Volume 32, Number 3—March 2026
Dispatch
Extraintestinal Entamoeba moshkovskii Infection, Eastern India
Cite This Article
Citation for Media
Abstract
Entamoeba moshkovskii is historically considered nonpathogenic. We report a case of severe extraintestinal infection in a patient from eastern India who had abdominal pain, fever, weight loss, anemia, and a splenic abscess. Molecular analysis confirmed E. moshkovskii as the causative agent. This case highlights this parasite’s potential to cause severe illness.
For more than a century, the Entamoeba histolytica protozoan was considered the sole pathogenic amoeba to cause diarrhea, dysentery, and liver abscess. Microscopy was once the diagnostic standard, but because of the morphologic similarity among Entamoeba species, PCR is now recommended (1,2). Major morphologically identical species indistinguishable from E. histolytica include E. dispar, E. moshkovskii, and E. bangladeshi (3,4). This group of organisms (with E. nuttalli) is called the E. histolytica complex. Within this group, E. moshkovskii was historically regarded as nonpathogenic; however, emerging evidence suggests potential pathogenicity (5–9). Reported prevalence of E. moshkovskii varies widely (≈1%–25%) across epidemiologic settings (5–10).
In Bangladesh, one study reported a 21.1% infection rate in children, whereas another reported a 2.95% prevalence with an association to diarrhea in infants (8,11). Similarly, a 3-year surveillance study in eastern India identified a 3.12% prevalence among diarrheal patients, supporting its classification as an emerging pathogen rather than a nonpathogenic species (5,6). In a murine model of intestinal amebiasis, E. moshkovskii was also found to cause diarrhea, weight loss, and colitis (8).
Emerging epidemiologic evidence suggests E. moshkovskii is a potential enteropathogen. We present a clinically documented case of an extraintestinal infection caused by E. moshkovskii that resulted in severe health complications in a patient from eastern India.
A 36-year-old man from Kolkata, in West Bengal, India, sought care in July 2024 for a 6-day history of intermittent fever, characterized by 1–2 spikes per day and a recorded high temperature of 102°F. The patient reported fever with malaise but no chills or rigors, along with a 5-day history of dull, continuous left upper quadrant abdominal pain that worsened with deep inspiration, sneezing, or when lying in the left lateral decubitus position. A slow-developing heavy feeling in the left upper abdomen, along with nausea, a bad taste in the mouth, and 1 episode of nonbilious vomiting, also occurred. The patient reported weight loss from 53 kg to 47 kg over 4 weeks, despite preserved appetite. In addition, he had a history of occasional nonproductive cough without wheezing, hemoptysis, or shortness of breath. The patient had a left-lobe hepatic abscess diagnosed in March 2023 in Kolkata and was treated empirically with intravenous injection and oral metronidazole. The causative agent was not identified. The bad taste in the patient’s mouth was likely not related to the previous metronidazole treatment, because he had not been on the medication in the previous 3 weeks.
The patient was a cigarette smoker but had abstained from alcohol for 4 years. On examination, his heart rate was 104 beats/min and blood pressure 146/74 mmHg; oxygen saturation (98% room air) and respiratory rate (16 breaths/min) were unremarkable. Physical examination revealed pallor, hepatomegaly (2 cm below right costal margin), splenomegaly (6 cm below left costal margin), and left upper quadrant tenderness. The patient had absent breath sounds over the left intercostal and subaxillary areas but showed no signs of cyanosis, clubbing, or lymphadenopathy. Neurologic examination results were unremarkable. Iron deficiency anemia was noted. Serologic testing revealed negative results for HIV, hepatitis B surface antigen, hepatitis C virus antibody, malarial parasite detection antigen, and Brucella IgM and IgG. The patient also had a nonreactive Rose Bengal plate test and standard tube agglutination test for Brucella. A chest radiograph revealed an elevated left hemidiaphragm and left pleural effusion. An abdominal ultrasound revealed hepatomegaly and a large splenic abscess (18 cm x 14 cm × 17 cm, 2000 mL) with mild subdiaphragmatic fluid. Contrast-enhanced computed tomography of the chest and abdomen confirmed a collapsed left lower lobe and a splenic abscess. The patient was managed with drainage and supportive care.
Pleural fluid analysis with Gram and Ziehl-Neelsen stains showed no microorganisms, and Mycobacterium tuberculosis was not detected. The wet mount showed predominantly macrophages, erythrocytes, epithelial cells, and many protozoal structures resembling E. histolytica with erythrophagia (Figure 1). However, splenic abscess aspirate did not reveal amoebae or amoeba-like structures. The direct smear revealed degenerated blood elements and necrosis, with no granuloma or malignant cells. We conducted additional characterization of the E. histolytica–like organism by using molecular methods.
