Yellow Fever Vaccine & Malaria Prevention Information, by Country
CDC Yellow Book 2024
Preparing International TravelersThe following pages present country-specific information on yellow fever (YF) vaccine requirements and recommendations, and malaria transmission information and prevention recommendations. Country-specific maps are included to aid in interpreting the information. The information in this chapter was accurate at the time of publication; however, it is subject to change at any time due to changes in disease transmission or, in the case of YF, changing entry requirements for travelers. Updated information reflecting changes since publication can be found in the online version of this book and on the Centers for Disease Control and Prevention (CDC) Travelers’ Health website. Recommendations for prevention of other travel-associated illnesses can also be found on the CDC Travelers’ Health website.
Yellow Fever Vaccine
Entry Requirements
Entry requirements for proof of YF vaccination under the International Health Regulations (IHR) differ from CDC’s YF vaccination recommendations. Under the IHR, countries are permitted to establish YF vaccine entry requirements to prevent the importation and transmission of YF virus within their boundaries. Certain countries require proof of vaccination from travelers arriving from all countries (Table 5-25); some countries require proof of vaccination only for travelers above a certain age coming from countries with risk for YF virus transmission. The World Health Organization (WHO) defines areas with risk for YF virus transmission as countries or areas where YF virus activity has been reported currently or in the past, and where vectors and animal reservoirs exist.
Unless issued a medical waiver by a yellow fever vaccine provider, travelers must comply with entry requirements for proof of vaccination against YF.
WHO publishes a list of YF vaccine country entry requirements and recommendations for international travelers approximately annually. But because entry requirements are subject to change at any time, health care professionals and travelers should refer to the online version of this book and the CDC Travelers’ Health website for any updates before departure.
CDC Recommendations
CDC’s YF vaccine recommendations are guidance intended to protect travelers from acquiring YF virus infections during international travel. These recommendations are based on a classification system for destination-specific risk for YF virus transmission: endemic, transitional, low potential for exposure, and no risk (Table 2-08). CDC recommends YF vaccination for travel to areas classified as having endemic or transitional risk (Maps 5-10 and 5-11). Because of changes in YF virus circulation, however, recommendations can change; therefore, before departure, travelers and clinicians should check CDC’s destination pages for up-to-date YF vaccine information.
Duration of Protection
In 2015, the US Advisory Committee on Immunization Practices published a recommendation that 1 dose of YF vaccine provides long-lasting protection and is adequate for most travelers. The recommendation also identifies specific groups of travelers who should receive additional doses, and others for whom additional doses should be considered (see Sec. 5, Part 2, Ch. 26, Yellow Fever). In July 2016, WHO officially amended the IHR to stipulate that a completed International Certificate of Vaccination or Prophylaxis is valid for the lifetime of the vaccinee, and YF vaccine booster doses are not necessary. Moreover, countries cannot require proof of revaccination (booster) against YF as a condition of entry, even if the traveler’s last vaccination was >10 years ago.
Ultimately, when deciding whether to vaccinate travelers, clinicians should take into account destination-specific risks for YF virus infection, and individual risk factors (e.g., age, immune status) for serious YF vaccine–associated adverse events, in the context of the entry requirements. See Sec. 5, Part 2, Ch. 26, Yellow Fever, for a full discussion of YF disease and vaccination guidance.
Table 2-08 Yellow fever (YF) vaccine recommendation categories1
| YF VACCINE RECOMMENDATION CATEGORY | RATIONALE |
|---|---|
| Recommended | Vaccination recommended for all travelers ≥9 months old going to areas with endemic or transitional YF risk, as determined by persistent or periodic YF virus transmission |
| Generally not recommended | Vaccination generally not recommended for travel to areas where the potential for YF virus exposure is low, as determined by absence of reports of human YF and past evidence suggestive of only low levels of YF virus transmission Vaccination might be considered for a small subset of travelers at increased risk for exposure to YF virus due to prolonged travel, heavy mosquito exposure, or inability to avoid mosquito bites |
| Not recommended | Vaccination not recommended for travel to areas where there is no risk for YF virus transmission, as determined by absence of past or present evidence of YF virus circulation in the area or environmental conditions not conducive to YF virus transmission |
Malaria Prevention
The following recommendations to protect travelers from malaria were developed using the best available data from multiple sources. Countries are not required to submit malaria surveillance data to CDC. On an ongoing basis, CDC actively solicits data from multiple sources, including WHO (main and regional offices); national malaria control programs; international organizations; CDC overseas offices; US military; academic, research, and aid organizations; and the published scientific literature. The reliability and accuracy of those data are also assessed.
