Volume 20, Number 5—May 2014
Research
Bovine Leukemia Virus DNA in Human Breast Tissue
Cite This Article
Citation for Media
Abstract
Bovine leukemia virus (BLV), a deltaretrovirus, causes B-cell leukemia/lymphoma in cattle and is prevalent in herds globally. A previous finding of antibodies against BLV in humans led us to examine the possibility of human infection with BLV. We focused on breast tissue because, in cattle, BLV DNA and protein have been found to be more abundant in mammary epithelium than in lymphocytes. In human breast tissue specimens, we identified BLV DNA by using nested liquid-phase PCR and DNA sequencing. Variations from the bovine reference sequence were infrequent and limited to base substitutions. In situ PCR and immunohistochemical testing localized BLV to the secretory epithelium of the breast. Our finding of BLV in human tissues indicates a risk for the acquisition and proliferation of this virus in humans. Further research is needed to determine whether BLV may play a direct role in human disease.
Bovine leukosis (B-cell leukemia/lymphoma), first described in 1871 in Lithuania, was believed to be an infectious disease because it spread through herds of cattle. In 1969, a virus isolated from cultured lymphocytes of cattle in an afflicted herd was identified as the agent of bovine leukosis (1). Since then, bovine leukemia virus (BLV) has been extensively investigated. It is a deltaretrovirus, closely related to human T-cell leukemia virus (HTLV) 1 (2), and has typical retroviral genome regions: LTR (long terminal repeat, promoter region); gag (group-specific antigen, capsid region); pol (polymerase, reverse transcription region, which synthesizes a DNA copy of the BLV RNA genome); and env (envelope). However, unlike other oncogenic retroviruses, deltaretroviruses have an additional region, tax (trans-activating region of the X gene), which has regulatory functions and is oncogenic to host cells. tax causes malignant transformation not through integration and insertional mutagenesis, as many retroviruses do, but by inhibition of DNA repair (base excision pathway) and trans-activating disruption of cellular growth control mechanisms (2).
BLV-infected cattle herds are found worldwide. In the United States, ≈38% of beef herds, 84% of all dairy herds, and 100% of large-scale dairy operation herds are infected (3,4). On average, clinical leukosis develops in <5% of these cattle, which are excluded from the market as a result (1), but BLV-infected lymphocytes are also found in the blood and milk of subclinically infected cows (2). Concerns that this virus might infect humans through exposure to food products from subclinically infected animals prompted 10 studies that used what were then (1975–1979) state-of-the-art immunologic methods to test serum samples from a collective total of 1,761 humans, including cancer patients, farm workers, and veterinarians (5). In these studies no antibodies against BLV were detected, prompting Burridge to conclude in his review article, “There is no epidemiological or serological evidence from human studies to indicate that BLV can infect man” (5).
The advent of immunoblotting, ≈100 times more sensitive than techniques of the 1970s (6), enabled the detection of antibodies reactive with recombinant purified BLV p24 capsid protein in serum samples from 39% of 257 self-selected human volunteers (7). This study could not determine whether the antibodies were a response to infection or merely to heat-inactivated BLV consumed in food products. However, injection of sheep with raw milk from BLV-positive cows stimulated antibody production, whereas injection with pasteurized control milk did not (8,9). The finding of antibodies to BLV in humans prompted us to investigate human tissues for evidence of infection with BLV by using liquid-phase PCR (L-PCR), sequencing, in situ PCR, and immunohistochemical (IHC) testing. We focused on breast tissue because, in cattle, BLV DNA and p24 were detected in mammary tissue, whereas only BLV DNA was detected in lymphocytes (10).
Cells and DNA used to test cross-reactivity of BLV primers and anti-BLV p24 monoclonal antibody (mAb) with other viruses are listed with their sources in Tables 1 and 2. Except for fetal lamb kidney (FLK) cells, cell lines were stored in liquid nitrogen until use. Before experiments were performed, the species of origin of all cell lines was authenticated by using the 2-pronged method (cytochrome oxidase housekeeping gene primers) (11).
