:
e0121303 . DOIPubMedGoogle Scholar - Duffy CW, Amambua-Ngwa A, Ahouidi AD, Diakite M, Awandare GA, Ba H, et al. Multi-population genomic analysis of malaria parasites indicates local selection and differentiation at the gdv1 locus regulating sexual development. Sci Rep. 2018;8:15763. DOIPubMedGoogle Scholar
- Jombart T. adegenet: a R package for the multivariate analysis of genetic markers. Bioinformatics. 2008;24:1403–5. DOIPubMedGoogle Scholar
- Paradis E. pegas: an R package for population genetics with an integrated-modular approach. Bioinformatics. 2010;26:419–20. DOIPubMedGoogle Scholar
- Pfeifer B, Wittelsbürger U, Ramos-Onsins SE, Lercher MJ. PopGenome: an efficient Swiss army knife for population genomic analyses in R. Mol Biol Evol. 2014;31:1929–36. DOIPubMedGoogle Scholar
- Gautier M, Vitalis R. rehh: an R package to detect footprints of selection in genome-wide SNP data from haplotype structure. Bioinformatics. 2012;28:1176–7. DOIPubMedGoogle Scholar
- Sallum MA, Peyton EL, Wilkerson RC. Six new species of the Anopheles leucosphyrus group, reinterpretation of An. elegans and vector implications. Med Vet Entomol. 2005;19:158–99. DOIPubMedGoogle Scholar
- Vythilingam I, Wong ML, Wan-Yussof WS. Current status of Plasmodium knowlesi vectors: a public health concern? Parasitology. 2018;145:32–40. DOIPubMedGoogle Scholar
- Moyes CL, Shearer FM, Huang Z, Wiebe A, Gibson HS, Nijman V, et al. Predicting the geographical distributions of the macaque hosts and mosquito vectors of Plasmodium knowlesi malaria in forested and non-forested areas. Parasit Vectors. 2016;9:242. DOIPubMedGoogle Scholar
- Sinka ME, Bangs MJ, Manguin S, Chareonviriyaphap T, Patil AP, Temperley WH, et al. The dominant Anopheles vectors of human malaria in the Asia-Pacific region: occurrence data, distribution maps and bionomic précis. Parasit Vectors. 2011;4:89. DOIPubMedGoogle Scholar
- Jiram AI, Vythilingam I, NoorAzian YM, Yusof YM, Azahari AH, Fong MY. Entomologic investigation of Plasmodium knowlesi vectors in Kuala Lipis, Pahang, Malaysia. Malar J. 2012;11:213. DOIPubMedGoogle Scholar
- Vythilingam I, Lim YA, Venugopalan B, Ngui R, Leong CS, Wong ML, et al. Plasmodium knowlesi malaria an emerging public health problem in Hulu Selangor, Selangor, Malaysia (2009-2013): epidemiologic and entomologic analysis. Parasit Vectors. 2014;7:436. DOIPubMedGoogle Scholar
- Vythilingam I, Noorazian YM, Huat TC, Jiram AI, Yusri YM, Azahari AH, et al. Plasmodium knowlesi in humans, macaques and mosquitoes in peninsular Malaysia. Parasit Vectors. 2008;1:26. DOIPubMedGoogle Scholar
- Wong ML, Ahmed MA, Sulaiman WYW, Manin BO, Leong CS, Quan FS, et al. Genetic diversity of zoonotic malaria parasites from mosquito vector and vertebrate hosts. Infect Genet Evol. 2019;73:26–32. DOIPubMedGoogle Scholar
- Anthony TG, Conway DJ, Cox-Singh J, Matusop A, Ratnam S, Shamsul S, et al. Fragmented population structure of plasmodium falciparum in a region of declining endemicity. J Infect Dis. 2005;191:1558–64. DOIPubMedGoogle Scholar
- Auburn S, Benavente ED, Miotto O, Pearson RD, Amato R, Grigg MJ, et al. Genomic analysis of a pre-elimination Malaysian Plasmodium vivax population reveals selective pressures and changing transmission dynamics. Nat Commun. 2018;9:2585. DOIPubMedGoogle Scholar
- Miotto O, Almagro-Garcia J, Manske M, Macinnis B, Campino S, Rockett KA, et al. Multiple populations of artemisinin-resistant Plasmodium falciparum in Cambodia. Nat Genet. 2013;45:648–55. DOIPubMedGoogle Scholar
- Herdiana H, Irnawati I, Coutrier FN, Munthe A, Mardiati M, Yuniarti T, et al. Two clusters of Plasmodium knowlesi cases in a malaria elimination area, Sabang Municipality, Aceh, Indonesia. Malar J. 2018;17:186. DOIPubMedGoogle Scholar
- Imwong M, Madmanee W, Suwannasin K, Kunasol C, Peto TJ, Tripura R, et al. Asymptomatic natural human infections with the simian malaria parasites Plasmodium cynomolgi and Plasmodium knowlesi. J Infect Dis. 2019;219:695–702. DOIPubMedGoogle Scholar
Figures
Cite This ArticleOriginal Publication Date: June 25, 2020
Table of Contents – Volume 26, Number 8—August 2020
| EID Search Options |
|---|
|
|
|
|
|
|

Please use the form below to submit correspondence to the authors or contact them at the following address:
Address for correspondence author: David J. Conway, Department of Infection Biology, Faculty of Infectious and Tropical Diseases, London School of Hygiene and Tropical Medicine, Keppel St, London WC1E 7HT, UK
Top