If the information is available, trends in malaria incidence and other data are considered in the context of malaria control activities within a given country or other mitigating factors (e.g., natural disasters, wars, the coronavirus disease 2019 pandemic) that can affect the ability to control malaria or accurately count and report it. Factors such as the volume of travel to that country and the number of acquired cases reported in the US surveillance system are also examined. In developing its recommendations, CDC considers areas within countries where malaria transmission occurs, substantial occurrences of antimalarial drug resistance, the proportions of species present, and the available malaria prophylaxis options.
Clinicians should use these recommendations in conjunction with an individual risk assessment and consider not only the destination but also the detailed itinerary, including specific cities, types of accommodations, season, and style of travel, as well as special health conditions (e.g., pregnancy). Several medications are available for malaria prophylaxis. When deciding which drug to use, consider the itinerary and length of trip, travelers’ previous adverse reactions to antimalarials, drug allergies, medical history, and drug costs. For a thorough discussion of malaria and guidance for prophylaxis, see Sec. 5, Part 3, Ch. 16, Malaria.
Brazil
Yellow Fever Vaccine
Entry requirements: None
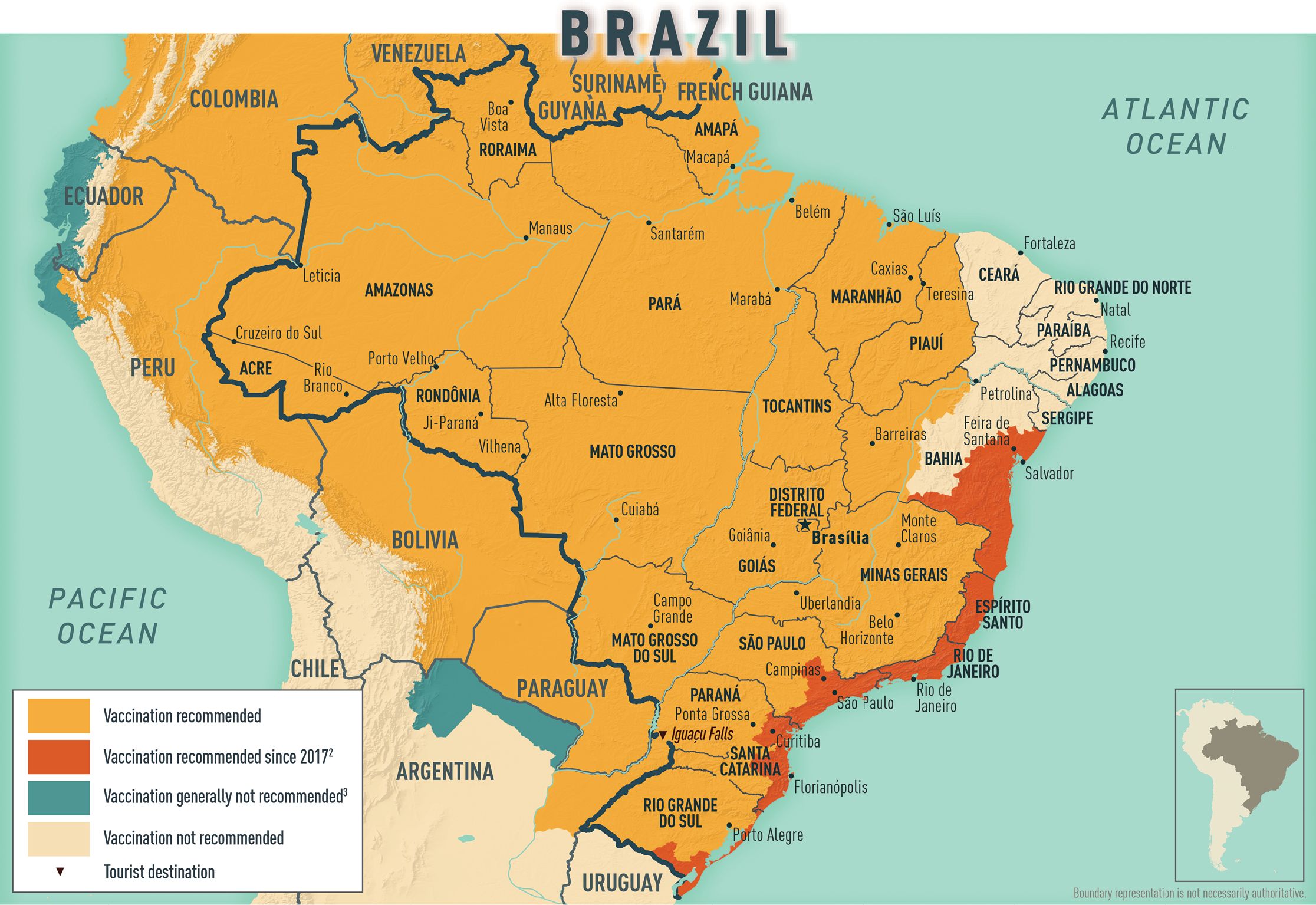
CDC recommendations: Recommended for travelers ≥9 months old going to the states of Acre, Amapá, Amazonas, Distrito Federal (including the capital city, Brasília), Espírito Santo,* Goiás, Maranhão, Mato Grosso, Mato Grosso do Sul, Minas Gerais, Pará, Paraná,* Piauí, Rio de Janeiro (including the city of Rio de Janeiro and all coastal islands),* Rio Grande do Sul,* Rondônia, Roraima, Santa Catarina,* São Paulo (including the city of São Paulo and all coastal islands),* Tocantins, and designated areas of Bahia*. Vaccination is also recommended for travelers going to Iguaçu Falls. Not recommended for travel