Coded human samples were acquired from the Cooperative Human Tissue Network, a National Cancer Institute–supported tissue bank. Specimens were selected, without regard to patient age, race, or diagnosis, from archived breast tissues acquired from female patients who underwent breast surgery during 2000–2005 at participating hospitals in 4 catchments areas: Birmingham, Alabama; Pennsylvania; Ohio; and Oakland, California. The Institutional Review Board of the University of California, Berkeley (Berkeley, CA, USA) approved human subject use. Bovine control tissue came from the University of Wisconsin, Madison (Madison, WI, USA), as described (10).
PCR primers (Table 3) were examined for BLV specificity by using BLAST (http://blast.ncbi.nlm.nih.gov/Blast.cgi) to search for similar sequences in the nucleotide collection database, which includes exogenous viruses and human endogenous retroviruses such as HERV-K. The search was optimized for highly similar sequences.
For DNA extraction and quality control, cell lines were rinsed with Dulbecco phosphate-buffered saline (DPBS) and pelleted (500 × g for 3–5 min); DNA was then extracted by using the QIAamp DNA Mini Kit (QIAGEN, Hilden, Germany) according to the manufacturer’s cell protocol. DNA from human tissue specimens was extracted from frozen or deparaffinized formalin-fixed paraffin-embedded (FFPE) sections (5 µm thick) by using the QIAamp DNA Mini Kit according to the manufacturer’s tissue protocol. Overnight proteinase K digestion was extended 3–6 h to result in complete digestion, free of visible tissue particles. Extracted DNA quality was confirmed by amplification of a housekeeping gene sequence: human glyceraldehyde-3-phosphate dehydrogenase (GAPDH) for human, rhesus monkey, baboon, and bat material; murine GAPDH for mouse and rat cell lines; and bovine GAPDH for bovine, ovine, and feline cell lines (Table 4). Molecular contamination of extracted human DNA by BLV control DNA was monitored by using sheep-specific primers for the FLK cell line and plasmid vector primers for the C72/taxNeo line (bovine mammary epithelial cells stably transfected with BLV tax gene) (12), the only 2 BLV-containing cell lines previously grown in the laboratory in which testing was conducted.
For nested L-PCR, extracted DNA (0.85 µg) was added to 50 μL of PCR mix (2.0 mmol/L MgCl2, 0.2 mmol/L dNTPs, 0.025 U/µL Taq polymerase [all from Promega, Madison, WI, USA], and 0.2 μmol/L outer primers for each BLV gene [Table 3]) in Hot Start Micro 50 tubes (MolecularBio Products, San Diego, CA, USA). For the second (nested) round of amplification, 2 μL of the first-round PCR product was added to a new tube of the same reaction mix with inner primers for the corresponding genome region (Table 3). Cycling conditions are given in Table 3. Reaction mix and conditions, including MgCl2 concentrations, were optimized by using the BLV-positive FLK fibroblasts and BLV-negative bat lung fibroblasts (Tb1Lu). Sensitivity of the nested L-PCR was determined by using the housekeeping gene GAPDH, which occurs in humans as 1 copy/cell (13). DNA was extracted from 1 million cells of the human cell line MCF7 (Table 1) and serially diluted to an end point equivalent of 1 cell. Nested L-PCR performed on each dilution established the sensitivity at ≤10 copies.
L-PCR, which requires initial tissue digestion to extract DNA, is prone to molecular contamination. We reduced this probability by separating PCR procedure steps into 3 locations: 1) preparation of PCR mixes in a dedicated, locked, DNA-free room with entrance and ventilation system separate from the main laboratory and with dedicated laminar flow hood, small equipment, plasticware, and reagents; 2) addition of template DNA in a different room in a small hood dedicated to specimen handling, with DNA easily decontaminated by UV light and DNA decontamination solution (RNase AWAY; Molecular BioProducts, San Diego, CA, USA) applied to equipment and surfaces before and after handling each specimen; and 3) processing and addition of positive control samples last, and only in a fume hood (velocity = 127 fpm) vented to atmospheric air, thus preventing contaminating aerosols from entering the general work area.