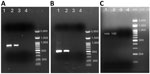
We extracted DNA by using the QIAamp DNA Mini Kit (QIAGEN, https://www.qiagen.com). We performed conventional PCR targeting the 18S rRNA gene on splenic aspirate and pleural fluid by using previously published species-specific primers for E. histolytica and E. moshkovskii (5,12). Both the splenic abscess aspirate and pleural fluid DNA tested negative for E. histolytica but positive for E. moshkovskii (Figure 2, panel A). To confirm the presence of E. moshkovskii, we targeted the chitinase gene for testing. We designed primer sequences to amplify the upstream and downstream regions of the chitinase open reading frame (Appendix Table). Both splenic abscess aspirate and pleural fluid samples again tested positive for E. moshkovskii when targeting the chitinase locus (Figure 2 panel B). We then purified the PCR products by using the Roche PCR Gel Extraction kit (Roche, https://www.roche.com) and sequenced bidirectionally with the BigDye Terminator v3.1 kit (Thermo Fisher Scientific, https://www.thermofisher.com).
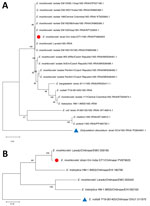
BLAST analysis (https://blast.ncbi.nlm.nih.gov) confirmed the organism as E. moshkovskii, showing 100% identity to the reference Laredo strain (18S rRNA; GenBank accession no. AF149906.1). However, the chitinase gene corresponding to AmoebaDB (https://amoebadb.org/amoeba/app) entry EMO_056190 exhibited 3 mutations at positions 36 T/C, 127 G/A, and *2 T/G. The mutation at position 36 was synonymous, whereas the substitution at position 127 resulted in a nonsynonymous change, replacing glutamic acid with lysine. We constructed phylogenetic trees generated from the obtained 18S rRNA and chitinase gene sequences to confirm species identity and evaluate evolutionary relationships (Figure 3, panels A, B).
Of note, PCR analysis of DNA isolated from the patient’s stool samples by using the 18S rRNA targeted assay detected the presence of E. moshkovskii and not E. histolytica (Figure 2, panel C). In addition, results of ELISA testing for E. histolytica–specific IgG in serum were negative. Taken together, those findings indicate that neither the hepatic abscess nor the splenic abscess was associated with E. histolytica infection.
The patient was initially started on intravenous (IV) meropenem empirically. However, after confirmation of E. moshkovskii infection, we changed the treatment to IV metronidazole (750 mg IV/8 h for 1 d), followed by oral metronidazole (400 mg 3×/d after food for 7 d). The patient was also prescribed ferrous sulfate 200 mg (1 dose by mouth), B-complex tablets, and other medications as needed. Pigtail drainage continued for the first week. The patient recovered after 3 weeks.
E. moshkovskii DNA alongside E. histolytica in 5 of 115 liver abscess cases was previously reported; however, extraintestinal E. moshkovskii infection, particularly in pleural fluid, is extremely rare (13). In the case we report, E. moshkovskii might have reached the pleural cavity through direct extension or rupture of a splenic abscess.
Metronidazole is the primary drug for treating amoebiasis and was effective in this patient, showing empirical activity against E. moshkovskii (14). However, the optimal dosage for this species remains undefined. Drug sensitivity differs among Entamoeba spp.; for instance, E. moshkovskii is resistant to emetine, which is effective against E. histolytica (15). Diagnosis is also challenging. Molecular tests often target only E. histolytica, leaving E. moshkovskii infections unrecognized. In addition, E. moshkovskii produces fewer cysts than E. histolytica, reducing the reliability of microscopic detection and increasing the risk for false negatives (5).
Because of the high regional prevalence of E. moshkovskii, extraintestinal infections caused by this species need attention. Our case underscores the need to better understand transmission and to develop improved diagnostic methods to ensure effective management and prevent drug resistance in E. moshkovskii.
Dr. Sardar conducted his doctoral research at the Indian Council of Medical Research—National Institute for Research in Bacterial Infections and is currently a postdoctoral fellow at The University of Tokyo. His research focuses on the biology and pathogenicity of Entamoeba species.
Acknowledgments
We thank the hospital staff for their support in sample collection and obtaining the patient’s history.
This study was reviewed and approved by the Institutional Human Ethics Committee of the Indian Council of Medical Research—National Institute for Research in Bacterial Infections. Informed consent was obtained from the participant.
Representative sequences obtained in this study were deposited in GenBank under the accession numbers PV882953 for the 18S rRNA and PV879623 for the chitinase locus.
This study was funded by the Indian Council of Medical Research.
Author contributions: study conceptualization, S.K.S. and S.G.; study methodology, S.K.S, A.G., T.H., and B.G.; data curation, S.K.S., A.G., and T.H.; study validation, K.D. and S.G.; writing original draft, S.K.S.; formal statistical analysis, S.K.S.; visualization, S.G.; investigation, S.G.; funding acquisition, S.G.; manuscript review and editing, S.G.; study supervision, S.G.