limited to any areas not listed above, including the cities of Fortaleza and Recife *In 2017, in response to a large YF outbreak in multiple eastern states, CDC expanded its vaccination recommendations for travelers going to Brazil. The expanded YF vaccination recommendations for these states are preliminary. For updates, refer to the CDC Travelers’ Health website.
Malaria Prevention
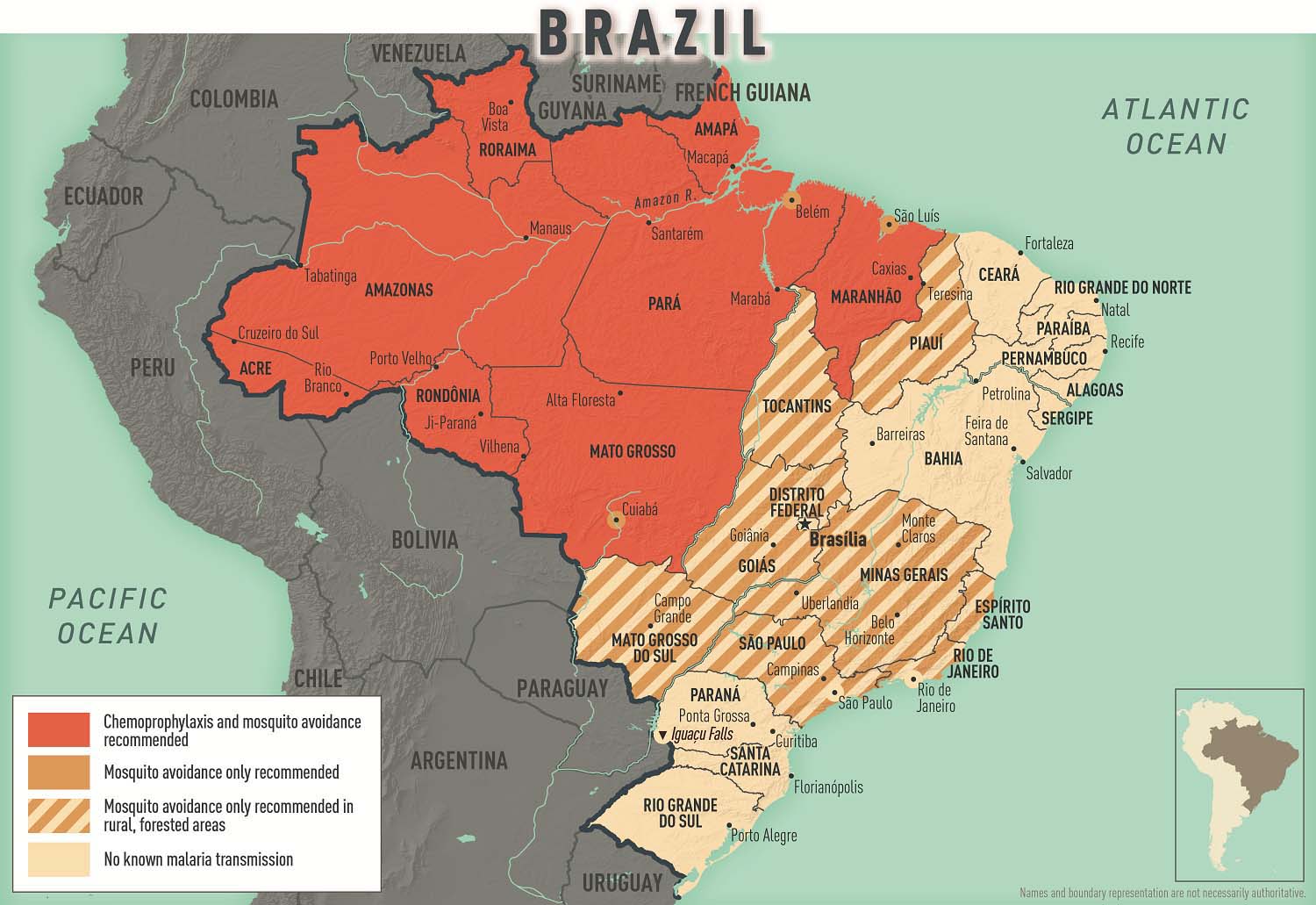
Transmission areas
- All areas in the states of Acre, Amapá, Amazonas, Rondônia, and Roraima
- Present in the states of Maranhão, Mato Grosso, and Pará, but rare cases in their capital cities (São Luis [capital of Maranhão], Cuiabá [capital of Mato Grosso], Belém [capital of Pará])
- Rare cases and sporadic foci of transmission in rural and forested areas in the states of Espírito Santo, Goiás, Minas Gerais, Mato Grosso do Sul, Piauí, Rio de Janeiro, São Paolo, and Tocantins
- No malaria transmission in the cities of Brasília (the capital), Rio de Janeiro, or São Paolo
- No malaria transmission at Iguaçu Falls
Drug resistance2
- Chloroquine
Species
- P. vivax (90%)
- P. falciparum (10%)
Recommended chemoprophylaxis
- Atovaquone-proguanil, doxycycline, mefloquine, tafenoquine3
- Areas with rare cases: No chemoprophylaxis recommended (insect bite precautions and mosquito avoidance only)4
Related Maps
- Map 2-03 Yellow fever vaccine recommendations for Brazil & neighboring countries
- Map 2-04 Malaria prevention in Brazil
Other Vaccines to Consider
See Health Information for Travelers to Brazil
Map 2-03 Yellow fever vaccine recommendations for Brazil & neighboring countries1
Footnotes
Yellow Fever Vaccine
1Current as of November 2022. This is an update of the 2010 map created by the Informal WHO Working Group on the Geographic Risk of Yellow Fever.
Malaria Prevention
2Refers to Plasmodium falciparum malaria, unless otherwise noted.
3Tafenoquine can cause potentially life-threatening hemolysis in people with glucose-6-phosphate-dehydrogenase (G6PD) deficiency. Rule out G6PD deficiency with a quantitative laboratory test before prescribing tafenoquine to patients.