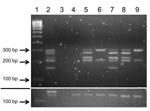
For sequencing, BLV amplicons obtained by nested L-PCR were separated by electrophoreses in a 1% agarose gel at 100 V, excised from the gel, and cleaned by using the Zymoclean Gel DNA Recovery Kit (Zymo Research, Irvine, CA, USA). The requested sample of 100 ng of DNA in 12 µL water and 1 μL of the 5 μmol/L sequencing primer stock solution of the inner primer pair for each genome region (Table 3) was submitted to the University of California, Berkeley, DNA Sequencing Facility for direct sequencing. For tax, inner primer 2 was used to obtain a longer product. DNA for sequencing was obtained from 2–5 amplifications and was sequenced at least once in forward (5′) and reverse (3′) directions. Sequences were checked against corresponding electropherograms. Variations from the reference sequence (GenBank accession no. EF600696) were considered valid only if they matched in both forward and reverse directions. Targeted sequences were relatively short because formalin causes DNA breaks, making it difficult to obtain sequences of >130 nt (14).
Nested in situ PCR (IS-PCR), adapted from Nuovo (15), was used to identify which cell types within tissues were BLV positive. Thick suspensions of detached, rinsed control cells were smeared on enhanced adherence glass microscope slides, air dried, and fixed for 16–18 h with 10% buffered neutral formalin. To enhance entry of PCR mix into cells, samples were made permeable by digestion with 2 mg/mL pepsin in 0.1 N HCl (40–80 min for tissue sections; 20 min for control cell smears), pepsin inactivation solution (100 mmol/L Tris-HCl, 100 mmol/L NaCl, pH 7.4) applied for 1 min, followed by a DPBS rinse and 5 min in absolute ethanol. Samples, run in duplicate, were surrounded with a 15 × 15 mm frame seal chamber (Bio-Rad, Hercules, CA, USA); 60 μL of PCR mix were then placed into the chamber, and the plastic cover was sealed over the frame. The PCR mixture was 4.0 mmol/L MgCl2, 0.4 mmol/L dNTPs, 1 μmol/L primers (Operon Biotechnologies, Huntsville, AL, USA), 0.06% bovine serum albumin, 8 μmol/L digoxigenin-11-dUTP (dig) (Hoffman-La Roche, Basel, Switzerland), and 0.053 U/μL Amplitaq Gold DNA Polymerase (Applied Biosystems, Foster City, CA, USA), a Taq polymerase activated only at ≥92°C, designed to reduce false positives from nonspecific DNA repair by Taq at cooler temperatures (15). Primers were the same as those used for L-PCR (Table 3). Slides were placed into an IS-PCR machine (Hybaid Thermo OmniSlide; Cambridge Biosystems, Cambridge, UK) for amplification. After each round, covers and chambers were removed, and slides were rinsed in DPBS. After the second round, endogenous peroxidase was quenched 30 min in 3% H2O2 in methanol. Label incorporated into PCR products was detected by anti-dig antibodies in an avidin-biotin-immunoperoxidase reaction (Hoffman-La Roche). The chromagen was diaminobenzidine. Outcome measurement was a semiquantitative judgment of color density of cells: 1+, light tan; 2+, medium tan; 3+, dark brown; 4+, almost black. Ratings of ≥2+ were considered positive. No donor information or results of L-PCR were available at the time of slide evaluation.
Initially, the BLV-positive control cell line (FLK) and the BLV-negative cell line (Tb1Lu) were used to optimize the reaction and ensure no false-positive reaction in the negative cell line. Control testing was run simultaneously with each batch of human tissue assays: 1) positive control, a smear of BLV-positive cells (FLK cell line) reacted with complete PCR mix; 2) negative controls, a smear of FLK cells and an adjacent serial section of each specimen reacted with PCR mix minus primers, to rule out false-positive reactions unique to each tissue resulting from unquenched endogenous peroxidase, nonspecific reaction of the sheep antibodies used in the final immunoperoxidase detection, or nonspecific DNA repair by Taq polymerase; 3) permeabilization control for entry of PCR mix into cells, with an adjacent serial section of each tissue reacted with PCR mix different from that for IS-PCR by omission of primers, 4.5 mmol/L concentration for MgCl2, and use of a different Taq polymerase (Promega) that reacts nonspecifically at cooler temperatures (4°C–50°C) to repair DNA. Specimens were scored positive only if mammary epithelial cells were positive and the background control slide (adjacent section) without primers was negative for the corresponding area of mammary epithelial cells. Specimens were scored negative only if the sample and its background control section were negative and the permeabilized control exhibited dig incorporation into cell nuclei resulting from Taq polymerase DNA repair, confirming entry of PCR mix into cells (15).