References
- Tanyuksel M, Petri WA Jr. Laboratory diagnosis of amebiasis. Clin Microbiol Rev. 2003;16:713–29. DOIPubMedGoogle Scholar
- Centers for Disease Control and Prevention. Post-travel diarrhea. In: CDC yellow book: health information for international travel. 2026 [cited 2026 Feb 28]. https://www.cdc.gov/yellow-book/hcp/post-travel-evaluation/post-travel-diarrhea.html
- Ngui R, Angal L, Fakhrurrazi SA, Lian YL, Ling LY, Ibrahim J, et al. Differentiating Entamoeba histolytica, Entamoeba dispar and Entamoeba moshkovskii using nested polymerase chain reaction (PCR) in rural communities in Malaysia. Parasit Vectors. 2012;5:187. DOIPubMedGoogle Scholar
- Royer TL, Gilchrist C, Kabir M, Arju T, Ralston KS, Haque R, et al. Entamoeba bangladeshi nov. sp., Bangladesh. Emerg Infect Dis. 2012;18:1543–5. DOIPubMedGoogle Scholar
- Sardar SK, Ghosal A, Haldar T, Maruf M, Das K, Saito-Nakano Y, et al. Prevalence and molecular characterization of Entamoeba moshkovskii in diarrheal patients from Eastern India. PLoS Negl Trop Dis. 2023;17:
e0011287 . DOIPubMedGoogle Scholar - Sardar SK, Ghosal A, Haldar T, Prasad A, Mal S, Saito-Nakano Y, et al. Genetic characterization of the Entamoeba moshkovskii population based on different potential genetic markers. Parasitology. 2024;151:429–39. DOIPubMedGoogle Scholar
- Sharma D, Rattan D, Kaur U, Dutta U, Sehgal R. First report on PCR-based detection of Entamoeba moshkovskii from a tertiary care center in north India. J Pure Appl Microbiol. 2025;19:100–5. DOIGoogle Scholar
- Shimokawa C, Kabir M, Taniuchi M, Mondal D, Kobayashi S, Ali IK, et al. Entamoeba moshkovskii is associated with diarrhea in infants and causes diarrhea and colitis in mice. J Infect Dis. 2012;206:744–51. DOIPubMedGoogle Scholar
- Kyany’a C, Eyase F, Odundo E, Kipkirui E, Kipkemoi N, Kirera R, et al. First report of Entamoeba moshkovskii in human stool samples from symptomatic and asymptomatic participants in Kenya. Trop Dis Travel Med Vaccines. 2019;5:23. DOIPubMedGoogle Scholar
- López MC, León CM, Fonseca J, Reyes P, Moncada L, Olivera MJ, et al. Molecular epidemiology of Entamoeba: first description of Entamoeba moshkovskii in a rural area from central Colombia. PLoS One. 2015;10:
e0140302 . DOIPubMedGoogle Scholar - Ali IK, Hossain MB, Roy S, Ayeh-Kumi PF, Petri WA Jr, Haque R, et al. Entamoeba moshkovskii infections in children, Bangladesh. Emerg Infect Dis. 2003;9:580–4. DOIPubMedGoogle Scholar
- Mukherjee AK, Chowdhury P, Bhattacharya MK, Ghosh M, Rajendran K, Ganguly S. Hospital-based surveillance of enteric parasites in Kolkata. BMC Res Notes. 2009;2:110. DOIPubMedGoogle Scholar
- Kumar M, Nath G, Parija SC. Detection of Entamoeba dispar and Entamoeba moshkovskii DNA in liver abscess pus: newer perspectives to be considered in diagnosis of amoebiasis. Int J Infect Dis. 2016;45:1364. DOIGoogle Scholar
- Shrivastav MT, Malik Z, Somlata . Somlata. Revisiting drug development against the neglected tropical disease, amebiasis. Front Cell Infect Microbiol. 2021;10:
628257 . DOIPubMedGoogle Scholar - Dreyer DA. Growth of a strain of entamoeba histolytica at room temperature. Tex Rep Biol Med. 1961;19:393–6.PubMedGoogle Scholar
Figures
Cite This ArticleOriginal Publication Date: March 10, 2026
1These authors contributed equally to this article.
Table of Contents – Volume 32, Number 3—March 2026
| EID Search Options |
|---|
|
|
|
|
|
|

Please use the form below to submit correspondence to the authors or contact them at the following address:
Sandipan Ganguly, ICMR–National Institute for Research in Bacterial Infections, P-33, C.I.T. Road, Scheme-XM, Beliaghata, Kolkata 700010, India
Top