4Mosquito avoidance includes applying topical mosquito repellant, sleeping under an insecticide-treated mosquito net, and wearing protective clothing (e.g., long pants and socks, long-sleeve shirt). For additional details on insect bite precautions, see Sec. 4, Ch. 6, Mosquitoes, Ticks & Other Arthropods.
5Primaquine can cause potentially life-threatening hemolysis in people with G6PD deficiency. Rule out G6PD deficiency with a quantitative laboratory test before prescribing primaquine to patients.
6P. knowlesi is a malaria species with a simian (macaque) host. Human cases have been reported from most countries in Southwest Asia and are associated with activities in forest or forest-fringe areas. P. knowlesi has no known resistance to antimalarials.
Yellow Fever Maps
1Current as of November 2022. This is an update of the 2010 map created by the Informal WHO Working Group on the Geographic Risk of Yellow Fever.
2In 2017, the Centers for Disease Control and Prevention (CDC) expanded its YF vaccination recommendations for travelers going to Brazil because of a large YF outbreak in multiple states in that country. Please refer to the CDC Travelers’ Health website for more information and updated recommendations.
3YF vaccination is generally not recommended for travel to areas where the potential for YF virus exposure is low. Vaccination might be considered, however, for a small subset of travelers going to these areas who are at increased risk for exposure to YF virus due to prolonged travel, heavy exposure to mosquitoes, or inability to avoid mosquito bites. Factors to consider when deciding whether to vaccinate a traveler include destination-specific and travel-associated risks for YF virus infection; individual, underlying risk factors for having a serious YF vaccine–associated adverse event; and destination entry requirements.
The following authors contributed to the previous version of this chapter: Mark D. Gershman, Emily S. Jentes, Rhett J. Stoney (Yellow Fever) Kathrine R. Tan, Paul M. Arguin (Malaria)