IHC testing for BLV p24 was performed by using formalin-fixed cell smears and deparaffinized FFPE tissue sections (5 μm) on superadherent microscope slides. Samples were quenched of endogenous peroxidase for 30 min in 3% H2O2 in methanol and rinsed in DPBS; antigens were then unmasked by incubation in citrate buffer (0.1 mmol/L sodium citrate, 0.04 mmol/L citric acid, pH 6.0) for 25 min at 95°C in individual plastic containers to prevent cross-transfer of tissue material. Unmasking was followed by a DPBS rinse, and an avidin-biotin-immunoperoxidase procedure (Vectastain Elite ABC Kit; Vector Laboratories, Burlingame, CA, USA) was performed according to the manufacturer’s instructions. Blocking serum was 1.5% fetal horse serum in DPBS. Primary antibody in blocking serum (1:10) was a hybridoma-produced mouse mAb against BLV p24 (AIDS Research and Reference Reagent Program, National Institute of Allergy and Infectious Diseases; www.aidsreagent.org) (16); specificity was validated by immunoblot reactivity with purified recombinant p24 (7). Secondary antibody was 1.5% horse anti-mouse IgG (Vector) in blocking serum. The chromagen was diaminobenzidine. Negative controls were the BLV-negative control cell line Tb1Lu and adjacent human tissue sections reacted with fresh hybridoma medium (diluted 1:10 in blocking buffer) substituted for primary antibody. The positive control was FLK, which is known to replicate BLV. Outcome measurement was a semiquantitative judgment of color density, as described for IS-PCR.
Human samples were selected from 219 FFPE breast tissue samples; 97 (44%) samples had positive results for BLV by nested IS-PCR that used primers from the most conserved BLV genome region, tax. Five tax-positive samples and 1 tax-negative sample (as a control) were chosen for in-depth molecular analysis on the basis of BLV status and sample size large enough and with enough mammary epithelial cells to yield sufficient material for multiple assays and extensive quality control tests. BLV was detected in DNA extracted from human tissues by using nested L-PCR. All 5 tax-positive samples chosen were positive for the LTR region but showed varying results for other BLV genome regions (Table 5; Figure 1). Sequences of all samples positive for BLV had high identity (E value <1.2) only to BLV nucleotide sequences deposited in GenBank, which suggests that these isolates did not represent some other entity. Variations from the BLV reference sequence were infrequent, and all involved base substitutions (Figure 2).
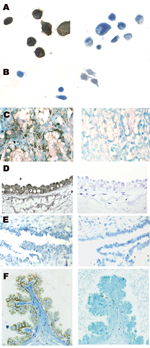
Nested IS-PCR was used to identify the cell type in which the PCR product was localized. Figure 3 shows IS-PCR results obtained by using tax primers for the BLV-negative human sample (no. 143), 1 of the BLV-positive samples (no. 010), the positive and negative cell line controls, and a BLV tax-positive bovine mammary gland sample. The site of the amplified BLV DNA was the secretory mammary epithelium, identified by an anatomic pathologist (H.M.J.). Table 5 summarizes the results for L-PCR and IS-PCR (tax) testing of the 6 tissue samples that were studied in depth.
Validation of the IS-PCR results for a subset of 7 samples (3 tax negative and 4 tax positive) was performed by an independent laboratory by using control cell smears and coded FFPE sections sent from our laboratory with no information about the human patients, tissue pathology, or our results. The detection method was PCR in situ hybridization, in which the PCR occurs in situ but with no label incorporated during amplification (15). Labeled probes specific for the BLV tax region were applied after amplification. The independent laboratory confirmed with 100% concordance the results we obtained by using nested IS-PCR and L-PCR for the tax genome region (Table 6).
Of the 215 human breast tissue samples tested by IHC, 12 (6%) had positive results for BLV p24. The reaction was confined to secretory mammary epithelium, and distribution of both BLV DNA and p24 in mammary epithelial cells was duct and lobule specific; that is, virtually all cells in the duct or lobule would be BLV positive but neighboring ducts or lobules could be completely negative. A BLV p24–positive breast tissue sample is shown in Figure 3, panel F.
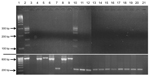
GenBank searches indicated complete identity (E value ≤1.1) of PCR primers (Table 3) only with BLV sequences. L-PCR further substantiated primer specificity (Figure 4; Table 1) by demonstrating no cross-reactivity (amplicon generation) by using template DNA from cell lines harboring representatives of all mammalian and avian oncogenic retroviral subfamilies and human lentiviruses; viruses previously reported in human breast tissues (Epstein-Barr virus, human papillomavirus, mouse mammary tumor virus–like sequences) (17); and human endogenous retrovirus HERV-K (Table 1). Extracted DNA met quality control standards of exhibiting a housekeeping gene (GAPDH; Figure 4). mAb specificity was supported by lack of cross-reactivity with cell lines or by tissues replicating viruses previously identified in human breast tissues (Epstein-Barr virus, human papillomavirus, and mouse mammary tumor virus–like sequences) (17) or that cause chronic human infections (hepatitis B virus, human T-cell leukemia viruses 1 and 2, HIV 1 and 2, human herpesviruses 1 and 2, and cytomegalovirus) (Table 2).
Several methods were used to test for cell and molecular contamination. The no-template-DNA control run with each assay ruled out contamination in commercial DNA isolation columns and PCR reagents; in addition, contamination of human DNA samples by FLK DNA was ruled out by testing human DNA with sheep-specific primers (11), and contamination of FLK with other cell lines, including human, was ruled out by using species-specific primers (11). Sequences from the human isolates also provided strong evidence against contamination; none were exact matches with each other or with any Genbank BLV sequence, which includes the FLK-positive control cell line and bovine specimens previously sequenced in our laboratory. Furthermore, the FLK stock cell line has a signature base substitution (bp 5194; GenBank accession no. EF600696) in the env region; this substitution is unique among sequences deposited in GenBank and was not detected in the human BLV isolates we investigated (Figure 2). The positive results obtained by using in situ methods (IHC and IS-PCR) demonstrating the signal within individual formalin-fixed mammary epithelial cells are further evidence that the positive reactions do not represent contamination of PCR mix. Previous reports have documented that contaminating DNA cannot cause a false-positive reaction with PCR in situ hybridization in FFPE tissues (15). Negative controls (adjacent tissue sections without BLV-specific primers or antibody) run with each in situ assay were all negative, which suggests that positive signals were not false positives resulting from nonspecific reactions.
During the 4 decades since BLV was identified in cattle, there has been considerable interest in determining whether humans could become infected with BLV, especially because cattle are a major food source. As in early serologic studies, previous cellular and molecular studies reported no evidence of human infection with BLV. An explanation for some of the negative findings (18–22) could be that sufficiently sensitive reagents and techniques such as PCR and sequencing were not available when the studies were conducted. Most previous studies also focused on leukocytes, the cell type involved in bovine leukemia/lymphoma (18–27). We focused instead on mammary epithelium, in which we had detected BLV DNA and protein in cattle (10). In situ techniques (i.e., IS-PCR, PCR in situ hybridization, and IHC) enabled confirmation that BLV was localized within mammary epithelial cells.
Evidence for BLV DNA and protein in humans is not surprising. Many viruses, including those that are oncogenic, are known to cross species naturally, and most microbial species pathogenic in humans are speculated to have had a animal origin at some point in human evolution. Once in the human population, most zoonotic viruses can be transmitted among humans, a process that poses the most serious threat to human health (28). BLV is known to cross species readily; the virus infects capybara, zebus, and water buffaloes naturally and sheep, goats, pigs, rabbits, rats, and chickens experimentally (1). Human cells (fibroblasts) are susceptible to infection with BLV in vitro (29).
The lack of gag, pol, and env sequences in some of the BLV-positive panel samples and the presence of LTR and tax sequences in all of them is consistent with results reported for the closely related HTLV-1 (30). Deletions in gag, pol, and env were observed in HTLV-1 isolates from 25.7%–56% of adult T-cell leukemia patients, and frequency increased with clinical progression of leukemia (30). Such deletions are postulated to be advantageous to the virus by enabling escape from immune surveillance. In contrast, the LTR and tax regions of HTLV-1 are highly conserved. Analogous results were observed in specimens from cattle infected with BLV; deletions involving parts of gag and env and all of pol were frequent (2).
Overall, the human BLV isolates differed from the reference sequence by only a few base substitutions, a finding that fits well with the biology of deltaretroviruses, which have infrequent interhost transmission and remain largely latent within the host, probably as a strategy to escape the host’s immune response (31). Infectivity of deltaretroviruses occurs primarily by cell–cell contact, not by extracellular virions (32), and BLV virions have not been found in the peripheral blood of infected cattle (2). In our study, evidence of the capsid protein p24 was infrequent (12/215 [6%]) but consistent with the concept that, in some humans, BLV could be replicating.
Mutation rate tends to be lower for latent viruses than for viruses that escape immune attack by rapidly mutating to outpace the host’s immune response (e.g., HIV [2,31]). Four of the 7 LTR base substitutions we observed (in 3 human BLV isolates) occurred in the PU box, a short (12 bp) protein-binding region that, when mutated, decreases basal gene expression (2,33). This expression would facilitate evasion of the host’s immune response and would thus be likely to be selected for during virus evolution. Transcription in BLV-infected bovine lymphocytes is rare, identified in only ≈1/50,000 peripheral blood cells (2).
In summary, multiple lines of evidence support the conclusion that the BLV DNA and protein we found are more likely to represent the in vivo presence of BLV in humans than to represent some other virus, molecular laboratory contamination, or an artifactual nonspecific reaction. In view of the potential public health implications of BLV in humans, future research should address how humans acquire BLV infection, how frequently BLV infection occurs in different populations, and whether the virus is associated with human disease.
Dr Buehring is professor of virology in the Infectious Diseases and Vaccinology Division, School of Public Health, University of California, Berkeley. Her research interest is BLV, including its presence in the mammary gland of cows, hormone responsiveness to glucocorticoids, and the effects of its infection in humans.
Acknowledgments
We thank Gary Firestone for providing the GR cell line; Kathryn Radke for the FLK, Bat2Cl6, and Tb1Lu cell lines; Carl Hanson for the formalin-fixed HIV- and HTLV-infected cells; Feng Wang-Johanning for the HERV-K DNA; and Craig Meyers for the formalin-fixed keratinocyte raft cultures. We also thank Dr Firestone and Fenyong Liu for their critical reviews of the manuscript and Christina Di Loreto and Michael Van Ness for assistance with photography.
This research was supported by the following grants: California Breast Cancer Research Program of the University of California (6PB-0075); US Army Medical Research and Materiel Command Breast Cancer Research Program (DAMD 17-02-1-0320); and grants from the Dr. Susan Love Research Foundation and the Avon Foundation.
References
- Schwartz I, Levy D. Pathobiology of bovine leukemia virus. Vet Res. 1994;25:521–36 .PubMedGoogle Scholar
- Gillet N, Florins A, Boxus M, Burteau C, Nigro A, Vandermeers F, Mechanisms of leukemogenesis induced by bovine leukemia virus: prospects for novel anti-retroviral therapies in humans. Retrovirology. 2007;4:18–50 . DOIPubMedGoogle Scholar
- US Department of Agriculture Animal and Plant Health Services. Info sheet: bovine leukosis virus (BLV) in U.S. beef cattle. February 1999 [cited 2013 Jan 23]. http://www.aphis.usda.gov/animal_health/nahms/beefcowcalf/downloads/beef97/Beef97_is_BLV.pdf
- US Department of Agriculture Animal and Plant Health Services, Veterinary Services, Center for Epidemiology and Animal Health. Info sheet: bovine leukosis virus (BLV) on U.S. dairy operations, 2007. October 2008 [cited 2013 Aug 20]. http://www.aphis.usda.gov/animal_health/nahms/dairy/downloads/dairy07/Dairy07_is_BLV.pdf
- Burridge MJ. The zoonotic potential of bovine leukemia virus. Vet Res Commun. 1981;5:117–26. DOIPubMedGoogle Scholar
- Choi KY, Liu RB, Buehring GC. Relative sensitivity and specificity of agar gel immunodiffusion, enzyme immunosorbent assay, and immunoblotting for detection of anti-bovine leukemia virus antibodies in cattle. J Virol Methods. 2002;104:33–9 . DOIPubMedGoogle Scholar
- Buehring GC, Philpott SM, Choi KY. Humans have antibodies reactive with bovine leukemia virus. AIDS Res Hum Retroviruses. 2003;19:1105–13 . DOIPubMedGoogle Scholar
- Baumgartener L, Olson C, Onuma M. Effect of pasteurization and heat treatment on bovine leukemia virus. J Am Vet Med Assoc. 1976;169:1189–91 .PubMedGoogle Scholar
- Chung YS, Prior HC, Duffy PF, Rogers RJ, Macenzie AR. The effect of pasteurization on bovine leucosis virus–infected milk. Aust Vet J. 1986;63:379–80. DOIPubMedGoogle Scholar
- Buehring GC, Kramme PM, Schultz RD. Evidence for bovine leukemia virus in the mammary epithelial cells of infected cows. Lab Invest. 1994;71:359–65 .PubMedGoogle Scholar
- Cooper JK, Sykes G, King S, Cottril K, Ivanova NV, Hanner R, Species identification in cell culture: a two-pronged molecular approach. In Vitro Cell Dev Biol Anim. 2007;43:344–51 and. DOIPubMedGoogle Scholar
- Motton DD, Buehring GC. Bovine leukemia virus alters growth properties and casein synthesis in mammary epithelial cells. J Dairy Sci. 2003;86:2826–38. DOIPubMedGoogle Scholar
- Ercolani L, Florence B, Denaro M, Alexander M. Isolation and complete sequence of a functional human glyceraldehyde-3-phosphate dehydrogenase gene. J Biol Chem. 1988;263:15335–41 .PubMedGoogle Scholar
- Gillio-Tos A, De Marco L, Fiano V, Garcia-Bragado F, Dikshit R, Boffetta P, Efficient DNA extraction from 25-year-old paraffin-embedded tissues: study of 365 samples. Pathology. 2007;39:345–8. DOIPubMedGoogle Scholar
- Nuovo GJ. PCR in situ hybridization: protocols and applications. Philadelphia: Lippincott-Raven; 1996. pp. 193–244.
- Kramme PM, Thomas CB, Schultz RD. Temporal stability of the virus load of cattle infected with bovine leukemia virus. Vet Immunol Immunopathol. 1995;45:347–54. DOIPubMedGoogle Scholar
- Joshi D, Buehring GC. Are viruses associated with breast cancer? Scrutinizing the molecular evidence. Breast Cancer Res Treat. 2012;135:1–15 . DOIPubMedGoogle Scholar
- Ressang AA. Examination for evidence of infection of humans with bovine leukemia virus [in Dutch]. Tijdschr Diergeneeskd. 1977;102:333–5 .PubMedGoogle Scholar
- Olson C, Driscoll DM. Bovine leukosis: investigation of risk for man. J Am Vet Med Assoc. 1978;173:1470–2.
- Kettmann R, Burny A, Cleuter Y, Ghysdael J, Mammerickx M. Distribution of bovine leukemia virus proviral sequences in tissues of bovine, ovine, and human origin. Ann Rech Vet. 1978;9:837–44 .PubMedGoogle Scholar
- Thiry L, Sprecher-Goldberger S, Jacquemin P, Cogniaux J, Burny A, Portetelle D, Bovine leukemia virus–related antigens in lymphocyte cultures infected with AIDS-associated viruses. Science. 1985;227:1482–4 . DOIPubMedGoogle Scholar
- Bender AP, Robison LL, Kashmiri SV, McClain K, Woods WG, Smithson WA, No involvement of bovine leukemia virus in childhood acute lymphoblastic leukemia and non-Hodgkin’s lymphoma. Cancer Res. 1988;48:2919–22 .PubMedGoogle Scholar
- Chen IS, Haislip AM, Myers LW, Ellison GW, Merrrill JE. Failure to detect human T-cell leukemia virus–related sequences in multiple sclerosis blood. Arch Neurol. 1990;47:1064–5 . DOIPubMedGoogle Scholar
- Heneine W, Woods TC, Sinha SD, Khan AS, Chapman LE, Schonberger LB, Lack of evidence for infection with known human and animal retroviruses in patients with chronic fatigue syndrome. Clin Infect Dis. 1994;18(Suppl 1):S121–5 . DOIPubMedGoogle Scholar
- Perzova RN, Loughran TP, Dube S, Ferrer J, Esteban E, Poiesz BJ. Lack of BLV and PTLV DNA sequences in the majority of patients with large granular lymphocyte leukaemia. Br J Haematol. 2000;109:64–70 . DOIPubMedGoogle Scholar
- Lee J, Kim Y, Kang CS, Cho DH, Shin DH, Yum YN, Investigation of the bovine leukemia virus proviral DNA in human leukemia and lung cancers in Korea. J Korean Med Sci. 2005;20:603–6 . DOIPubMedGoogle Scholar
- Burmeister T, Schwartz S, Hummel M, Hoelzer D, Thiel E. No genetic evidence for involvement of Deltaretroviruses in adult patients with precursor and mature T-cell neoplasms. Retrovirology. 2007;4:11–7 . DOIPubMedGoogle Scholar
- Christou L. The global burden of bacterial and zoonotic infections. Clin Microbiol Infect. 2011;17:326–30. DOIPubMedGoogle Scholar
- Diglio CA, Ferrer JF. Induction of syncytia by the bovine C-type leukemia virus. Cancer Res. 1976;36:1056–67 .PubMedGoogle Scholar
- Kamihira S, Sugahara K, Tsuruda K, Uemura A, Akamatsu N, Nagai H, Proviral status of HTLV-1 integrated into the host genomic DNA of adult T-cell leukemia cells. Clin Lab Haematol. 2005;27:235–41. DOIPubMedGoogle Scholar
- Duffy S, Shackleton LA, Holmes EC. Rates of evolutionary change in viruses: pattern and determinants. Nat Rev Genet. 2008;9:267–76. DOIPubMedGoogle Scholar
- Igakura T, Stinchcombe JC, Goon PK, Taylor GP, Griffiths GM, Tanaka Y, Spread of HTLV between lymphocytes by virus-induced polarization of the cytoskeleton. Science. 2003;299:1713–6 . DOIPubMedGoogle Scholar
- Zhao X, Jimenez C, Sentsui H, Buehring GC. Sequence polymorphisms in the long terminal repeat of bovine leukemia virus: Evidence for selection pressures in regulatory sequences. Virus Res. 2007;124:113–24. DOIPubMedGoogle Scholar
Figures
Tables
Cite This Article1Current affiliation: Texas A&M Health Science Center College of Medicine, College Station, Texas, USA.
Table of Contents – Volume 20, Number 5—May 2014
| EID Search Options |
|---|
|
|
|
|
|
|

Please use the form below to submit correspondence to the authors or contact them at the following address:
Gertrude Case Buehring, Division of Infectious Diseases and Vaccinology, School of Public Health, 16 Barker Hall, University of California, Berkeley, CA 94720-7354, USA:Gertrude Case Buehring, Division of Infectious Diseases and Vaccinology, School of Public Health, 16 Barker Hall, University of California, Berkeley, CA 94720-7354, USA:Gertrude Case Buehring, Division of Infectious Diseases and Vaccinology, School of Public Health, 16 Barker Hall, University of California, Berkeley, CA 94720-7354, USA:
Top